Exogenous Application of Methyl Jasmonate and Salicylic Acid Mitigates Drought-Induced Oxidative Damages in French Bean (Phaseolus vulgaris L.)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material, Experimental Conditions, and Treatments
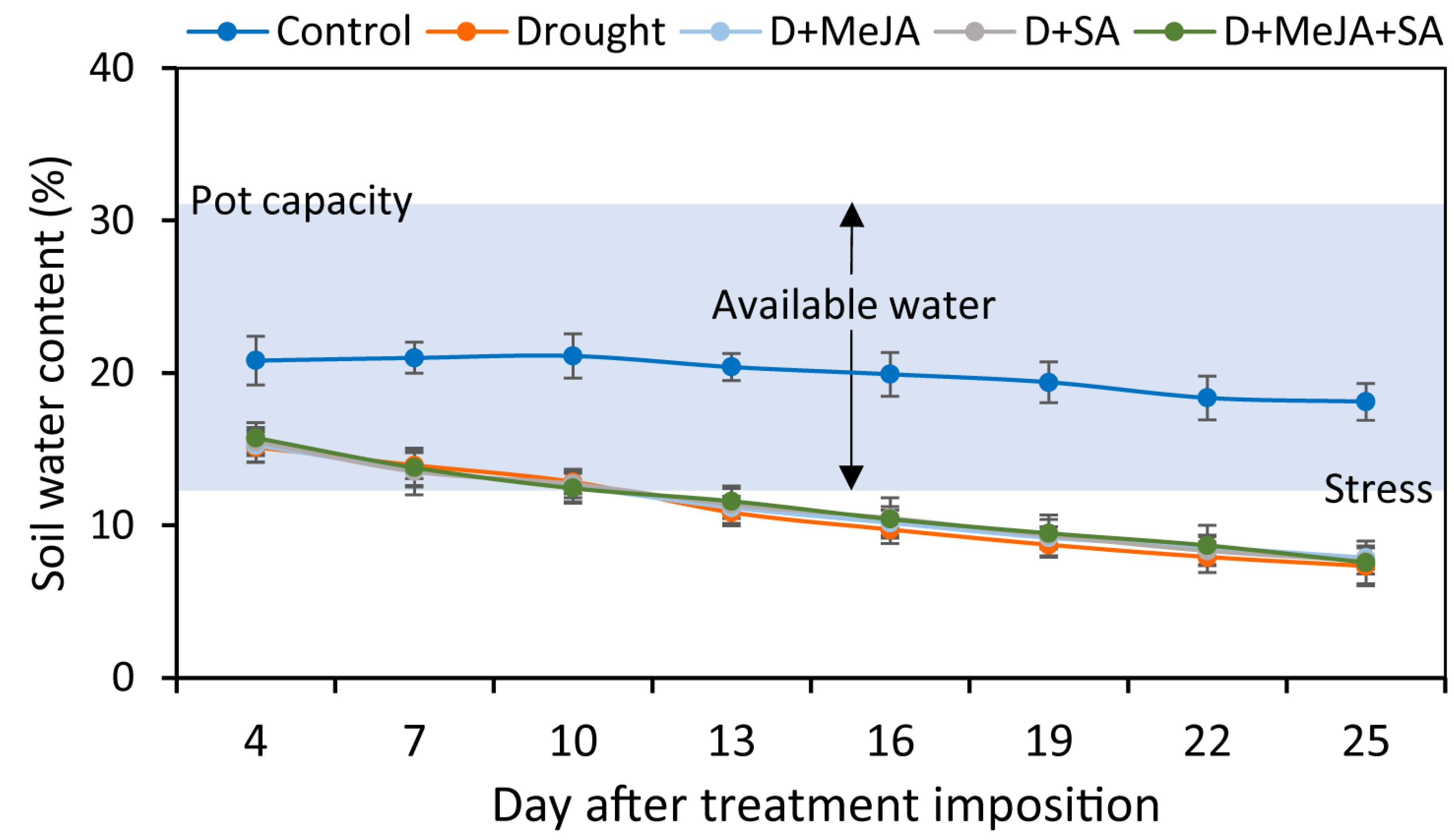
2.2. Pot Capacity and Water Content of the Experimental Soil
2.3. Growth Parameters
2.4. Physiological Parameters
2.4.1. Leaf Chlorophyll and Pigment Content
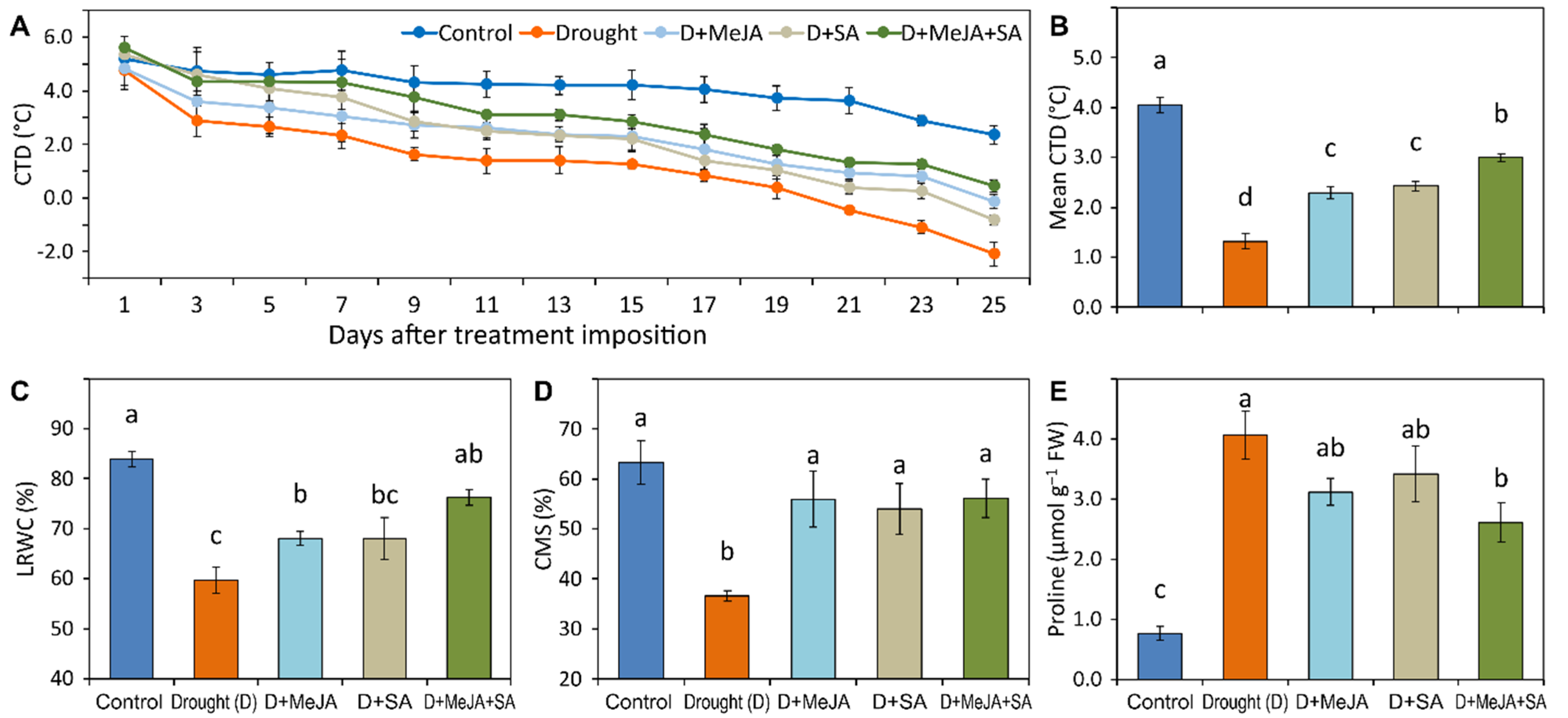
2.4.2. Canopy Temperature Depression
2.4.3. Leaf Relative Water Content
2.4.4. Cell Membrane Stability
2.5. Biochemical Observations
2.5.1. Proline Content
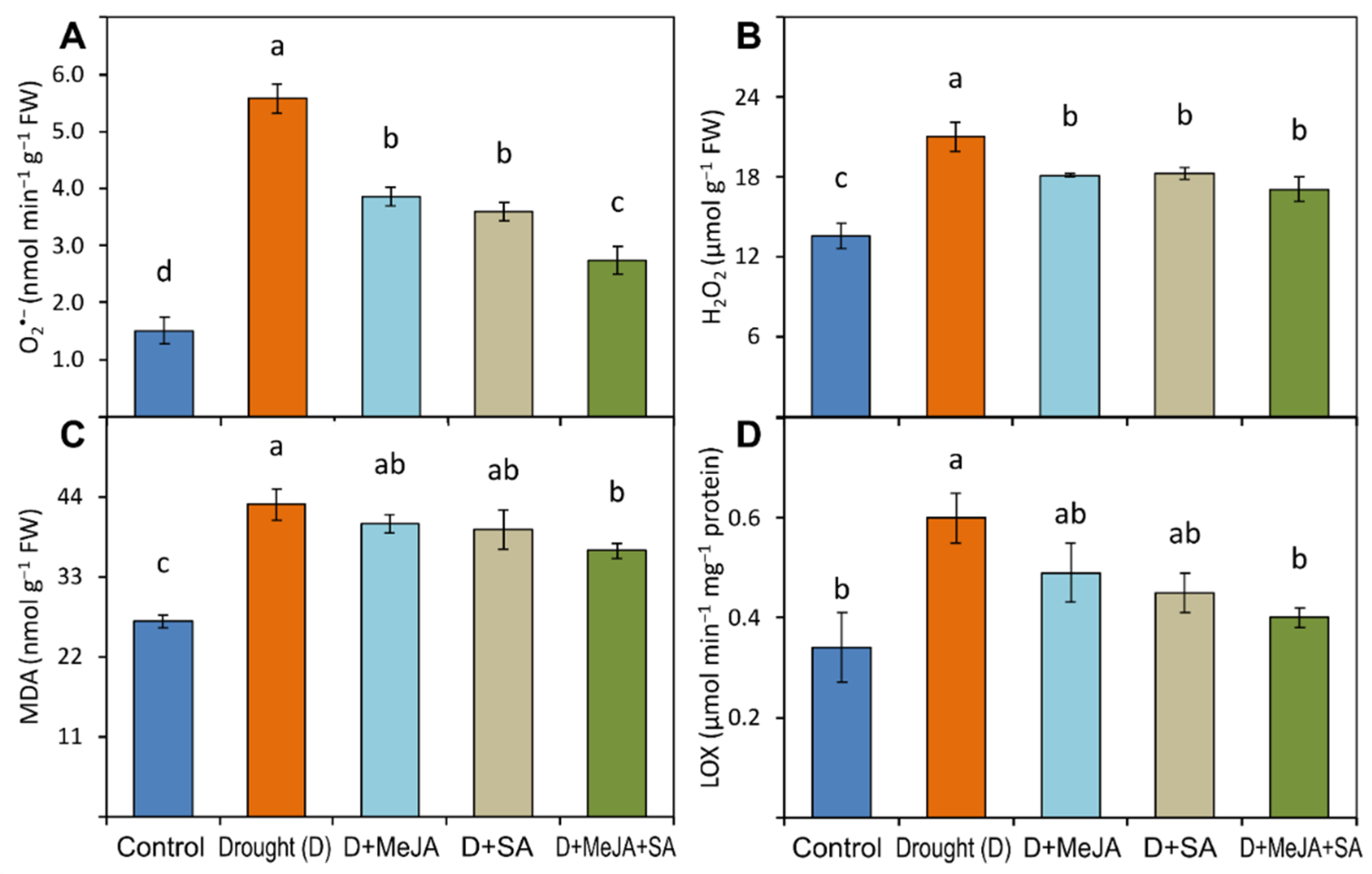
2.5.2. Oxidative Stress Indicators
2.5.3. Extraction and Quantitation of Soluble Protein
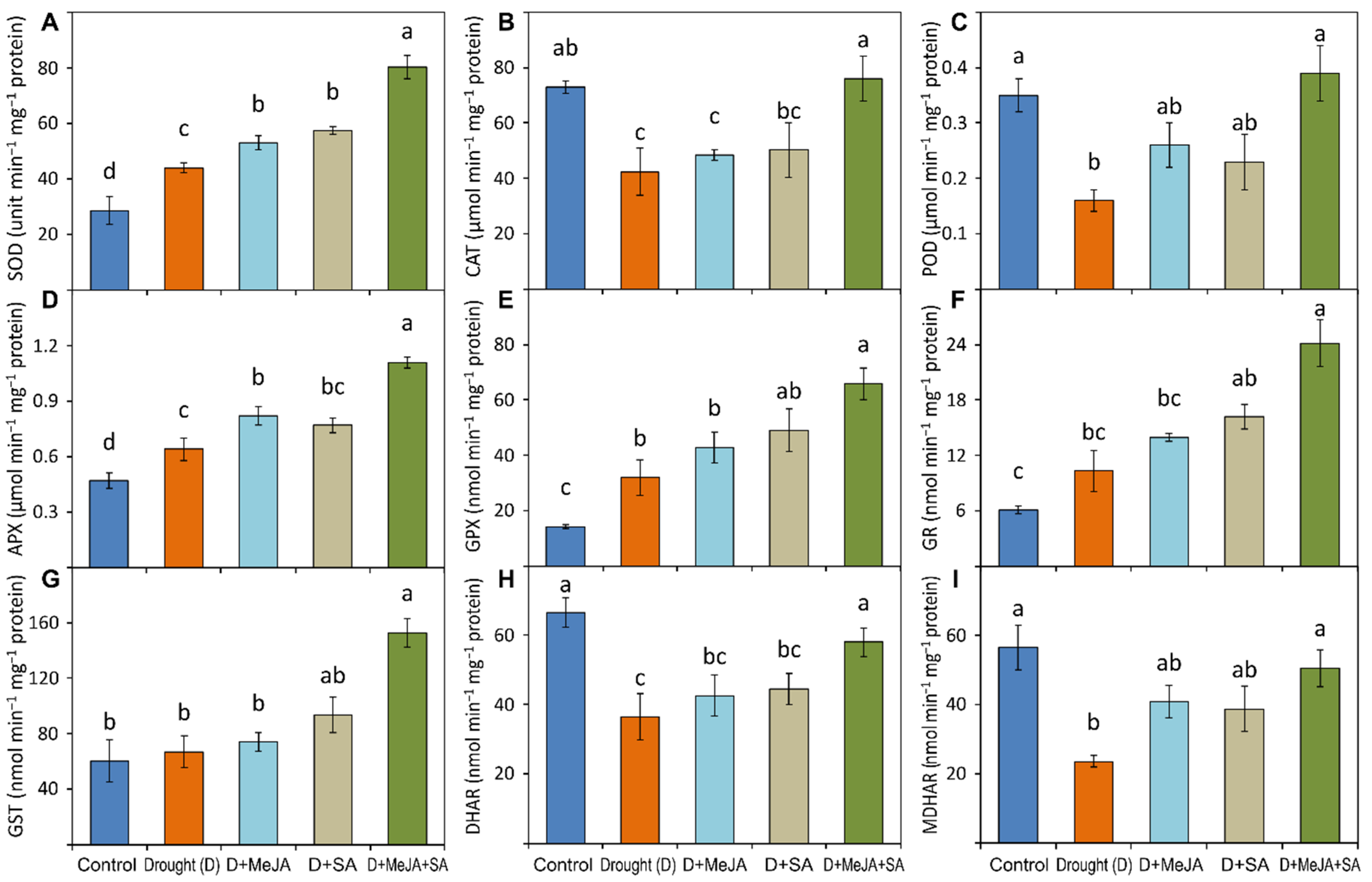
2.5.4. Assays of Enzymatic Activities
2.6. Statistical Analysis
3. Results
3.1. Soil Water Content
3.2. MeJA and SA Enhanced Plant Growth
3.3. Impact of MeJA and SA on Photosynthetic Pigments
3.4. MeJA and SA Improved Physiological Traits
3.5. MeJA and SA Suppressed the Generation of Oxidative Stress Indicators
