The Frequency of a Magnetic Field Reduces the Viability and Proliferation of Numerous Tumor Cell Lines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Cultures
2.2. Electromagnetic Field Exposure System
2.3. Exposure Conditions
2.4. Metabolic Activity Assay
2.5. Proliferation
2.6. Apoptosis Assay
2.7. Statistical Analysis
3. Results
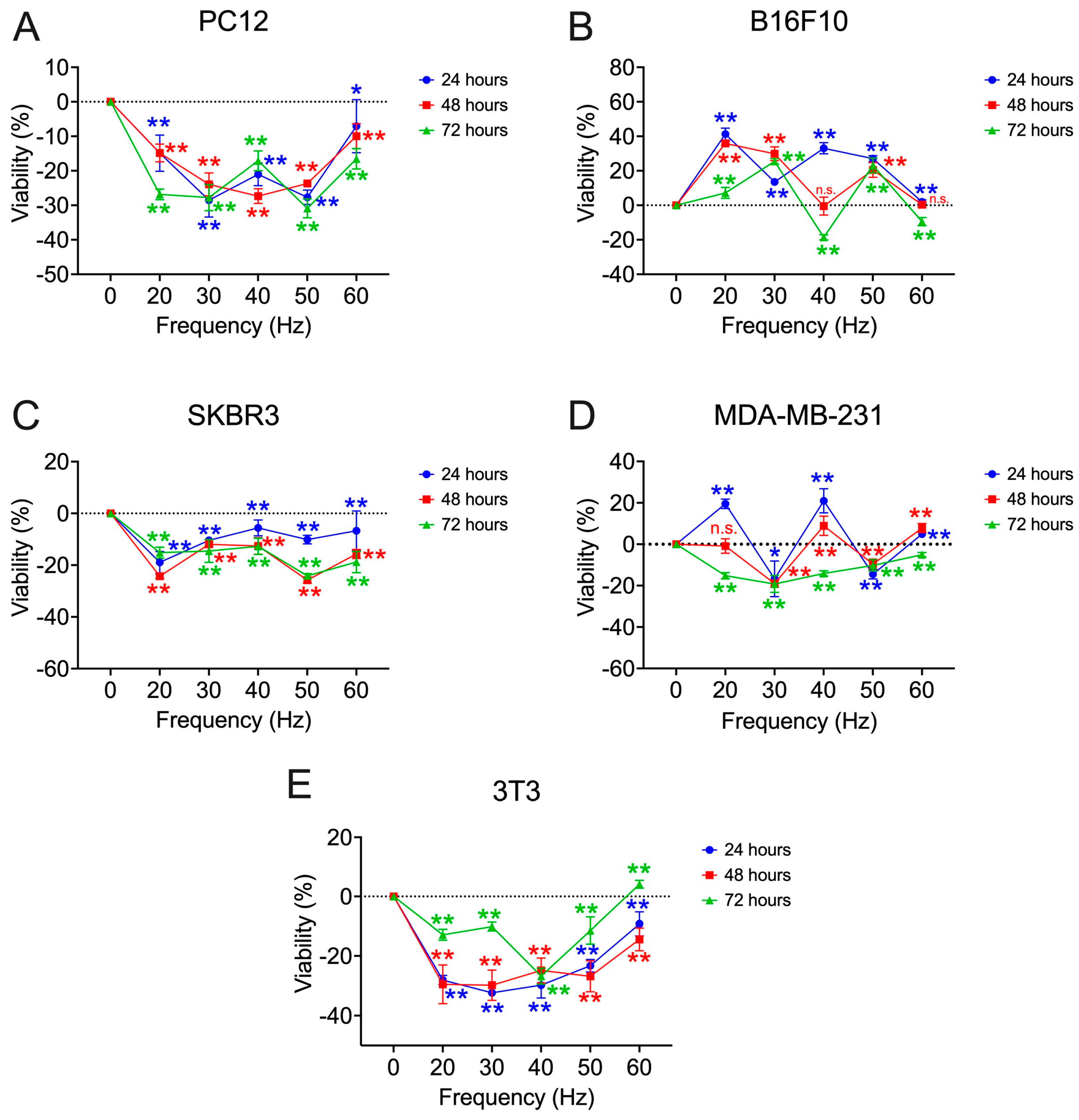
3.1. Viability
3.2. Proliferation
3.3. Apoptosis
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ELF-EMF | Extremely low frequency electromagnetic field |
| DMEM | Dulbecco’s modified Eagle’s medium |
| RPMI | Roswell Park Memorial Institute medium |
| ATCC | American Type Culture Collection |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide |
| DMSO | dimethyl sulfoxide |
References
- Matthews, H.K.; Bertoli, C.; de Bruin, R.A.M. Cell Cycle Control in Cancer. Nat. Rev. Mol. Cell Biol. 2022, 23, 74–88. [Google Scholar] [CrossRef] [PubMed]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Non-Ionizing Radiation, Part 1: Static and Extremely Low-Frequency (ELF) Electric and Magnetic Fields; IARC Working Group on the Evaluation of Carcinogenic Risks to Humans: Lyon, France, 2002; Volume 80, ISBN 9283212800. [Google Scholar]
- Zhang, G.; Liu, X.; Liu, Y.; Zhang, S.; Yu, T.; Chai, X.; He, J.; Yin, D.; Zhang, C. The Effect of Magnetic Fields on Tumor Occurrence and Progression: Recent Advances. Prog. Biophys. Mol. Biol. 2023, 179, 38–50. [Google Scholar] [CrossRef]
- Maffei, M.E. Magnetic Fields and Cancer: Epidemiology, Cellular Biology, and Theranostics. Int. J. Mol. Sci. 2022, 23, 1339. [Google Scholar] [CrossRef]
- Löscher, W.; Liburdy, R.P. Animal and Cellular Studies on Carcinogenic Effects of Low Frequency (50/60-Hz) Magnetic Fields. Mutat. Res./Rev. Mutat. Res. 1998, 410, 185–220. [Google Scholar] [CrossRef]
- Kocaman, A.; Altun, G.; Kaplan, A.A.; Deniz, Ö.G.; Yurt, K.K.; Kaplan, S. Genotoxic and Carcinogenic Effects of Non-Ionizing Electromagnetic Fields. Environ. Res. 2018, 163, 71–79. [Google Scholar] [CrossRef]
- Holmberg, B. Magnetic Fields and Cancer: Animal and Cellular Evidence--an Overview. Env. Health Perspect. 1995, 103, 63–67. [Google Scholar] [CrossRef]
- Kavet, R. EMF and Current Cancer Concepts. Bioelectromagnetics 1996, 17, 339–357. [Google Scholar] [CrossRef]
- Khan, M.W.; Juutilainen, J.; Naarala, J.; Roivainen, P. Residential Extremely Low Frequency Magnetic Fields and Skin Cancer. Occup. Environ. Med. 2022, 79, 49–54. [Google Scholar] [CrossRef]
- Zhang, Y.; Lai, J.; Ruan, G.; Chen, C.; Wang, D.W. Meta-Analysis of Extremely Low Frequency Electromagnetic Fields and Cancer Risk: A Pooled Analysis of Epidemiologic Studies. Environ. Int. 2016, 88, 36–43. [Google Scholar] [CrossRef]
- Huss, A.; Spoerri, A.; Egger, M.; Kromhout, H.; Vermeulen, R. Occupational Extremely Low Frequency Magnetic Fields (ELF-MF) Exposure and Hematolymphopoietic Cancers—Swiss National Cohort Analysis and Updated Meta-Analysis. Environ. Res. 2018, 164, 467–474. [Google Scholar] [CrossRef]
- Seomun, G.; Lee, J.; Park, J. Exposure to Extremely Low-Frequency Magnetic Fields and Childhood Cancer: A Systematic Review and Meta-Analysis. PLoS ONE 2021, 16, e0251628. [Google Scholar] [CrossRef] [PubMed]
- Brabant, C.; Geerinck, A.; Beaudart, C.; Tirelli, E.; Geuzaine, C.; Bruyère, O. Exposure to Magnetic Fields and Childhood Leukemia: A Systematic Review and Meta-Analysis of Case-Control and Cohort Studies. Rev. Environ. Health 2023, 38, 229–253. [Google Scholar] [CrossRef]
- Johansson, O. Disturbance of the Immune System by Electromagnetic Fields—A Potentially Underlying Cause for Cellular Damage and Tissue Repair Reduction Which Could Lead to Disease and Impairment. Pathophysiology 2009, 16, 157–177. [Google Scholar] [CrossRef]
- Destefanis, M.; Viano, M.; Leo, C.; Gervino, G.; Ponzetto, A.; Silvagno, F. Extremely Low Frequency Electromagnetic Fields Affect Proliferation and Mitochondrial Activity of Human Cancer Cell Lines. Int. J. Radiat. Biol. 2015, 91, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Shayeghan, M.; Mehdizadeh, R.; Forouzesh, F.; Sanati, H.; Javidi, M.A.; Madjid Ansari, A. Effect of Extremely Low–Frequency Electromagnetic Field on Apoptosis Induction and Expression of Estrogen Receptor, Progesterone Receptor, and ERBB2 in BT-474 Cells. Iran. J. Breast Dis. 2022, 15, 36–49. [Google Scholar] [CrossRef]
- Wang, M.H.; Chen, K.W.; Ni, D.X.; Fang, H.J.; Jang, L.S.; Chen, C.H. Effect of Extremely Low Frequency Electromagnetic Field Parameters on the Proliferation of Human Breast Cancer. Electromagn. Biol. Med. 2021, 40, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Bergandi, L.; Lucia, U.; Grisolia, G.; Salaroglio, I.C.; Gesmundo, I.; Granata, R.; Borchiellini, R.; Ponzetto, A.; Silvagno, F. Thermomagnetic Resonance Effect of the Extremely Low Frequency Electromagnetic Field on Three-Dimensional Cancer Models. Int. J. Mol. Sci. 2022, 23, 7955. [Google Scholar] [CrossRef]
- Oh, I.R.; Raymundo, B.; Jung, S.A.; Kim, H.J.; Park, J.K.; Kim, C.W. Extremely Low-Frequency Electromagnetic Field Altered PPARγ and CCL2 Levels and Suppressed CD44+/CD24− Breast Cancer Cells Characteristics. Bull. Korean Chem. Soc. 2020, 41, 812–823. [Google Scholar] [CrossRef]
- Nezamtaheri, M.S.; Goliaei, B.; Shariatpanahi, S.P.; Ansari, A.M. Differential Biological Responses of Adherent and Non-Adherent (Cancer and Non-Cancerous) Cells to Variable Extremely Low Frequency Magnetic Fields. Sci. Rep. 2022, 12, 14225. [Google Scholar] [CrossRef]
- Mahmoudinasab, H.; Saadat, M. Short-Term Exposure to 50-Hz Electromagnetic Field and Alterations in NQO1 and NQO2 Expression in MCF-7 Cells. Open Access Maced. J. Med. Sci. 2016, 4, 548–550. [Google Scholar] [CrossRef]
- Mahmoudinasab, H.; Sanie-Jahromi, F.; Saadat, M. Effects of Extremely Low-Frequency Electromagnetic Field on Expression Levels of Some Antioxidant Genes in Human MCF-7 Cells. Mol. Biol. Res. Commun. 2016, 5, 77–85. [Google Scholar] [PubMed]
