1. Introduction
Cabbage (
Brassica oleracea var.
capitata L.) belongs to the family Brassicaceae, which includes several economically important vegetable species grown worldwide. In Europe,
B. oleracea varieties such as cabbage, broccoli, and kale are gaining interest due to their high nutritional value and human health benefits [
1,
2]. Cabbage is a biennial plant that is self-incompatible and requires mandatory vernalization, which greatly affects the breeding process. Therefore, new approaches in plant breeding are needed to accelerate the production of new varieties with desirable agronomic traits.
Protoplasts, plant cells without a cell wall, are a unique system that can be used in basic research such as physiological and biochemical studies, and for new breeding strategies based on protoplast transformation or fusion [
3,
4]. Somatic hybridization by protoplast fusion has been used for the transfer of desirable resistance genes to diseases or other stress factors in many plants that cannot be crossed in a traditional way [
5,
6,
7]. In addition, protoplast-based transient transformation methods that rely on direct DNA uptake using polyethylene glycol (PEG) or electroporation are simple, highly efficient, and useful for numerous cell-based assays including the study of gene expression regulation, signaling pathways, protein interactions, etc. [
8]. Moreover, with the advent of genome editing techniques, protoplasts are gaining more interest as they offer many advantages over stable transformation methods using
Agrobacterium tumefaciens, which are time-consuming and laborious [
9]. Protoplast transformation has been used as a rapid screening method to validate genome editing reagents in many plant species [
10,
11,
12,
13,
14]. Moreover, they can also be regenerated into whole plants with targeted modifications [
13,
14,
15].
However, in order to apply protoplast technology in practice, the successful isolation of protoplasts is first required. Methods for the enzymatic isolation of protoplasts using cell wall degrading enzymes such as cellulases and pectinases were introduced by Cocking in 1960 [
16]. The most commonly used enzymes are Cellulase Onozuka RS and Pectolyase Y-23; the latter can also be replaced by Macerozyme R10 [
17]. For the optimal digestion of plant material, various factors such as the choice of enzymes and their concentrations, the duration of digestion, and the condition of the donor material must be tested [
3,
8,
17,
18]. The most common donor material is leaves, but protoplasts can also be isolated from petals or seedling organs such as hypocotyls or cotyledons [
18,
19]. In
B. oleracea L., protoplasts have been isolated from leaves and hypocotyls using different enzyme solutions. Commonly, Cellulase Onozuka RS at concentrations of 0.1% to 1% is used in combination with 0.1–1% Macerozyme R10 or 0.1% Pectolyase Y-23. Lower yields (1.8 × 10
4 to 0.8 × 10
6 protoplasts/g fw) have been reported for hypocotyls than for leaves (1.3 to 3.2 × 10
6 protoplasts/g fw) [
20,
21,
22,
23]. The latter are a good source of obtaining a high number of uniform cells. Moreover, when using in vitro plants, protoplasts can be isolated from micropropagated clones grown under controlled conditions, thus minimizing the negative effects of poor physiological conditions on isolation efficiency [
18].
Isolated protoplasts can be used in different transient assays, or if a regeneration protocol is available, plants with improved traits can be obtained by protoplast fusion or transformation. However, protoplast regeneration is a very complex process involving many steps such as cell wall recovery, cell cycle re-entry, callus formation, and de novo tissue regeneration [
24]. It also depends on many factors including the protoplast isolation protocol, culture conditions, concentration, and combination of plant growth regulators in culture media, etc., which need to be optimized to achieve plant regeneration from protoplasts [
3,
18,
19,
25]. In the initial phase, high osmotic pressure is required to maintain the protoplast’s integrity before the cell wall is rebuilt, and growth regulators such as auxins and cytokinins are essential for protoplast division. However, during culture, the requirements of protoplasts may change, and the composition of the medium must be adjusted [
3,
26]. For this reason, a liquid medium is usually used for the culture of protoplasts. Liquid media based on Murashige and Skoog [
27] or Kao and Michayluk [
28] with modifications are usually used for the cultivation and regeneration of Brassica protoplasts [
23,
29,
30,
31,
32,
33]. To prevent the agglutination of protoplasts and promote mitotic divisions, protoplasts can be immobilized in agarose or alginate [
3,
18,
19], an approach often used for
B. oleracea L. protoplasts [
20,
22,
23,
29].