3.6. Antioxidant Enzyme Activities and MeJA and SA Pre-Treatment
3.7. Comparative Assessment of Responses across Treatments
3.8. Principal Component Analysis (PCA)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Graham, P.H.; Ranalli, P. Common bean (Phaseolus vulgaris L.). Field Crop. Res. 1997, 53, 131–146. [Google Scholar] [CrossRef]
- Rosales-Serna, R.; Kohashi-Shibata, J.; Acosta-Gallegos, J.A.; Trejo-Lopez, C.; Ortizcereceres, J.; Kelly, J. Biomass distribution, maturity acceleration and yield in drought-stressed common bean cultivars. Field Crops Res. 2004, 85, 203–211. [Google Scholar] [CrossRef]
- Nazrul, M.I.; Shaheb, M.R. Performance of French bean (Phaseolus vulgaris L.) genotypes in Sylhet region of Bangladesh. Bangladesh Agron. J. 2016, 19, 37–44. [Google Scholar] [CrossRef] [Green Version]
- Assefa, T.; Wu, J.; Beebe, S.E.; Rao, I.M.; Marcomin, D.; Claude, R.J. Improving adaptation to drought stress in small red common bean: Phenotypic differences and predicted genotypic effects on grain yield, yield components and harvest index. Euphytica 2015, 203, 477–489. [Google Scholar] [CrossRef]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Ghobadi, M.; Taherabadi, S.; Ghobadi, M.