- Ramazi, S.; Salimian, M.; Allahverdi, A.; Kianamiri, S.; Abdolmaleki, P. Synergistic Cytotoxic Effects of an Extremely Low-Frequency Electromagnetic Field with Doxorubicin on MCF-7 Cell Line. Sci. Rep. 2023, 13, 8844. [Google Scholar] [CrossRef]
- Lazzarini, R.; Eléxpuru-Zabaleta, M.; Piva, F.; Giulietti, M.; Fulgenzi, G.; Tartaglione, M.F.; Zingaretti, L.; Tagliabracci, A.; Valentino, M.; Santarelli, L.; et al. Effects of Extremely Low-Frequency Magnetic Fields on Human MDA-MB-231 Breast Cancer Cells: Proteomic Characterization. Ecotoxicol. Environ. Saf. 2023, 253, 114650. [Google Scholar] [CrossRef]
- Elexpuru-Zabaleta, M.; Lazzarini, R.; Tartaglione, M.F.; Piva, F.; Ciarapica, V.; Marinelli Busilacchi, E.; Poloni, A.; Valentino, M.; Santarelli, L.; Bracci, M. A 50 Hz Magnetic Field Influences the Viability of Breast Cancer Cells 96 h after Exposure. Mol. Biol. Rep. 2023, 50, 1005–1017. [Google Scholar] [CrossRef]
- Xu, A.; Wang, Q.; Lin, T. Low-Frequency Magnetic Fields (Lf-Mfs) Inhibit Proliferation by Triggering Apoptosis and Altering Cell Cycle Distribution in Breast Cancer Cells. Int. J. Mol. Sci. 2020, 21, 2952. [Google Scholar] [CrossRef]
- Shoorche, A.B.; Mohammadkarim, A.; Jadidi, M.; Bahraminasab, M.; Bagheri, H.; Gholami, S. Human Osteosarcoma Cells in Response to ELF-MF: Morphological Remodeling Compared to Cell Proliferation. J. Cancer Res. Ther. 2023, 19, 1311–1315. [Google Scholar] [CrossRef] [PubMed]
- Koziorowska, A.; Romerowicz-Misielak, M.; Sołek, P.; Koziorowski, M. Extremely Low Frequency Variable Electromagnetic Fields Affect Cancer and Noncancerous Cells in Vitro Differently: Preliminary Study. Electromagn. Biol. Med. 2018, 37, 35–42. [Google Scholar] [CrossRef]
- Shahidain, R.; Mullins, R.D.; Sisken, J.E. Calcium Spiking Activity and Baseline Calcium Levels in ROS 17/2.8 Cells Exposed to Extremely Low Frequency Electromagnetic Fields (ELF EMF). Int. J. Radiat. Biol. 2001, 77, 241–248. [Google Scholar] [CrossRef]
- Yuan, L.Q.; Wang, C.; Zhu, K.; Li, H.M.; Gu, W.Z.; Zhou, D.M.; Lai, J.Q.; Zhou, D.; Lv, Y.; Tofani, S.; et al. The Antitumor Effect of Static and Extremely Low Frequency Magnetic Fields against Nephroblastoma and Neuroblastoma. Bioelectromagnetics 2018, 39, 375–385. [Google Scholar] [CrossRef]
- Patruno, A.; Costantini, E.; Ferrone, A.; Pesce, M.; Francesca, D.; Trubiani, O.; Reale, M. Short ELF-EMF Exposure Targets SIRT1/NRF2/HO-1 Signaling in THP-1 Cells. Int. J. Mol. Sci. 2020, 21, 7284. [Google Scholar] [CrossRef]
- Srdjenovic, B.; Mrdjanovic, J.; Galovic, A.J.; Kladar, N.; Bozin, B.; Jurisic, V.; Bogdanovic, G. Effect of ELF-EMF on Antioxidant Status and Micronuclei in K562 Cells and Normal Lymphocytes. Cent. Eur. J. Biol. 2014, 9, 931–940. [Google Scholar] [CrossRef]
- Garip, A.; Akan, Z. Effect of ELF-EMF on Number of Apoptotic Cells; Correlation with Reactive Oxygen Species and HSP. Acta Biol. Hung. 2010, 61, 158–167. [Google Scholar] [CrossRef]
- Patruno, A.; Tabrez, S.; Pesce, M.; Shakil, S.; Kamal, M.A.; Reale, M. Effects of Extremely Low Frequency Electromagnetic Field (ELF-EMF) on Catalase, Cytochrome P450 and Nitric Oxide Synthase in Erythro-Leukemic Cells. Life Sci. 2015, 121, 117–123. [Google Scholar] [CrossRef] [PubMed]
- AyeŞ, I.G.; Zafer, A.; Şule, O.; IŞil, I.T.; Kalkan, T. Differentiation of K562 Cells under ELF-EMF Applied at Different Time Courses. Electromagn. Biol. Med. 2010, 29, 122–130. [Google Scholar] [CrossRef]
- Wolf, F.I.; Torsello, A.; Tedesco, B.; Fasanella, S.; Boninsegna, A.; D’Ascenzo, M.; Grassi, C.; Azzena, G.B.; Cittadini, A. 50-Hz Extremely Low Frequency Electromagnetic Fields Enhance Cell Proliferation and DNA Damage: Possible Involvement of a Redox Mechanism. Biochim. Biophys. Acta Mol. Cell Res. 2005, 1743, 120–129. [Google Scholar] [CrossRef]
- Ruiz Gómez, M. 25 Hz Electromagnetic Field Exposure Has No Effect on Cell Cycle Distribution and Apoptosis in U-937 and HCA-2/1cch Cells. Bioelectrochemistry 2001, 53, 137–140. [Google Scholar] [CrossRef]
- Chen, L.; Xia, Y.; Lu, J.; Xie, Q.; Ye, A.; Sun, W. A 50-Hz Magnetic-Field Exposure Promotes Human Amniotic Cells Proliferation via SphK–S1P–S1PR Cascade Mediated ERK Signaling Pathway. Ecotoxicol. Environ. Saf. 2020, 194, 110407. [Google Scholar] [CrossRef]
- Tang, J.Y.; Yeh, T.W.; Huang, Y.T.; Wang, M.H.; Jang, L.S. Effects of Extremely Low-Frequency Electromagnetic Fields on B16F10 Cancer Cells. Electromagn. Biol. Med. 2019, 38, 149–157. [Google Scholar] [CrossRef]
- Maleki, N.S.M.; Entezari, M.; Abdi, S.; Tekiyehmaroof, N. Electromagnetic Fields Change the Expression of Suppressor of Cytokine Signaling 3 (SOCS3) and Cathepsin L2 (CTSL2) Genes in Adenocarcinoma Gastric (AGS) Cell Line. Int. J. Cancer Manag. 2022, 15, 117270. [Google Scholar] [CrossRef]
- Mansoury, F.; Babaei, N.; Abdi, S.; Entezari, M.; Doosti, A. Extremely Low Frequency Magnetic Fields Induce MTOR and Hsa_Circ_100338 Expression Changes in Gastric Cancer and Normal Fibroblast Cell Lines. Cell J. 2022, 24, 364–369. [Google Scholar] [CrossRef]
- Nieminen, V.; Juntunen, M.; Naarala, J.; Luukkonen, J. Static or 50 Hz Magnetic Fields at 100 ΜT Do Not Modify the Clonogenic Survival of Doxorubicin-Treated MCF-7 Cancer Cells. Bioelectrochemistry 2022, 147, 108196. [Google Scholar] [CrossRef]
- Yuan, L.-Q.; Wang, C.; Lu, D.-F.; Zhao, X.-D.; Tan, L.-H.; Chen, X. Induction of Apoptosis and Ferroptosis by a Tumor Suppressing Magnetic Field through ROS-Mediated DNA Damage. Aging 2020, 12, 3662–3681. [Google Scholar] [CrossRef]