Several protocols for successful protoplast regeneration have been published, mainly for the Solanaceae and Poaceae families [
24,
34,
35,
36]. In Brassicaceae, protoplast research has mainly focused on
B. napus L. due to its importance in oilseed production [
8,
26,
37,
38,
39]. Sahab et al. [
8] published a detailed protocol for the isolation of leaf mesophyll protoplasts of
B. napus and their regeneration after PEG-mediated transformation. Although there are also publications on cabbage, plant regeneration remains limited to a few cultivars [
23,
29]. Recently, the effects of supplements such as peptidyl growth factors (phytosulfokines) and polyamines in the culture medium on the division frequency of protoplasts have been studied, but despite the modifications, plant regeneration from cabbage protoplasts remains low [
30,
31,
32].
In this study, factors affecting protoplast regeneration such as the composition of the enzyme solution (concentration and choice of cell-wall digesting enzymes) for successful protoplast isolation and different culture protocols were tested. In addition, the protoplast-to-plant regeneration potential in five different cabbage cultivars was investigated.
3. Discussion
Traditional breeding of
B. oleracea varieties has led to significant improvement in productivity and quality [
40,
41], but to overcome the limitations of current breeding programs and meet the needs of an ever-growing population, the application of novel technologies is necessary. Many protoplast-based approaches have already been used in plant research including genome editing using CRISPR/Cas9 [
4,
25]. Nevertheless, there are obstacles associated with protoplast regeneration, and in many plant species, efficient protoplast regeneration remains a technical barrier [
4]. In addition, isolating a sufficient amount of highly viable protoplasts is also a challenge in some plant species [
42].
In protoplast isolation, the selection of enzymes and their concentration play a crucial role as they directly affect the efficiency and viability of the isolated protoplasts [
3,
18,
43]. To obtain a high yield of viable cabbage protoplasts, the effect of different concentrations of Pectolyase Y-23 on the quantity and quality of isolated protoplasts was first tested. Leaves of sterile in vitro plants were used in all experiments to reduce the negative effects of uncontrolled growth conditions and the presence of endogenous bacteria, as suggested by Moon et al. [
43]. While there were no significant differences in the yield of isolated protoplasts, higher concentrations of Pectolyase Y-23 affected the viability. Increasing the enzyme concentration can result in higher yields, but excess enzyme can cause phytotoxicity and consequently reduce the viability [
44,
45], which was also evident in our experiments.
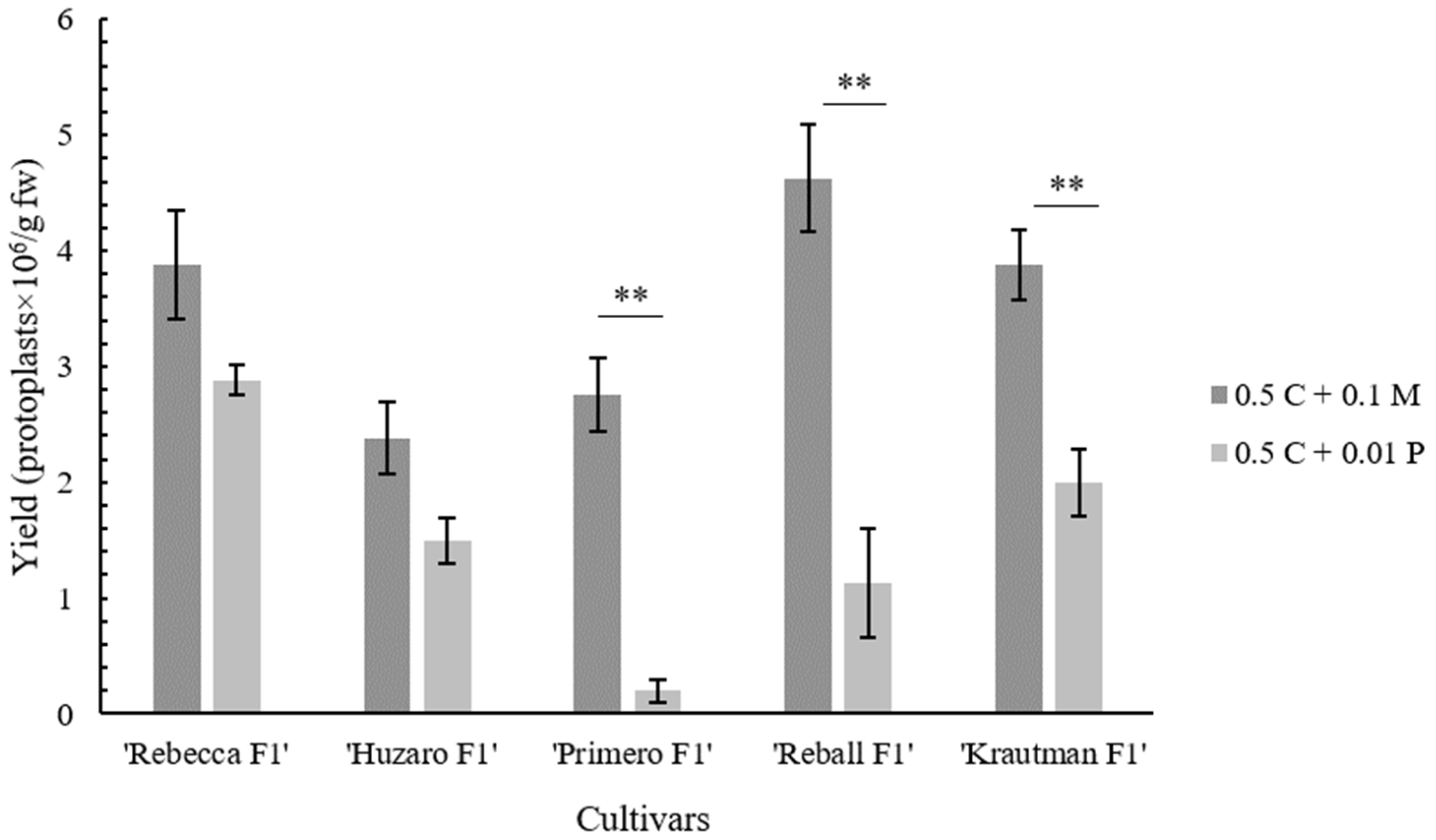
Macerozyme R-10 instead of Pectolyase Y-23 was then used in enzymatic solution and found that the replacement of Pectolyase Y-23 with Macerozyme R-10 improved the isolation efficiency by more than twofold to 4.63 × 10