-E.; Mohammadi, G.-R. Jalali-Honarmand, S. Antioxidant capacity, photosynthetic characteristics and water relations of sunflower (Helianthus annuus L.) cultivars in response to drought stress. Ind. Crop. Prod. 2013, 50, 29–38. [Google Scholar] [CrossRef]
- Lesznyák, M.; Hunyadi-Borbély, É.; Csajbók, J. The role of nutrient-water-supply and the cultivation in the yield of pea (Pisum sativum L.). Cereal Res. Commun. 2008, 36, 1079–1082. [Google Scholar]
- Trenberth, K.E.; Dai, A.; Van Der Schrier, G.; Jones, P.D.; Barichivich, J.; Briffa, K.R.; Sheffield, J. Global warming and changes in drought. Nat. Clim. Chang. 2014, 4, 17–22. [Google Scholar] [CrossRef]
- Schwalm, C.R.; Anderegg, W.R.L.; Michalak, A.M.; Fisher, J.B.; Biondi, F.; Koch, G.; Litvak, M.; Ogle, K.; Shaw, J.D.; Wolf, A.; et al. Global patterns of drought recovery. Nature 2017, 548, 202–205. [Google Scholar] [CrossRef]
- MoEF. Bangladesh Climate Change Strategy and Action Plan 2009; Ministry of Environment and Forests: Dhaka, Bangladesh, 2009.