- Dehghani-Soltani, S.; Eftekhar-Vaghefi, S.H.; Babaee, A.; Basiri, M.; Mohammadipoor-Ghasemabad, L.; Vosough, P.; Ahmadi-Zeidabadi, M. Pulsed and Discontinuous Electromagnetic Field Exposure Decreases Temozolomide Resistance in Glioblastoma by Modulating the Expression of O6-Methylguanine-DNA Methyltransferase, Cyclin-D1, and P53. Cancer Biother. Radiopharm. 2021, 36, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Sołek, P.; Mytych, J.; Łannik, E.; Majchrowicz, L.; Koszła, O.; Koziorowska, A.; Koziorowski, M. Cancer On-Target: Selective Enhancement of 3-Bromopyruvate Action by an Electromagnetic Field in Vitro. Free Radic. Biol. Med. 2022, 180, 153–164. [Google Scholar] [CrossRef]
- Cios, A.; Ciepielak, M.; Lieto, K.; Matak, D.; Lewicki, S.; Palusińska, M.; Stankiewicz, W.; Szymański, Ł. Extremely Low-Frequency Electromagnetic Field (ELF-EMF) Induced Alterations in Gene Expression and Cytokine Secretion in Clear Cell Renal Carcinoma Cells. Med. Pr. 2024, 75, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Cios, A.; Ciepielak, M.; Stankiewicz, W.; Szymański, Ł. The Influence of the Extremely Low Frequency Electromagnetic Field on Clear Cell Renal Carcinoma. Int. J. Mol. Sci. 2021, 22, 1342. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Im, S.H.; Yoon, Y.J.; Kim, H.M.; Lee, H.J.; Park, G.S. A 60 Hz Uniform Electromagnetic Field Promotes Human Cell Proliferation by Decreasing Intracellular Reactive Oxygen Species Levels. PLoS ONE 2018, 13, e0199753. [Google Scholar] [CrossRef]
- Focke, F.; Schuermann, D.; Kuster, N.; Schär, P. DNA Fragmentation in Human Fibroblasts under Extremely Low Frequency Electromagnetic Field Exposure. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2010, 683, 74–83. [Google Scholar] [CrossRef]
- Rezaie-Tavirani, M.; Hasanzadeh, H.; Seyyedi, S.; Zali, H. Proteomic Analysis of the Effect of Extremely Low-Frequency Electromagnetic Fields (ELF-EMF) with Different Intensities in SH-SY5Y Neuroblastoma Cell Line. J. Lasers Med. Sci. 2017, 8, 79–83. [Google Scholar] [CrossRef]
- Mehdizadeh, R.; Madjid Ansari, A.; Forouzesh, F.; Shahriari, F.; Shariatpanahi, S.P.; Salaritabar, A.; Javidi, M.A. P53 Status, and G2/M Cell Cycle Arrest, Are Determining Factors in Cell-Death Induction Mediated by ELF-EMF in Glioblastoma. Sci. Rep. 2023, 13, 10845. [Google Scholar] [CrossRef]
- Merla, C.; Liberti, M.; Consales, C.; Denzi, A.; Apollonio, F.; Marino, C.; Benassi, B. Evidences of Plasma Membrane-Mediated ROS Generation upon ELF Exposure in Neuroblastoma Cells Supported by a Computational Multiscale Approach. Biochim. Biophys. Acta Biomembr. 2019, 1861, 1446–1457. [Google Scholar] [CrossRef] [PubMed]
- Amiri, M.; Basiri, M.; Eskandary, H.; Akbarnejad, Z.; Esmaeeli, M.; Masoumi-Ardakani, Y.; Ahmadi-Zeidabadi, M. Cytotoxicity of Carboplatin on Human Glioblastoma Cells Is Reduced by the Concomitant Exposure to an Extremely Low-Frequency Electromagnetic Field (50 Hz, 70 G). Electromagn. Biol. Med. 2018, 37, 138–145. [Google Scholar] [CrossRef]
- Azanza, M.; Del Moral, A. Cell Membrane Biochemistry and Neurobiological Approach to Biomagnetism. Prog. Neurobiol. 1994, 44, 517–601. [Google Scholar] [CrossRef] [PubMed]
- Liboff, A.R. Cyclotron Resonance in Membrane Transport. In Interactions between Electromagnetic Fields and Cells; Chiabrera, A., Nicolini, C., Schwan, H.P., Eds.; Plenum Press: London, UK, 1985; pp. 281–296. ISBN 978-0306420832. [Google Scholar]
- Liboff, A.R.; Rozek, R.J.; Sherman, M.L.; McLeod, B.R.; Smith, S.D. Ca 2+-45 Cyclotron Resonance in Human Lymphocytes. Electromagn. Biol. Med. 1987, 6, 13–22. [Google Scholar] [CrossRef]
- Liboff, A.R.; McLeod, B.R.; Smith, S.D. Ion Cyclotron Resonance Effects of ELF Fields in Biological Systems. In Extremely Low Frequency Electromagnetic Fields: The Question of Cancer; Wilson, B.W., Stevens, R.G., Anderson, L.E., Eds.; Batelle Press: Columbus, OH, USA, 1990; pp. 251–289. ISBN 978-0935470482. [Google Scholar]
- Lednev, V.V. Possible Mechanism for the Influence of Weak Magnetic Fields on Biological Systems. Bioelectromagnetics 1991, 12, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Markov, M.S. “Biological Windows”: A Tribute to W. Ross Adey. Environmentalist 2005, 25, 67–74. [Google Scholar] [CrossRef]
- Bawin, S.M.; Adey, W.R. Sensitivity of Calcium Binding in Cerebral Tissue to Weak Environmental Electric Fields Oscillating at Low Frequency. Proc. Natl. Acad. Sci. USA 1976, 73, 1999–2003. [Google Scholar] [CrossRef]
- Blackman, C.F.; Benane, S.G.; Kinney, L.S.; Joines, W.T.; House, D.E. Effects of ELF Fields on Calcium-Ion Efflux from Brain Tissue in Vitro. Radiat. Res. 1982, 92, 510–520. [Google Scholar] [CrossRef]
- Blackman, C.F.; Benane, S.G.; Rabinowitz, J.R.; House, D.E.; Joines, W.T. A Role for the Magnetic Field in the Radiation-induced Efflux of Calcium Ions from Brain Tissue in Vitro. Bioelectromagnetics 1985, 6, 327–337. [Google Scholar] [CrossRef]
- Blackman, C.F.; Benane, S.G.; House, D.E.; Joines, W.T. Effects of ELF (1–120 Hz) and Modulated (50 Hz) RF Fields on the Efflux of Calcium Ions from Brain Tissue in Vitro. Bioelectromagnetics 1985, 6, 1–11. [Google Scholar] [CrossRef]
- López de Mingo, I.; Rivera González, M.-X.; Maestú Unturbe, C. The Cellular Response Is Determined by a Combination of Different ELF-EMF Exposure Parameters: A Scope Review. Int. J. Mol. Sci. 2024, 25, 5074. [Google Scholar] [CrossRef] [PubMed]
- López de Mingo, I.; Rivera González, M.-X.; Ramos Gómez, M.; Maestú Unturbe, C. The Frequency of a Magnetic Field Determines the Behavior of Tumor and Non-Tumor Nerve Cell Models. Int. J. Mol. Sci. 2025, 26, 2032. [Google Scholar] [CrossRef] [PubMed]
- Rivera González, M.-X.; López de Mingo, I.; Amuneke Ramírez, A.; Maestú Unturbe, C. Design and Characterisation of a Cell Exposure System with High Magnetic Field Homogeneity: RILZ Coils. Front. Bioeng. Biotechnol. 2024, 12, 1337899. [Google Scholar] [CrossRef] [PubMed]
- Council of the European Union 1999/519/EC: Council Recommendation of 12 July 1999 on the Limitation of Exposure of the General Public to Electromagnetic Fields (0 Hz to 300 GHz). Off. J. Eur. Communities 1999, 1–12.
- Bawin, S.M.; Sheppard, A.; Adey, W.R. 203—Possible Mechanisms of Weak Electromagnetic Field Coupling in Brain Tissue. Bioelectrochemistry Bioenerg. 1978, 5, 67–76. [Google Scholar] [CrossRef]
- Adey, W.R. Tissue Interactions with Nonionizing Electromagnetic Fields. Physiol. Rev. 1981, 61, 435–514. [Google Scholar] [CrossRef]
- Adair, R.K. Criticism of Lednev’s Mechanism for the Influence of Weak Magnetic Fields on Biological Systems. Bioelectromagnetics 1992, 13, 231–235. [Google Scholar] [CrossRef]
- Liboff, A.R.; Parkinson, W.C. Search for Ion-cyclotron Resonance in an Na + -transport System. Bioelectromagnetics 1991, 12, 77–83. [Google Scholar] [CrossRef]
- Parkinson, W.C.; Hanks, C.T. Search for Cyclotron Resonance in Cells in Vitro. Bioelectromagnetics 1989, 10, 129–145. [Google Scholar] [CrossRef]
- Liboff, A.R.; McLeod, B.R. Kinetics of Channelized Membrane Ions in Magnetic Fields. Bioelectromagnetics 1988, 9, 39–51. [Google Scholar] [CrossRef]
- Smith, S.D.; McLeod, B.R.; Liboff, A.R.; Cooksey, K. Calcium Cyclotron Resonance and Diatom Mobility. Bioelectromagnetics 1987, 8, 215–227. [Google Scholar] [CrossRef]
- Blackman, C.F.; Blanchard, J.P.; Benane, S.G.; House, D.E. Empirical Test of an Ion Parametric Resonance Model for Magnetic Field Interactions with PC-12 Cells. Bioelectromagnetics 1994, 15, 239–260. [Google Scholar] [CrossRef]
- Blanchard, J.P.; Blackman, C.F. Clarification and Application of an Ion Parametric Resonance Model for Magnetic Field Interactions with Biological Systems. Bioelectromagnetics 1994, 15, 217–238. [Google Scholar] [CrossRef]
- Azanza, M.J.; del Moral, A.; Pérez Bruzón, R.N. Frequency Resonance Effect of Neurons under Low-Frequency Weak Magnetic Field. J. Magn. Magn. Mater. 2007, 310, 2865–2867. [Google Scholar] [CrossRef]
- Blackman, C.F.; Blanchard, J.P.; Benane, S.G.; House, D.E. Experimental Determination of Hydrogen Bandwidth for the Ion Parametric Resonance Model. Bioelectromagnetics 1999, 20, 5–12. [Google Scholar] [CrossRef]
- Adey, W.R. Horizons in Science: Physical Regulation of Living Matter as an Emergent Concept in Health and Disease. In Electricity and Magnetism in Biology and Medicine; Springer US: Boston, MA, USA, 1999; pp. 53–57. ISBN 978-0-306-46041-8. [Google Scholar]
- Markov, M. Biological Mechanisms of the Magnetic Field Action. IEEE Trans. Magn. 1981, 17, 2334–2337. [Google Scholar] [CrossRef]
- Markov, M. Thermal vs. Nonthermal Mechanisms of Interactions between Electromagnetic Fields and Biological Systems. In Bioelectromagnetics Current Concepts. NATO Security Through Science Series; Markov, M.S., Ed.; Springer: Dordrecht, The Netherlands, 2006; pp. 1–15. ISBN 978-1-4020-4278-2. [Google Scholar]
- Markov, M.S. Biological Effects of Extremely Low Frequency Magnetic Fields. In Biomagnetic Stimulation; Springer US: Boston, MA, USA, 1994; pp. 91–103. ISBN 978-1-4757-9509-7. [Google Scholar]
- Markov, M.S. Benefit and Hazard of Electromagnetic Fields. In Electromagnetic Fields In Medicine And Biology; Markov, M., Ed.; CRC Press Taylor & Francis Group: New York, NY, USA, 2015; pp. 15–28. ISBN 978-1-4822-4851-7. [Google Scholar]
- de Groot, M.W.G.D.M.; van Kleef, R.G.D.M.; de Groot, A.; Westerink, R.H.S. In Vitro Developmental Neurotoxicity Following Chronic Exposure to 50 Hz Extremely Low-Frequency Electromagnetic Fields in Primary Rat Cortical Cultures. Toxicol. Sci. 2016, 149, 433–440. [Google Scholar] [CrossRef]
- Calcabrini, C.; Mancini, U.; De Bellis, R.; Diaz, A.R.; Martinelli, M.; Cucchiarini, L.; Sestili, P.; Stocchi, V.; Potenza, L. Effect of Extremely Low-Frequency Electromagnetic Fields on Antioxidant Activity in the Human Keratinocyte Cell Line NCTC 2544. Biotechnol. Appl. Biochem. 2017, 64, 415–422. [Google Scholar] [CrossRef]
- García-minguillán, O.; Prous, R.; Ramirez-castillejo, M.D.C.; Maestú, C. CT2A Cell Viability Modulated by Electromagnetic Fields at Extremely Low Frequency under No Thermal Effects. Int. J. Mol. Sci. 2020, 21, 152. [Google Scholar] [CrossRef]