- Jain, M.; Kataria, S.; Hirve, M.; Prajapati, R. Water deficit stress effects and responses in maize. In Plant Abiotic Stress Tolerance; Hasanuzzaman, M., Hakeem, K., Nahar, K., Alharby, H., Eds.; Springer: Cham, Switzerland, 2019; pp. 129–151. [Google Scholar]
- Gao, M.; Zhou, J.J.; Liu, H.; Zhang, W.; Hu, Y.; Liang, J.; Zhou, J. Foliar spraying with silicon and selenium reduces cadmium uptake and mitigates cadmium toxicity in rice. Sci. Total Environ. 2018, 631–632, 1100–1108. [Google Scholar] [CrossRef]
- Singh, M.; Kushwaha, B.K.; Singh, S.; Kumar, V.; Singh, V.P.; Prasad, S.M. Sulphur alters chromium (VI) toxicity in Solanum melongena seedlings: Role of sulphur assimilation and sulphur-containing antioxidants. Plant. Physiol. Biochem. 2017, 112, 183–192. [Google Scholar] [CrossRef]
- Fazeli, F.; Ghorbanli, M.; Niknam, V. Effect of drought on biomass, protein content, lipid peroxidation and antioxidant enzymes in two sesame cultivars. Plant. Biol. 2007, 51, 98–103. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Ascorbate and glutathione: The heart of the redox hub. Plant. Physiol. 2011, 155, 2–18. [Google Scholar] [CrossRef] [Green Version]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant. Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Noctor, G.; Lelarge-Trouverie, C.; Mhamdi, A. The metabolomics of oxidative stress. Phytochemistry 2014, 112, 33–53. [Google Scholar] [CrossRef]
- Alam, M.M.; Hasanuzzaman, M.; Nahar, K.; Fujita, M. Exogenous salicylic acid ameliorates short term drought stress in mustard (Brassica juncea L.) seedlings by up-regulating the antioxidant defense and glyoxalase system. Aust. J. Crop. Sci. 2013, 7, 1053–1063. [Google Scholar]
- Hasanuzzaman, M.; Nahar, K.; Bhuiyan, T.F.; Anee, T.I.; Inafuku, M.; Oku, H.; Fujita, M. Salicylic acid: An all-rounder in regulating abiotic stress responses in plants. In Phytohormones-Signaling Mechanisms and Crosstalk in Plant Development and Stress Responses; El-Esawi, M.A., Ed.; IntechOpen: London, UK, 2017; pp. 31–75. [Google Scholar]
- Mir, M.A.; John, R.; Alyemeni, M.N.; Alam, P.; Ahmad, P. Jasmonic acid ameliorates alkaline stress by improving growth performance, ascorbate glutathione cycle and glyoxylase system in maize seedlings. Sci. Rep. 2018, 8, 2831. [Google Scholar] [CrossRef]
- Lazebnik, J.; Frago, E.; Dicke, M.; van Loon, J.J.A. Phytohormone mediation of interactions between herbivores and plant pathogens. J. Chem. Ecol. 2014, 40, 730–741. [Google Scholar] [CrossRef]
- Soares, A.M.S.; Oliveira, J.T.A.; Gondim, D.M.F.; Domingues, D.P.; Machado, O.L.T.; Jacinto, T. Assessment of stress–related enzymes in response to either exogenous salicylic acid or methyl jasmonate in Jatropha curcas L. leaves, an attractive plant to produce biofuel. South. Afr. J. Bot. 2016, 105, 163–168. [Google Scholar] [CrossRef]
- Li, L.; Lu, X.; Ma, H.; Lyu, D. Jasmonic acid regulates the ascorbate-glutathione cycle in Malus baccata Borkh roots under low root-zone temperature. Acta Physiol. Plant. 2017, 39, 174. [Google Scholar] [CrossRef]
- Alam, M.M.; Nahar, K.; Hasanuzzaman, M.; Fujita, M. Exogenous jasmonic acid modulates the physiology, antioxidant defense and glyoxalase systems in imparting drought stress tolerance in different Brassica species. Plant. Biotechnol. Rep. 2014, 8, 279–293. [Google Scholar] [CrossRef]
- Shyu, C.; Brutnell, T.P. Growth-defense balance in grass biomass production: The role of jasmonates. J. Exp. Bot. 2015, 66, 4165–4176. [Google Scholar] [CrossRef] [Green Version]
- Karamian, R.; Ghasemlou, F.; Amiri, H. Physiological evaluation of drought stress tolerance and recovery in Verbascum sinuatum plants treated with methyl jasmonate, salicylic acid and titanium dioxide nanoparticles. Plant. Biosyst. 2019, 154, 277–287. [Google Scholar] [CrossRef]
- Tayyab, N.; Naz, R.; Yasmin, H.; Nosheen, A.; Keyani, R.; Sajjad, M.; Hassan, M.N.; Roberts, T.H. Combined seed and foliar pre–treatments with exogenous methyl jasmonate and salicylic acid mitigate drought induced stress in maize. PLoS ONE 2020, 15, e0232269. [Google Scholar] [CrossRef]
- Anjum, S.A.; Tanveer, M.; Hussain, S.; Tung, S.A.; Samad, R.A.; Wang, L.; Khan, I.; ur Rehman, N.; Shah, A.N.; Shahzad, B. Exogenously applied methyl jasmonate improves the drought tolerance in wheat imposed at early and late developmental stages. Acta Physiol. Plant. 2016, 38, 25. [Google Scholar] [CrossRef]