- Morabito, C.; Steimberg, N.; Rovetta, F.; Boniotti, J.; Guarnieri, S.; Mazzoleni, G.; Mariggiò, M.A. Extremely Low-Frequency Electromagnetic Fields Affect Myogenic Processes in C2C12 Myoblasts: Role of Gap-Junction-Mediated Intercellular Communication. Biomed. Res. Int. 2017, 2017, 2460215. [Google Scholar] [CrossRef]
- Ma, Q.; Deng, P.; Zhu, G.; Liu, C.; Zhang, L.; Zhou, Z.; Luo, X.; Li, M.; Zhong, M.; Yu, Z.; et al. Extremely Low-Frequency Electromagnetic Fields Affect Transcript Levels of Neuronal Differentiation-Related Genes in Embryonic Neural Stem Cells. PLoS ONE 2014, 9, e90041. [Google Scholar] [CrossRef]
- Costantini, E.; Marconi, G.D.; Fonticoli, L.; Aielli, L.; Trubiani, O.; Rajan, T.S.; Pizzicannella, J.; Reale, M.; Diomede, F. Improved Osteogenic Differentiation by Extremely Low Electromagnetic Field Exposure: Possible Application for Bone Engineering. Histochem. Cell Biol. 2022, 158, 369–381. [Google Scholar] [CrossRef]
- Duan, W.; Liu, C.; Zhang, L.; He, M.; Xu, S.; Chen, C.; Pi, H.; Gao, P.; Zhang, Y.; Zhong, M.; et al. Comparison of the Genotoxic Effects Induced by 50 Hz Extremely Low-Frequency Electromagnetic Fields and 1800 MHz Radiofrequency Electromagnetic Fields in GC-2 Cells. Radiat. Res. 2015, 183, 305–314. [Google Scholar] [CrossRef]
- Park, J.E.; Seo, Y.K.; Yoon, H.H.; Kim, C.W.; Park, J.K.; Jeon, S. Electromagnetic Fields Induce Neural Differentiation of Human Bone Marrow Derived Mesenchymal Stem Cells via ROS Mediated EGFR Activation. Neurochem. Int. 2013, 62, 418–424. [Google Scholar] [CrossRef]
- Vianale, G.; Reale, M.; Amerio, P.; Stefanachi, M.; Di Luzio, S.; Muraro, R. Extremely Low Frequency Electromagnetic Field Enhances Human Keratinocyte Cell Growth and Decreases Proinflammatory Chemokine Production. Br. J. Dermatol. 2008, 158, 1189–1196. [Google Scholar] [CrossRef]
- Reale, M.; Kamal, M.A.; Patruno, A.; Costantini, E.; D’Angelo, C.; Pesce, M.; Greig, N.H. Neuronal Cellular Responses to Extremely Low Frequency Electromagnetic Field Exposure: Implications Regarding Oxidative Stress and Neurodegeneration. PLoS ONE 2014, 9, e104973. [Google Scholar] [CrossRef]
- Falone, S.; Grossi, M.R.; Cinque, B.; D’Angelo, B.; Tettamanti, E.; Cimini, A.; Di Ilio, C.; Amicarelli, F. Fifty Hertz Extremely Low-Frequency Electromagnetic Field Causes Changes in Redox and Differentiative Status in Neuroblastoma Cells. Int. J. Biochem. Cell Biol. 2007, 39, 2093–2106. [Google Scholar] [CrossRef]
- Yin, C.; Luo, X.; Duan, Y.; Duan, W.; Zhang, H.; He, Y.; Sun, G.; Sun, X. Neuroprotective Effects of Lotus Seedpod Procyanidins on Extremely Low Frequency Electromagnetic Field-Induced Neurotoxicity in Primary Cultured Hippocampal Neurons. Biomed. Pharmacother. 2016, 82, 628–639. [Google Scholar] [CrossRef]
- Colciago, A.; Audano, M.; Bonalume, V.; Melfi, V.; Mohamed, T.; Reid, A.J.; Faroni, A.; Greer, P.A.; Mitro, N.; Magnaghi, V. Transcriptomic Profile Reveals Deregulation of Hearing-Loss Related Genes in Vestibular Schwannoma Cells Following Electromagnetic Field Exposure. Cells 2021, 10, 1840. [Google Scholar] [CrossRef]
- Zuo, H.; Liu, X.; Li, Y.; Wang, D.; Hao, Y.; Yu, C.; Xu, X.; Peng, R.; Song, T. The Mitochondria/Caspase-Dependent Apoptotic Pathway Plays a Role in the Positive Effects of a Power Frequency Electromagnetic Field on Alzheimer’s Disease Neuronal Model. J. Chem. Neuroanat. 2020, 109, 101857. [Google Scholar] [CrossRef]
- Salek, F.; Baharara, J.; Shahrokhabadi, K.N.; Amini, E. The Guardians of Germ Cells; Sertoli-Derived Exosomes against Electromagnetic Field-Induced Oxidative Stress in Mouse Spermatogonial Stem Cells. Theriogenology 2021, 173, 112–122. [Google Scholar] [CrossRef]
- Samiei, M.; Aghazadeh, Z.; Abdolahinia, E.D.; Vahdati, A.; Daneshvar, S.; Noghani, A. The Effect of Electromagnetic Fields on Survival and Proliferation Rate of Dental Pulp Stem Cells. Acta Odontol. Scand. 2020, 78, 494–500. [Google Scholar] [CrossRef]
- Consales, C.; Panatta, M.; Butera, A.; Filomeni, G.; Merla, C.; Carrì, M.T.; Marino, C.; Benassi, B. 50-Hz Magnetic Field Impairs the Expression of Iron-Related Genes in the in Vitro SOD1 G93A Model of Amyotrophic Lateral Sclerosis. Int. J. Radiat. Biol. 2018, 95, 368–377. [Google Scholar] [CrossRef]
- Aparicio-Bautista, D.I.; Chávez-Valenzuela, D.; Ambriz-Álvarez, G.; Córdova-Fraga, T.; Reyes-Grajeda, J.P.; Medina-Contreras, Ó.; Rodríguez-Cruz, F.; García-Sierra, F.; Zúñiga-Sánchez, P.; Gutiérrez-Gutiérrez, A.M.; et al. An Extremely Low-Frequency Vortex Magnetic Field Modifies Protein Expression, Rearranges the Cytoskeleton, and Induces Apoptosis of a Human Neuroblastoma Cell Line. Bioelectromagnetics 2022, 43, 225–244. [Google Scholar] [CrossRef]
- Lim, H.M.; Nam, M.H.; Kim, Y.M.; Seo, Y.K. Increasing Odontoblast-like Differentiation from Dental Pulp Stem Cells through Increase of β-Catenin/p-Gsk-3β Expression by Low-Frequency Electromagnetic Field. Biomedicines 2021, 9, 1049. [Google Scholar] [CrossRef]
- Koziorowska, A.; Romerowicz-Misielak, M.; Gierczak, N.; Gniady, S.; Koziorowski, M. Electromagnetic Field of Extremely Low Frequency (60Hz and 120Hz) Effects the Cell Cycle Progression and the Metabolic Activity of the Anterior Pituitary Gland Cells in Vitro. In Proceedings of the 17th IEEE International Conference on Smart Technologies, EUROCON 2017—Conference Proceedings, Ohrid, Mcedonia, 6–8 July 2017; pp. 537–541. [Google Scholar] [CrossRef]
- Cho, S.; Lee, Y.; Lee, S.; Choi, Y.J.; Chung, H.W. Enhanced Cytotoxic and Genotoxic Effects of Gadolinium Following ELF-EMF Irradiation in Human Lymphocytes. Drug. Chem. Toxicol. 2014, 37, 440–447. [Google Scholar] [CrossRef]
- Kim, Y.M.; Cho, S.E.; Kim, S.C.; Jang, H.J.; Seo, Y.K. Effects of Extremely Low Frequency Electromagnetic Fields on Melanogenesis through P-ERK and P-SAPK/JNK Pathways in Human Melanocytes. Int. J. Mol. Sci. 2017, 18, 2120. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chuang, C.Y.; Shu, W.Y.; Chang, C.W.; Chen, C.R.; Fan, T.C.; Hsu, I.C. Distinct Epidermal Keratinocytes Respond to Extremely Low-Frequency Electromagnetic Fields Differently. PLoS ONE 2014, 9, e113424. [Google Scholar] [CrossRef]
- Moya-Gómez, A.; Font, L.P.; Burlacu, A.; Alpizar, Y.A.; Cardonne, M.M.; Brône, B.; Bronckaers, A. Extremely Low-Frequency Electromagnetic Stimulation (ELF-EMS) Improves Neurological Outcome and Reduces Microglial Reactivity in a Rodent Model of Global Transient Stroke. Int. J. Mol. Sci. 2023, 24, 11117. [Google Scholar] [CrossRef]
- Barbieri, M. What Is Code Biology? Biosystems 2018, 164, 1–10. [Google Scholar] [CrossRef]
- Tomkins, G.M. The Metabolic Code. Science 1975, 189, 760–763. [Google Scholar] [CrossRef] [PubMed]
- Trifonov, E.N. The Multiple Codes of Nucleotide Sequences. Bull. Math. Biol. 1989, 51, 417–432. [Google Scholar] [CrossRef]
- Trifonov, E.N. Interfering Contexts of Regulatory Sequence Elements. Bioinformatics 1996, 12, 423–429. [Google Scholar] [CrossRef]
- Trifonov, E.N. Elucidating Sequence Codes: Three Codes for Evolution. Ann. N. Y. Acad. Sci. 1999, 870, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Strahl, B.D.; Allis, C.D. The Language of Covalent Histone Modifications. Nature 2000, 403, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.M. Histone Acetylation and an Epigenetic Code. BioEssays 2000, 22, 836–845. [Google Scholar] [CrossRef]
- Turner, B.M. Cellular Memory and the Histone Code. Cell 2002, 111, 285–291. [Google Scholar] [CrossRef]
- Turner, B.M. Defining an Epigenetic Code. Nat. Cell Biol. 2007, 9, 2–6. [Google Scholar] [CrossRef]
- Kühn, S.; Hofmeyr, J.-H.S. Is the “Histone Code” an Organic Code? Biosemiotics 2014, 7, 203–222. [Google Scholar] [CrossRef]
- Gabius, H.-J. Biological Information Transfer Beyond the Genetic Code: The Sugar Code. Naturwissenschaften 2000, 87, 108–121. [Google Scholar] [CrossRef]
- Janke, C. The Tubulin Code: Molecular Components, Readout Mechanisms, and Functions. J. Cell Biol. 2014, 206, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Verhey, K.J.; Gaertig, J. The Tubulin Code. Cell Cycle 2007, 6, 2152–2160. [Google Scholar] [CrossRef]
- Raunser, S.; Gatsogiannis, C. Deciphering the Tubulin Code. Cell 2015, 161, 960–961. [Google Scholar] [CrossRef]
- Barisic, M.; Maiato, H. The Tubulin Code: A Navigation System for Chromosomes during Mitosis. Trends Cell Biol. 2016, 26, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Markov, M.S. Biophysical Estimation of the Environmental Importance of Electromagnetic Fields. Rev. Environ. Health 1994, 10, 75–84. [Google Scholar] [CrossRef]