- Yoshida, C.H.P.; Pacheco, A.C.; Lapaz, A.M.; Gorni, P.H.; Vítolo, H.F.; Bertoli, S.C. Methyl jasmonate modulation reduces photosynthesis and induces synthesis of phenolic compounds in sweet potatoes subjected to drought. Bragantia 2020, 79, 319–334. [Google Scholar] [CrossRef]
- Siboza, X.I.; Bertling, I. The effects of methyl jasmonate and salicylic acid on suppressing the production of reactive oxygen species and increasing chilling tolerance in ‘Eureka’ lemon [Citrus limon (L.) Burm F]. J. Hortic. Sci. Biotechnol. 2013, 88, 269–276. [Google Scholar] [CrossRef]
- Ahmmed, S.; Jahiruddin, M.; Razia, S.; Begum, R.A.; Biswas, J.C.; Rahman, A.S.M.M. Fertilizer Recommendation Guide-2018; Bangladesh Agricultural Research Council (BARC): Dhaka, Bangladesh, 2018. [Google Scholar]
- Ogbaga, C.C.; Stepien, P.; Johnson, G.N. Sorghum (Sorghum bicolor) varieties adopt strongly contrasting strategies in response to drought. Physiol. Plant. 2014, 152, 389–401. [Google Scholar] [CrossRef]
- Arnon, D. Copper enzymes isolated chloroplasts, polyphenol oxidase in Beta Vulgaris. Plant. Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Fischer, R.A.; Rees, D.; Sayre, K.D.; Lu, Z.M.; Condon, A.G.; Saavedra, L.A. Wheat yield progress associated with higher stomatal conductance and photosynthetic rate, and cooler canopies. Crop. Sci. 1998, 38, 1467–1475. [Google Scholar] [CrossRef]
- Shivakrishna, P.; Reddy, K.A.; Rao, D.M. Effect of PEG-6000 imposed drought stress on RNA content, relative water content (RWC), and chlorophyll content in peanut leaves and roots. Saudi J. Biol. Sci. 2018, 25, 285–289. [Google Scholar]
- Sairam, R.K.; Deshmukh, P.S.; Shukla, D.S. Tolerance of drought and temperature stress in relation to increased antioxidant enzyme activity in wheat. J. Agron. Crop. Sci. 1997, 178, 171–178. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teari, D. Rapid determination of free proline for water stress studies. Plant. Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Elstner, E.F.; Heupel, A. Inhibition of nitrite formation from hydroxylammoniumchloride: A simple assay for superoxide dismutase. Anal. Biochem. 1976, 70, 616–620. [Google Scholar] [CrossRef]
- Yang, S.-H.; Wang, L.-J.; Li, S.-H. Ultraviolet-B irradiation-induced freezing tolerance in relation to antioxidant system in winter wheat (Triticum aestivum L.) leaves. Env. Exp. Bot. 2007, 60, 300–307. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplast: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Mohi-Ud-Din, M.; Siddiqui, N.; Rohman, M.; Jagadish, S.V.K.; Ahmed, J.U.; Hassan, M.M.; Hossain, A.; Islam, T. Physiological and biochemical dissection reveals a trade-off between antioxidant capacity and heat tolerance in bread wheat (Triticum aestivum L.). Antioxidants 2021, 10, 351. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and susceptible method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Doderer, A.; Kokkelink, I.; van der Veen, S.; Valk, B.; Schram, A.; Douma, A. Purification and characterization of two lipoxygenase isoenzymes from germinating barley. Biochim. Biophys. Acta 1992, 112, 97–104. [Google Scholar] [CrossRef]
- Spitz, D.R.; Oberley, L.W. An assay for superoxide dismutase activity in mammalian tissue homogenates. Anal. Biochem. 1989, 179, 8–18. [Google Scholar] [CrossRef]
- Noctor, G.; Mhamdi, A.; Foyer, C.H. Oxidative stress and antioxidative systems: Recipes for successful data collection and interpretation. Plant Cell Environ. 2016, 39, 1140–1160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castillo, F.I.; Penel, I.; Greppin, H. Peroxidase release induced by ozone in sedum album leaves. Plant. Physiol. 1984, 74, 846–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elia, A.C.; Galarini, R.; Taticchi, M.I.; Dörr, A.J.; Mantilacci, L. Antioxidant responses and bioaccumulation in Ictalurus melas under mercury exposure. Ecotoxicol. Env. Saf. 2003, 55, 162–167. [Google Scholar] [CrossRef]
- Hossain, M.Z.; Hossain, M.D.; Fujita, M. Induction of pumpkin glutathione S-transferase by different stresses and its possible mechanisms. Biol. Plant. 2006, 50, 210–218. [Google Scholar] [CrossRef]
- Kolde, R. Pheatmap: Pretty Heatmaps, Version 1.0.12. rdrr.io 2019. Available online: https://rdrr.io/cran/pheatmap/ (accessed on 29 May 2021).