- Markov, M.S. Angiogenesis, Magnetic Fields and ‘Window Effects’. Cardiology 2010, 117, 54–56. [Google Scholar] [CrossRef] [PubMed]
- Adey, W.R. Potential Therapeutic Applications of Nonthermal Electromagnetic Fields: Ensemble Organization of Cells in Tissue as a Factor in Biological Field Sensing. In Bioelectromagnetic Medicine; Rosch, P.J., Markov, M.S., Eds.; Taylor & Francis: New York, NY, USA, 2004; Volume 1, pp. 1–15. ISBN 0-203-02165-7. [Google Scholar]
- Adey, W.R. Cell Membranes: The Electromagnetic Environment and Cancer Promotion. Neurochem. Res. 1988, 13, 671–677. [Google Scholar] [CrossRef]
- Adey, W.R. Biological Effects of Electromagnetic Fields. J. Cell. Biochem. 1993, 51, 410–416. [Google Scholar] [CrossRef]
- Simkó, M.; Mattsson, M. Extremely Low Frequency Electromagnetic Fields as Effectors of Cellular Responses in Vitro: Possible Immune Cell Activation. J. Cell. Biochem. 2004, 93, 83–92. [Google Scholar] [CrossRef]
- Funk, R.H.W.; Monsees, T.; Özkucur, N. Electromagnetic Effects—From Cell Biology to Medicine. Prog. Histochem. Cytochem. 2009, 43, 177–264. [Google Scholar] [CrossRef]
- Huang, Z.; Ito, M.; Zhang, S.; Toda, T.; Takeda, J.-I.; Ogi, T.; Ohno, K. Extremely Low-Frequency Electromagnetic Field Induces Acetylation of Heat Shock Proteins and Enhances Protein Folding. Ecotoxicol. Environ. Saf. 2023, 264, 115482. [Google Scholar] [CrossRef] [PubMed]
- Pipkin, J.L.; Hinson, W.G.; Young, J.F.; Rowland, K.L.; Shaddock, J.G.; Tolleson, W.H.; Duffy, P.H.; Casciano, D.A. Induction of Stress Proteins by Electromagnetic Fields in Cultured HL-60 Cells. Bioelectromagnetics 1999, 20, 347–357. [Google Scholar] [CrossRef]
- Bodega, G.; Forcada, I.; Suárez, I.; Fernández, B. Acute and Chronic Effects of Exposure to a 1-MT Magnetic Field on the Cytoskeleton, Stress Proteins, and Proliferation of Astroglial Cells in Culture. Environ. Res. 2005, 98, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.J.; Yu, Y.N.; Chiang, H.; Fu, Y.D.; Lu, D.Q. Effects of 50 Hz Magnetic Field Exposure on Protein Tyrosine Phosphorilation in Cultured Cells. Electro Magnetobiology 2001, 20, 207–214. [Google Scholar] [CrossRef]
- Bersani, F.; Marinelli, F.; Ognibene, A.; Matteucci, A.; Cecchi, S.; Santi, S.; Squarzoni, S.; Maraldi, N.M. Intramembrane Protein Distribution in Cell Cultures Is Affected by 50 Hz Pulsed Magnetic Fields. Bioelectromagnetics 1997, 18, 463–469. [Google Scholar] [CrossRef]
- Sun, W.J.; Chiang, H.; Fu, Y.T.; Yu, Y.N.; Xie, H.Y.; Lu, D.Q. Exposure to 50 Hz Electromagnetic Fields Induces the Phosphorylation and Activity of Stress-Activated Protein Kinase in Cultured Cells. Electro Magnetobiology 2001, 20, 415–423. [Google Scholar] [CrossRef]
- Löschinger, M. Stimulation of Protein Kinase A Activity and Induced Terminal Differentiation of Human Skin Fibroblasts in Culture by Low-Frequency Electromagnetic Fields. Toxicol. Lett. 1998, 96–97, 369–376. [Google Scholar] [CrossRef]
- Thumm, S.; Löschinger, M.; Glock, S.; Hämmerle, H.; Rodemann, H.P. Induction of CAMP-Dependent Protein Kinase A Activity in Human Skin Fibroblasts and Rat Osteoblasts by Extremely Low-Frequency Electromagnetic Fields. Radiat. Environ. Biophys. 1999, 38, 195–199. [Google Scholar] [CrossRef]
- Lange, S.; Richard, D.; Viergutz, T.; Kriehuber, R.; Weiss, D.G.; Simkó, M. Alterations in the Cell Cycle and in the Protein Level of Cyclin D1, P21CIP1, and P16INK4a after Exposure to 50 Hz MF in Human Cells. Radiat. Environ. Biophys. 2002, 41, 131–137. [Google Scholar] [CrossRef]
- Adey, W.R. The Sequence and Energetics of Cell Membrane Transductive Coupling to Intracellular Enzyme Systems. Bioelectrochemistry Bioenerg. 1986, 15, 447–456. [Google Scholar] [CrossRef]
- Kaczmarek, L.K.; Adey, W.R. The Efflux Of45Ca2+ and [3H]γ-Aminobutyric Acid from Cat Cerebral Cortex. Brain. Res. 1973, 63, 331–342. [Google Scholar] [CrossRef]
- Ebner, E. Resonance in Biological Systems: An Interdisciplinary View. In Contemporary Perspective on Science, Technology and Research Vol. 3; B P International: Bhanjipur, India, 2024; pp. 75–80. ISBN 978-81-968135-8-1. [Google Scholar]
- Blackman, C. Replication and Extension of Adey Group’s Calcium Efflux Results. In Electromagnetic Fields in Biology and Medicine; Markov, M., Ed.; CRC Press: Boca Raton, FL, USA, 2015; pp. 7–14. ISBN 978-1138749030. [Google Scholar]
- Santini, M.T.; Rainaldi, G.; Indovina, P.L. Cellular Effects of Extremely Low Frequency (ELF) Electromagnetic Fields. Int. J. Radiat. Biol. 2009, 85, 294–313. [Google Scholar] [CrossRef]
- Goldberg, R.B.; Creasey, W.A. A Review of Cancer Induction by Extremely Low Frequency Electromagnetic Fields. Is There a Plausible Mechanism? Med. Hypotheses 1991, 35, 265–274. [Google Scholar] [CrossRef]
- Evan, G.I.; Vousden, K.H. Proliferation, Cell Cycle and Apoptosis in Cancer. Nature 2001, 411, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Patergnani, S.; Danese, A.; Bouhamida, E.; Aguiari, G.; Previati, M.; Pinton, P.; Giorgi, C. Various Aspects of Calcium Signaling in the Regulation of Apoptosis, Autophagy, Cell Proliferation, and Cancer. Int. J. Mol. Sci. 2020, 21, 8323. [Google Scholar] [CrossRef]
- Valdespino-Gómez, V.M.; Valdespino-Castillo, P.M.; Valdespino-Castillo, V.E. Cell Signalling Pathways Interaction in Cellular Proliferation: Potential Target for Therapeutic Interventionism. Cirugía Y Cir. 2015, 83, 165–174. [Google Scholar] [CrossRef]
- Evan, G.I.; Brown, L.; Whyte, M.; Harrington, E. Apoptosis and the Cell Cycle. Curr. Opin. Cell Biol. 1995, 7, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Zarkovic, N. Roles and Functions of ROS and RNS in Cellular Physiology and Pathology. Cells 2020, 9, 767. [Google Scholar] [CrossRef] [PubMed]
- Morabito, C.; Guarnieri, S.; Fanò, G.; Mariggiò, M.A. Effects of Acute and Chronic Low Frequency Electromagnetic Field Exposure on PC12 Cells during Neuronal Differentiation. Cell. Physiol. Biochem. 2010, 26, 947–958. [Google Scholar] [CrossRef]
- Sahai, E.; Astsaturov, I.; Cukierman, E.; DeNardo, D.G.; Egeblad, M.; Evans, R.M.; Fearon, D.; Greten, F.R.; Hingorani, S.R.; Hunter, T.; et al. A Framework for Advancing Our Understanding of Cancer-Associated Fibroblasts. Nat. Rev. Cancer 2020, 20, 174–186. [Google Scholar] [CrossRef]

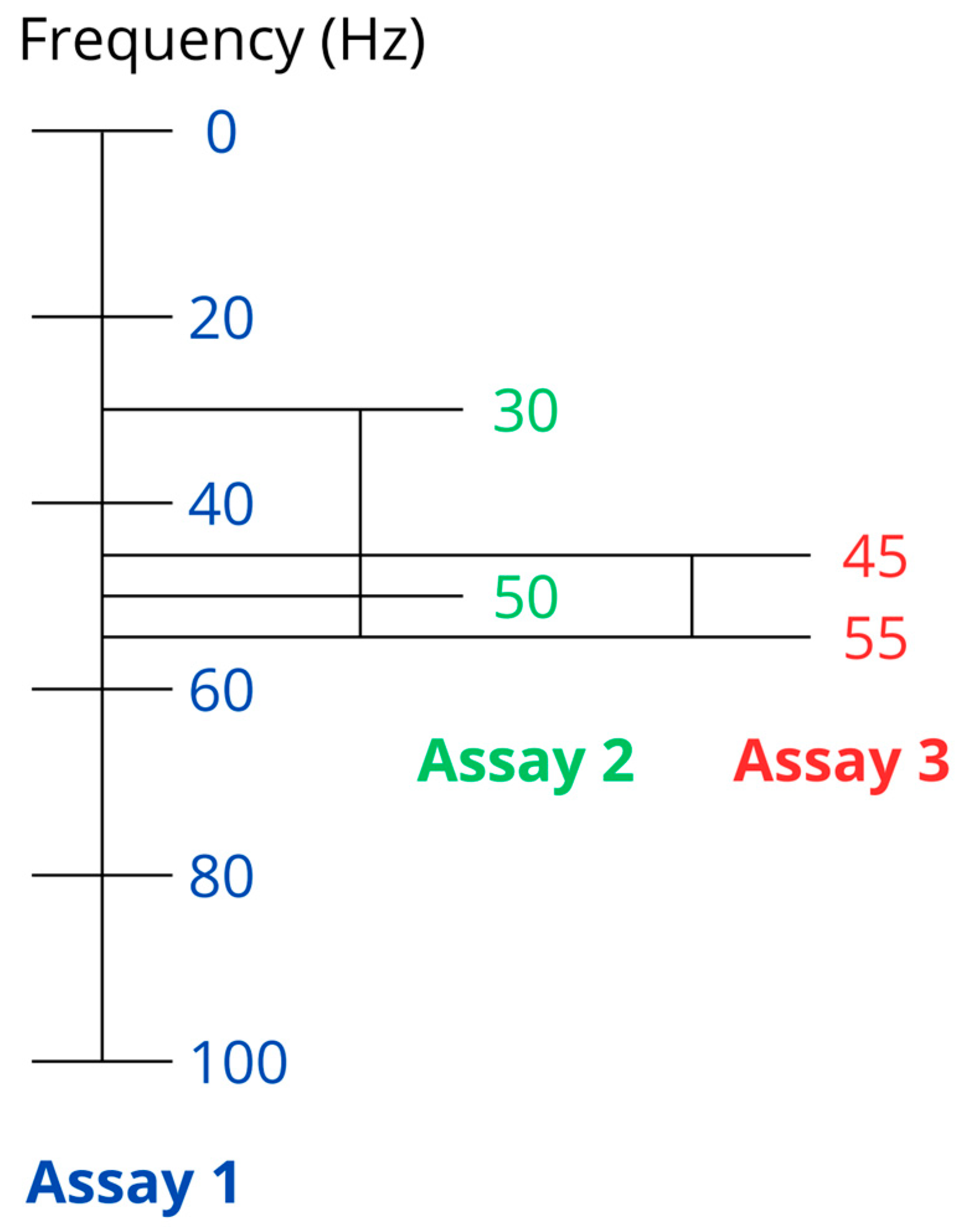
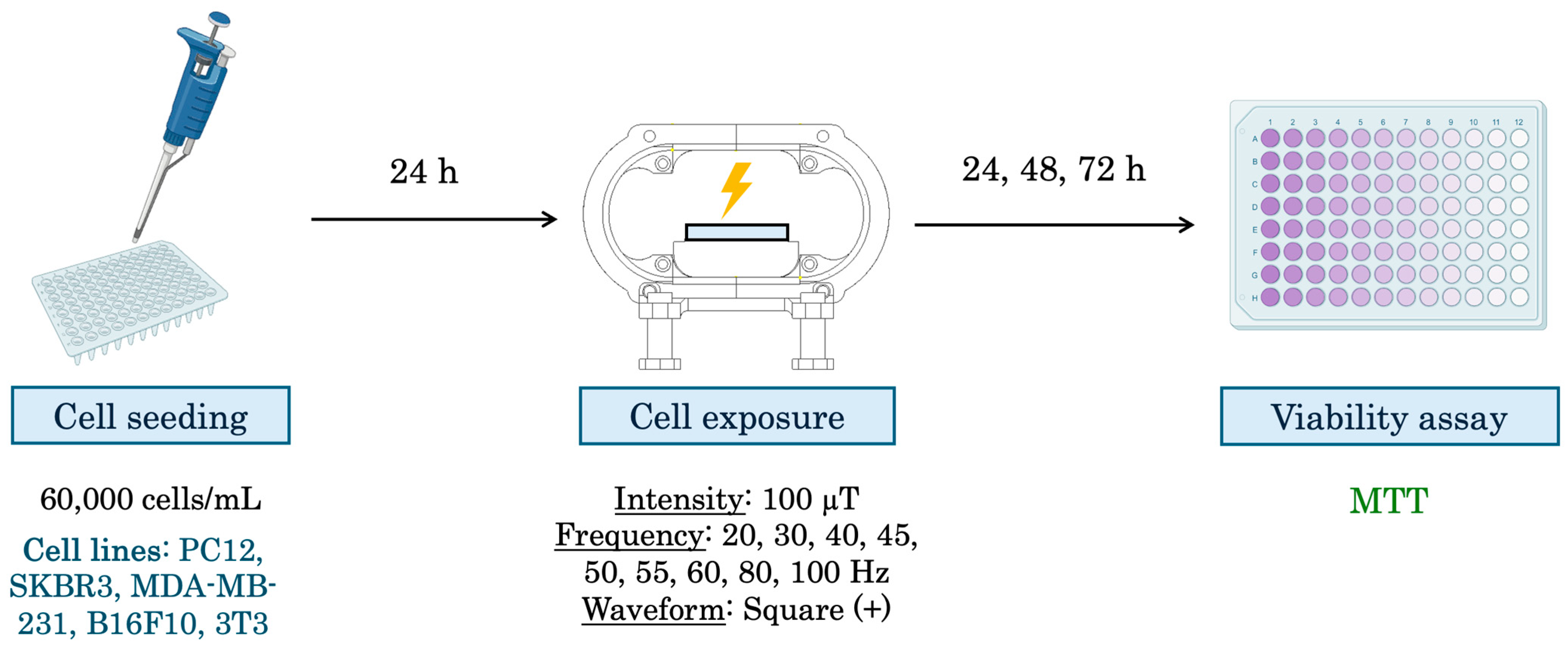

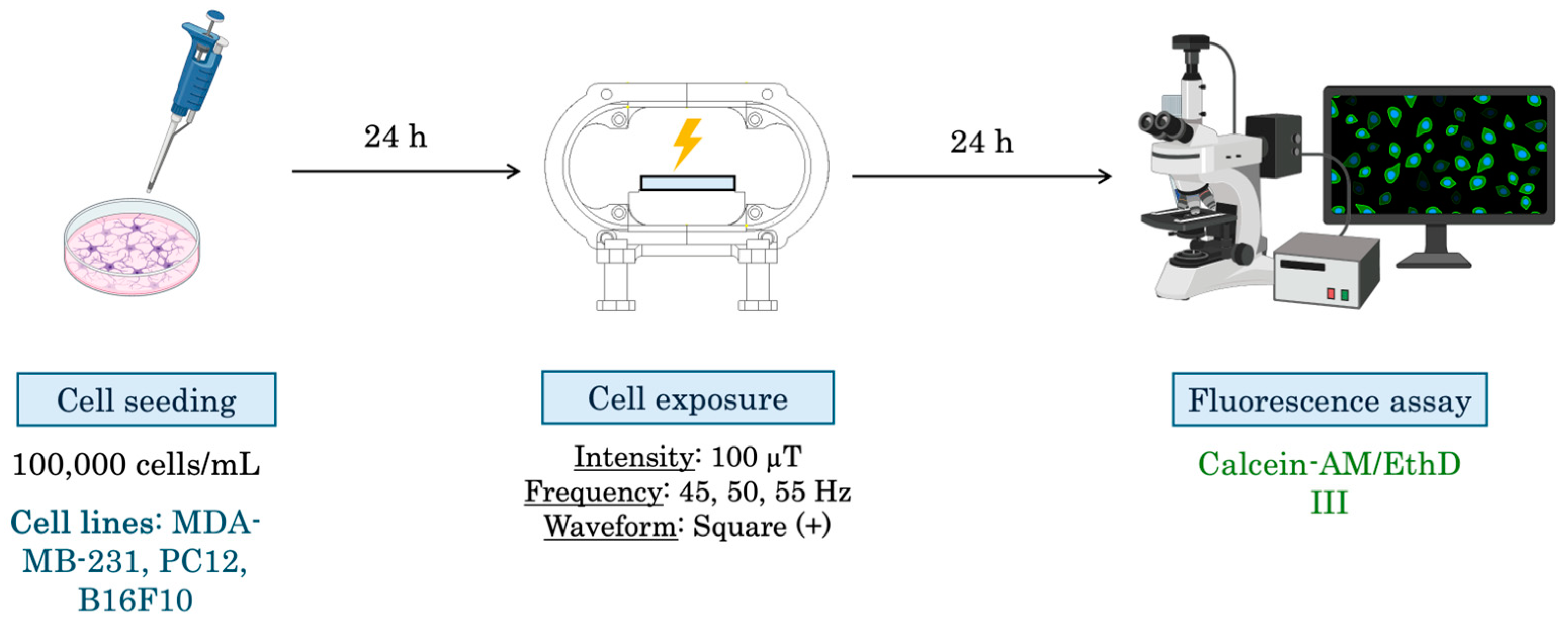

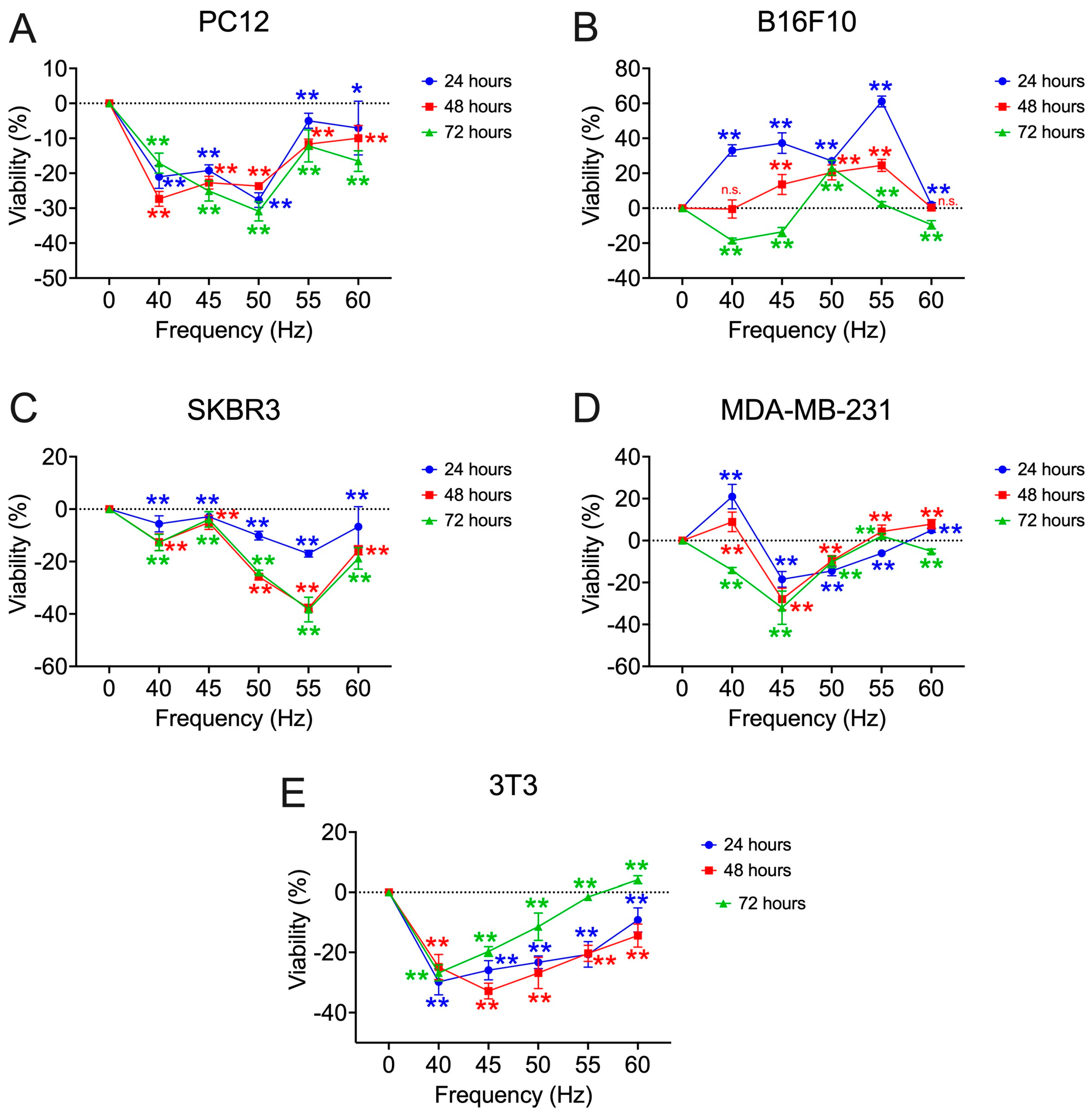
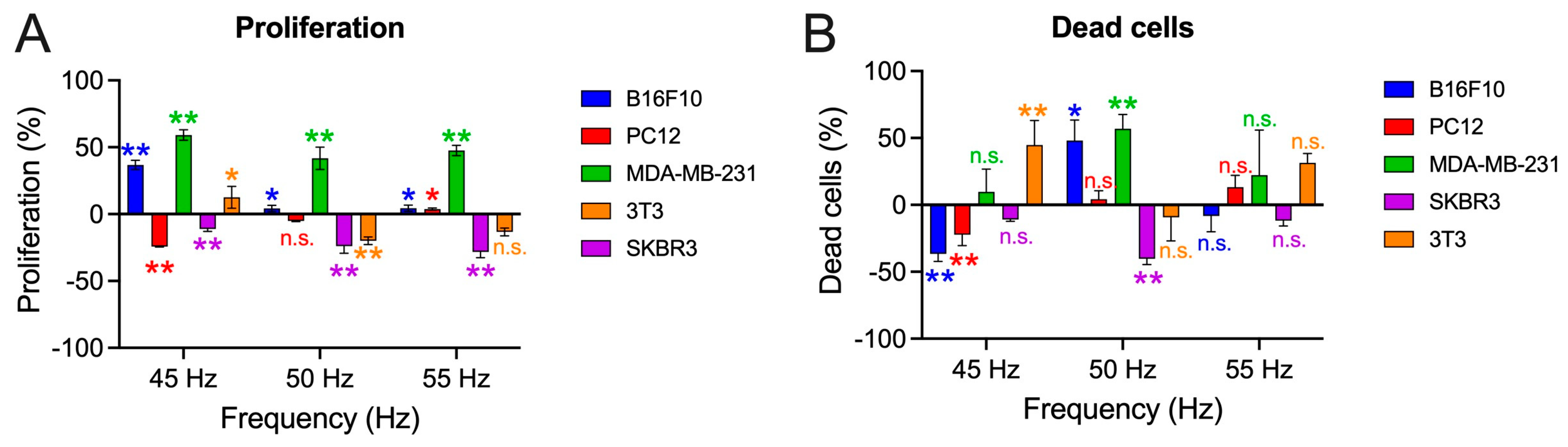
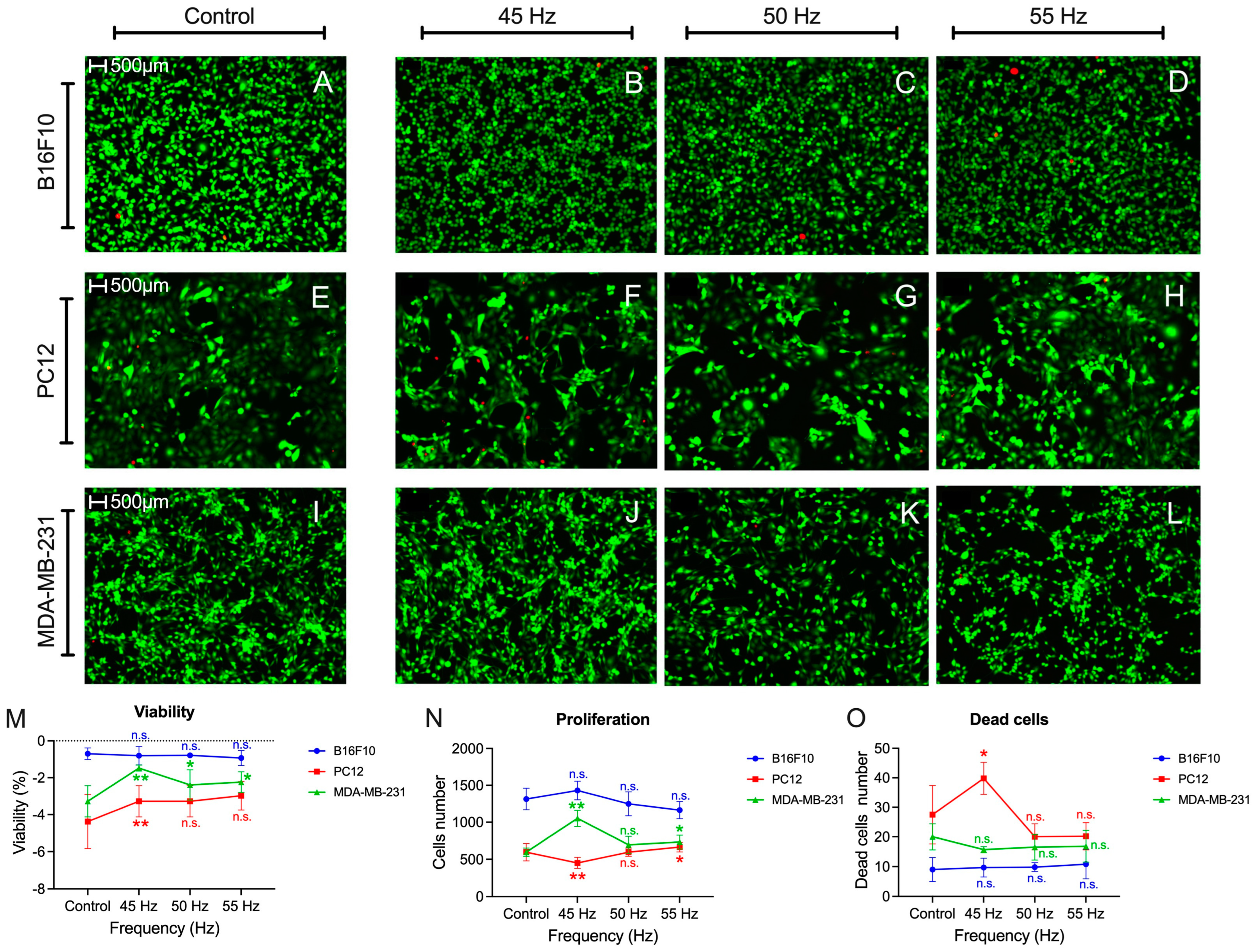

| Assay No. | Cell Line | Intensity [µT] | Frequency [Hz] | Exposure Time [Hours] | Biological Assay |
|---|---|---|---|---|---|
| 1 | PC12, SKBR3, MDA-MB-231, B16F10, 3T3 | 100 | 20, 40, 60, 80, 100 | 24, 48, 72 | Metabolic activity (MTT) |
| 2 | PC12, SKBR3, MDA-MB-231, B16F10, 3T3 | 100 | 30, 50 | 24, 48, 72 | Metabolic activity (MTT) |
| 3 | PC12, SKBR3, MDA-MB-231, B16F10, 3T3 | 100 | 45, 55 | 24, 48, 72 | Metabolic activity (MTT) |
| 4 | PC12, SKBR3, MDA-MB-231, B16F10, 3T3 | 100 | 45, 50, 55 | 24 | Proliferation and number of dead cells (Trypan Blue) |
| 5 | PC12, SKBR3, MDA-MB-231, B16F10, 3T3 | 100 | 50 | 24 | Viability/Apoptosis (Calcein/EthD) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López de Mingo, I.; Rivera González, M.X.; Ramos Gómez, M.; Maestú Unturbe, C. The Frequency of a Magnetic Field Reduces the Viability and Proliferation of Numerous Tumor Cell Lines. Biomolecules 2025, 15, 503. https://doi.org/10.3390/biom15040503
López de Mingo I, Rivera González MX, Ramos Gómez M, Maestú Unturbe C. The Frequency of a Magnetic Field Reduces the Viability and Proliferation of Numerous Tumor Cell Lines. Biomolecules. 2025; 15(4):503. https://doi.org/10.3390/biom15040503
Chicago/Turabian StyleLópez de Mingo, Isabel, Marco Xavier Rivera González, Milagros Ramos Gómez, and Ceferino Maestú Unturbe. 2025. "The Frequency of a Magnetic Field Reduces the Viability and Proliferation of Numerous Tumor Cell Lines" Biomolecules 15, no. 4: 503. https://doi.org/10.3390/biom15040503
APA StyleLópez de Mingo, I., Rivera González, M. X., Ramos Gómez, M., & Maestú Unturbe, C. (2025). The Frequency of a Magnetic Field Reduces the Viability and Proliferation of Numerous Tumor Cell Lines. Biomolecules, 15(4), 503. https://doi.org/10.3390/biom15040503