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Pardossi, A.; Incrocci., L.; Incrocci, G.; Malorgio, F.; Battista, P.; Bacci, L.; Rapi, B.; Marzialetti, P.; Hemming, J.; Balendonck, J. Root zone sensors for irrigation management in intensive agriculture. Sensors 2009, 9, 2809–2835. [Google Scholar] [CrossRef] [Green Version]
- Weng, E.; Luo, Y. Soil hydrological properties regulate grassland ecosystem responses to multifactor global change: A modeling analysis. J. Geophys. Res. 2008, 113, G03003. [Google Scholar] [CrossRef]
- Anjum, N.A.; Umar, S.; Ahmad, A. Oxidative Stress in Plant: Causes, Consequences and Tolerance; IK International Publishing House Pvt Ltd.: New Delhi, India, 2012. [Google Scholar]
- Huang, H.; Liu, B.; Liu, L.; Song, S. Jasmonate action in plant growth and development. J. Exp. Bot. 2017, 68, 1349–1359. [Google Scholar] [CrossRef] [Green Version]
- Abbaspour, H.; Saeidi–Sar, S.; Afshari, H. Improving drought tolerance of Pistacia vera L. seedlings by arbuscular mycorrhiza under greenhouse conditions. J. Med. Plant. Res. 2011, 5, 7065–7072. [Google Scholar] [CrossRef]
- Pandey, H.C.; Baig, M.J.; Bhatt, R.K. Effect of moisture stress on chlorophyll accumulation and nitrate reductase activity at vegetative and flowering stage in Avena species. Agric. Sci. Res. J. 2012, 2, 111–118. [Google Scholar]
- Dalal, V.K.; Tripathy, B.C. Water-stress induced downsizing of light-harvesting antenna complex protects developing rice seedlings from photo–oxidative damage. Sci. Rep. 2018, 8, 5955. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, H.I.; Latif, H.H. Improvement of drought tolerance of soybean plants by using methyl jasmonate. Physiol. Mol. Biol. Plants 2017, 23, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Razmi, N.; Ebadi, A.; Daneshian, J.; Jahanbakhsh, S. Salicylic acid induced changes on antioxidant capacity, pigments and grain yield of soybean genotypes in water deficit condition. J. Plant. Interact. 2017, 12, 457–464. [Google Scholar] [CrossRef]
- Royo, C.; Villegas, D.; Del Moral, L.G.; Elhani, S.; Aparicio, N.; Rharrabti, Y.; Araus, J.L. Comparative performance of carbon isotope discrimination and canopy temperature depression as predictors of genotype differences in durum wheat yield in Spain. Aust. J. Agric. Res. 2002, 53, 561–569. [Google Scholar] [CrossRef]
- Ahmed, H.G.M.-D.; Zeng, Y.; Yang, X.; Anwaar, H.A.; Mansha, M.Z.; Hanif, C.M.S.; Ikram, K.; Ullah, A.; Alghanem, S.M.S. Conferring drought-tolerant wheat genotypes through morpho-physiological and chlorophyll indices at seedling stage. Saudi J. Biol. Sci. 2020, 27, 2116–2123. [Google Scholar] [CrossRef]
- Mohi-Ud-Din, M.; Hossain, M.A.; Rohman, M.M.; Uddin, M.N.; Haque, M.S.; Ahmed, J.U.; Hossain, A.; Hassan, M.M.; Mostofa, M.G. Multivariate analysis of morpho-physiological traits reveals differential drought tolerance potential of bread wheat genotypes at the seedling stage. Plants 2021, 10, 879. [Google Scholar] [CrossRef]
- Balota, M.; Payne, W.A.; Evett, S.R.; Peters, T.R. Morphological and physiological traits associated with canopy temperature depression in three closely related wheat lines. Crop. Sci. 2008, 48, 1897–1910. [Google Scholar] [CrossRef] [Green Version]
- Kadioglu, A.; Saruhan, N.; Sağlam, A.; Terzi, R.; Acet, T. Exogenous salicylic acid alleviates effects of long–term drought stress and delays leaf rolling by inducing antioxidant system. Plant. Growth Regul. 2011, 64, 27–37. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycinebetaine and proline in improving plant abiotic resistance. Env. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Rampino, P.; Pataleo, S.; Gerardi, C.; Mita, G.; Perrotta, C. Drought stress response in wheat: Physiological and molecular analysis of resistant and sensitive genotypes. Plant. Cell Env. 2006, 29, 2143–2152. [Google Scholar] [CrossRef]
- Wang, G.P.; Zhang, X.Y.; Li, F.; Luo, Y.; Wang, W. Overaccumulation of glycine betaine enhances tolerance to drought and heat stress in wheat leaves in the protection of photosynthesis. Photosynthetica 2010, 48, 117–126. [Google Scholar] [CrossRef]
- Dixon, D.P.; Skipsey, M.; Edwards, R. Roles for glutathione transferases in plant secondary metabolism. Phytochemistry 2010, 71, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Fujita, M. Selenium pretreatment upregulates the antioxidant defense and methylglyoxal detoxification system and confers enhanced tolerance to drought stress in rapeseed seedlings. Biol. Trace Elem. Res. 2011, 143, 1758–1776. [Google Scholar] [CrossRef] [PubMed]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Fujita, M. Glutathione-induced drought stress tolerance in mung bean: Coordinated roles of the antioxidant defence and methylglyoxal detoxification systems. Aob Plants 2015, 7, plv069. [Google Scholar] [CrossRef] [Green Version]
- Nafie, E.; Hathout, T.; Mokadem, A.S.A. Jasmonic acid elicits oxidative defense and detoxification systems in Cucumis melo L. cells. Braz. J. Plant. Physiol. 2011, 23, 161–174. [Google Scholar] [CrossRef] [Green Version]

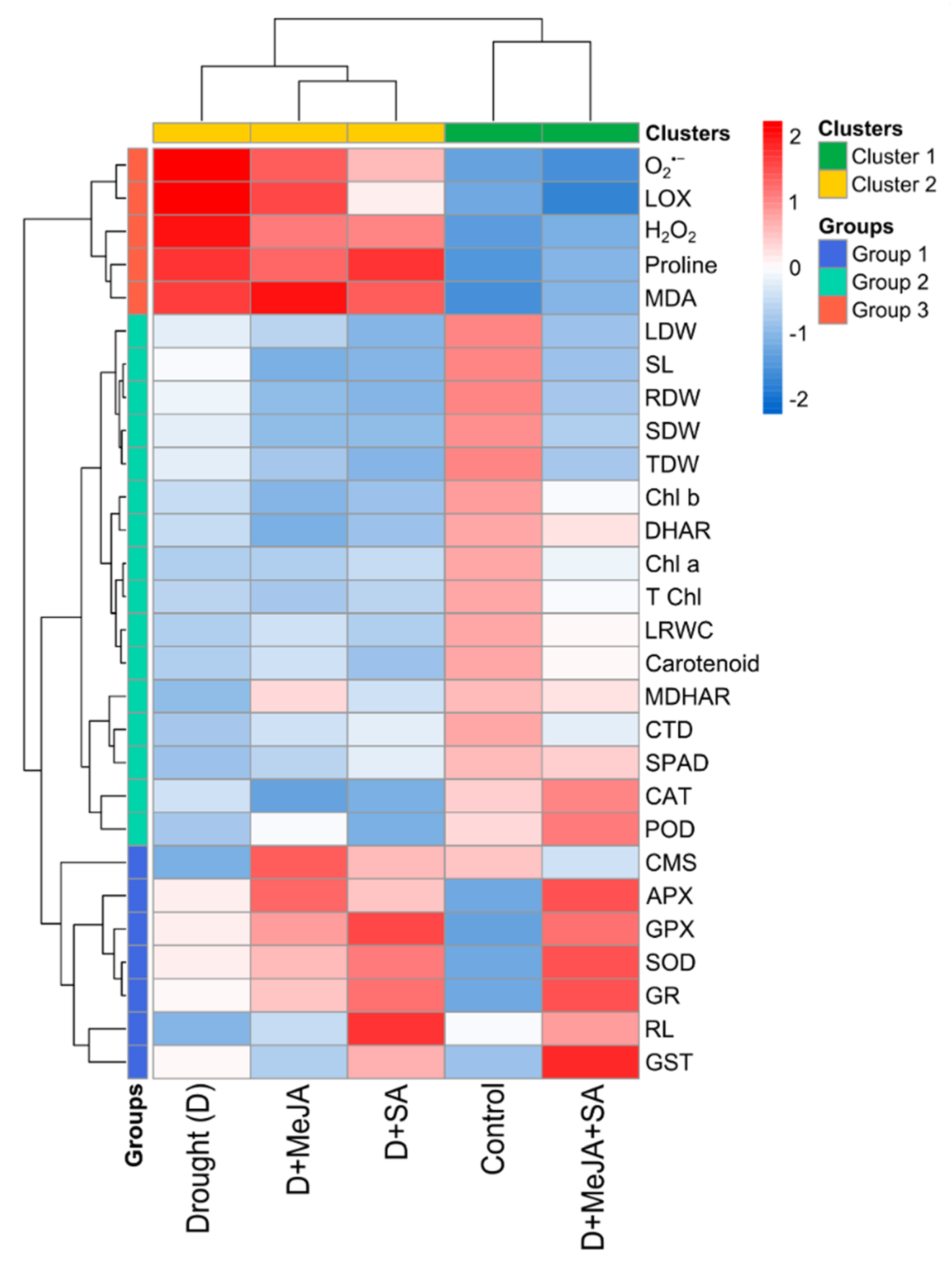
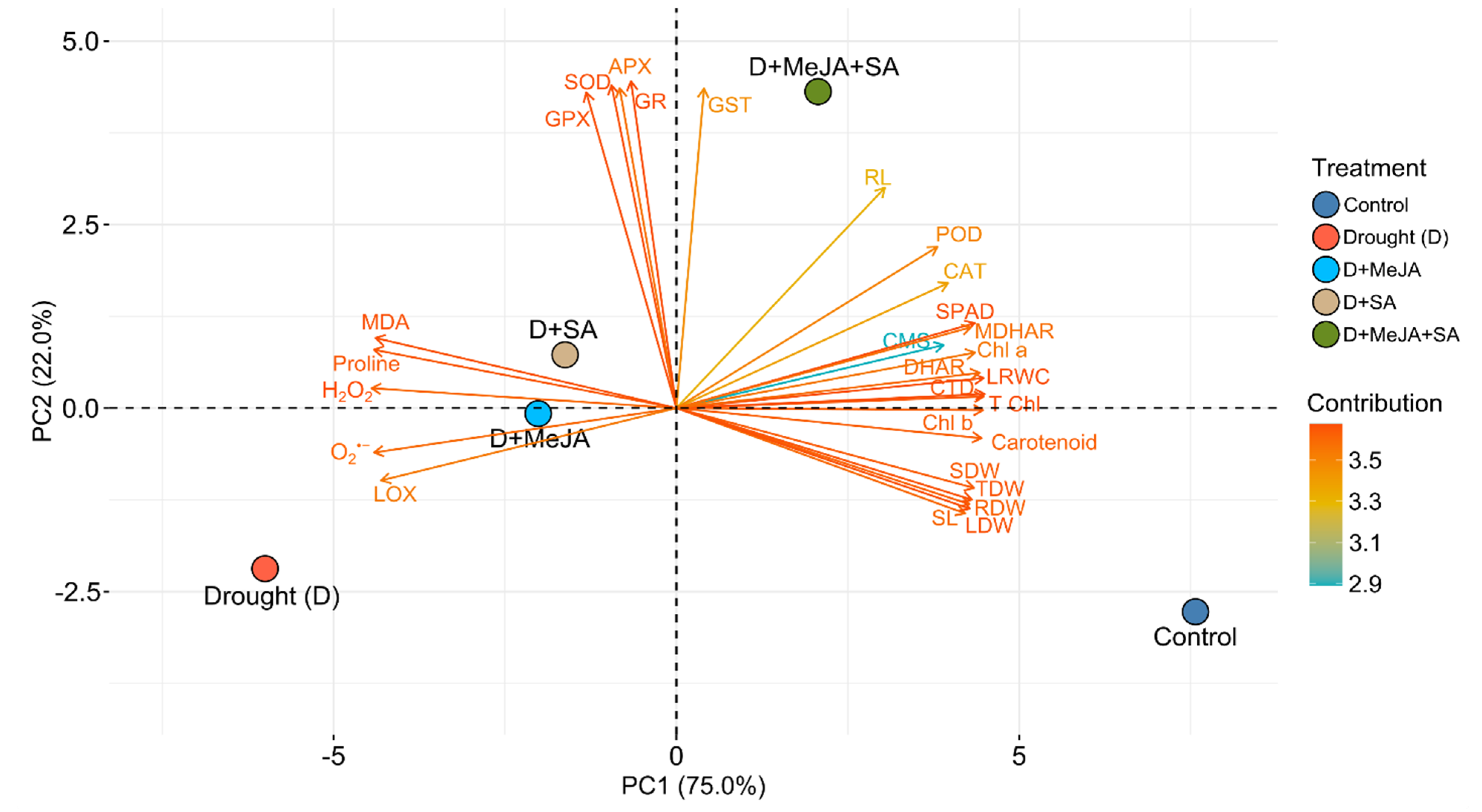
| Treatment | Root Length (cm) | Shoot Length (cm) | Root Dry Weight (g) | Shoot Dry Weight (g) | Leaf Dry Weight (g) | Total Dry Weight (g) |
|---|---|---|---|---|---|---|
| Control | 58.63 ± 3.45 a | 36.49 ± 0.87 a | 2.88 ± 0.26 a | 9.04 ± 0.74 a | 9.93 ± 0.77 a | 21.86 ± 1.62 a |
| Drought (D) | 55.69 ± 1.23 a | 21.27 ± 0.65 c | 1.38 ± 0.12 b | 2.77 ± 0.21 c | 2.61 ± 0.28 d | 6.76 ± 0.35 c |
| D + MeJA | 57.59 ± 2.13 a | 22.10 ± 0.49 c | 1.53 ± 0.17 b | 3.55 ± 0.19 c | 3.91 ± 0.37 b c | 8.99 ± 0.64 c |
| D + SA | 58.91 ± 2.55 a | 22.46 ± 0.63 c | 1.54 ± 0.18 b | 3.69 ± 0.49 c | 3.62 ± 0.13 c d | 8.85 ± 0.65 c |
| D + MeJA + SA | 59.81 ± 1.07 a | 25.63 ± 0.55 b | 1.86 ± 0.19 b | 5.09 ± 0.28 b | 4.89 ± 0.12 b | 11.84 ± 0.23 b |
| Treatments | SPAD | Leaf Pigments (mg g−1 Fresh Weight) | |||
|---|---|---|---|---|---|
| Chl a | Chl b | Total Chl | Carotenoids | ||
| Control | 49.85 ± 0.49 a | 1.39 ± 0.03 a | 0.49 ± 0.02 a | 1.97 ± 0.06 a | 0.50 ± 0.03 a |
| Drought (D) | 39.65 ± 1.18 c | 0.65 ± 0.10 c | 0.24 ± 0.05 c | 0.98 ± 0.07 c | 0.26 ± 0.01 c |
| D + MeJA | 43.65 ± 0.93 b | 0.87 ± 0.11 bc | 0.29 ± 0.04 bc | 1.25 ± 0.17 bc | 0.34 ± 0.05 bc |
| D + SA | 44.35 ± 1.52 b | 0.91 ± 0.16 bc | 0.30 ± 0.07 bc | 1.30 ± 0.25 bc | 0.33 ± 0.06 bc |
| D + MeJA + SA | 48.18 ± 0.94 a | 1.12 ± 0.06 b | 0.39 ± 0.02 ab | 1.61 ± 0.08 ab | 0.42 ± 0.03 ab |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohi-Ud-Din, M.; Talukder, D.; Rohman, M.; Ahmed, J.U.; Jagadish, S.V.K.; Islam, T.; Hasanuzzaman, M. Exogenous Application of Methyl Jasmonate and Salicylic Acid Mitigates Drought-Induced Oxidative Damages in French Bean (Phaseolus vulgaris L.). Plants 2021, 10, 2066. https://doi.org/10.3390/plants10102066
Mohi-Ud-Din M, Talukder D, Rohman M, Ahmed JU, Jagadish SVK, Islam T, Hasanuzzaman M. Exogenous Application of Methyl Jasmonate and Salicylic Acid Mitigates Drought-Induced Oxidative Damages in French Bean (Phaseolus vulgaris L.). Plants. 2021; 10(10):2066. https://doi.org/10.3390/plants10102066
Chicago/Turabian StyleMohi-Ud-Din, Mohammed, Dipa Talukder, Motiar Rohman, Jalal Uddin Ahmed, S. V. Krishna Jagadish, Tofazzal Islam, and Mirza Hasanuzzaman. 2021. "Exogenous Application of Methyl Jasmonate and Salicylic Acid Mitigates Drought-Induced Oxidative Damages in French Bean (Phaseolus vulgaris L.)" Plants 10, no. 10: 2066. https://doi.org/10.3390/plants10102066