6 protoplasts/g fw. Similar results were reported by Kiełkowska and Adamus [
23], but they obtained a lower yield (2.1 × 10
6 protoplasts/g fw) with a higher (1%) concentration of Cellulase Onozuka RS. The enzyme solution optimized in this study with Cellulase Onozuka RS and Macerozyme R-10 at concentrations of 0.5% and 0.1%, respectively, was suitable for the isolation of protoplasts with high viability from the leaves of all five different cabbage cultivars. This demonstrates the robustness and wide applicability of the isolation protocol optimized in this study.
In some plant species, higher isolation efficiencies (up to 1 × 10
7 protoplasts/g fw) were obtained by adjustments to the isolation protocol [
46,
47]. In
Arabidopsis, the so-called “Tape-
Arabidopsis Sandwich” isolation method has been developed, which does not require slicing of the leaves, which is time-consuming and can damage the cells. Instead, the epidermis is peeled off with tape and the mesophyll cells are exposed to the digesting enzymes. This approach exposes a larger surface area to the enzymes, resulting in the release of a larger amount of protoplasts and a shorter isolation procedure [
46]. This protocol was also applied to other Brassicaceae species such as
B. oleracea and
B. napus by Lin et al. [
24], but the yield of the obtained protoplasts was not disclosed. Nevertheless, protoplasts were used for PEG transformation, and successful genome editing by CRISPR/Cas9 was reported.
Purification of viable protoplasts by sucrose gradient centrifugation was used in all of the experiments to remove the cell wall debris and dead cells that could have negative effects and inhibit protoplast division and development [
25,
43]. To prevent the aggregation of protoplasts during culture, they were immobilized in alginate layers right after isolation, which has been reported to promote cell division and improve the plating efficiency [
23,
43,
48,
49].
The composition of protoplast culture media and the culture protocol are key factors influencing the division of protoplasts and their regeneration into plants. For this reason, three different protocols for the culture of cabbage protoplasts that differ in osmotic and plant growth regulators were examined. The latter are crucial in initial protoplast culture [
25,
26]. In all of the protocols tested, 2,4-D was used as recommended for Brassicaceae protoplasts [
50]. 2,4-D was also essential for the cell wall formation and initial growth of
B. napus protoplasts [
26]. Cell divisions of protoplasts of all cultivars could be observed in the first two weeks, but micro-calli from the ‘Primero F1’ protoplasts was produced only by Protocol 3, while Protocols 1 and 2 were more suitable for micro-calli induction in all other cultivars. The choice of osmotic stabilizer may have had an influence here. In Protocol 3, myo-inositol was used together with sucrose, as opposed to glucose, the main osmotic stabilizer in Protocols 1 and 2. Myo-inositol was proposed by Jie et al. [
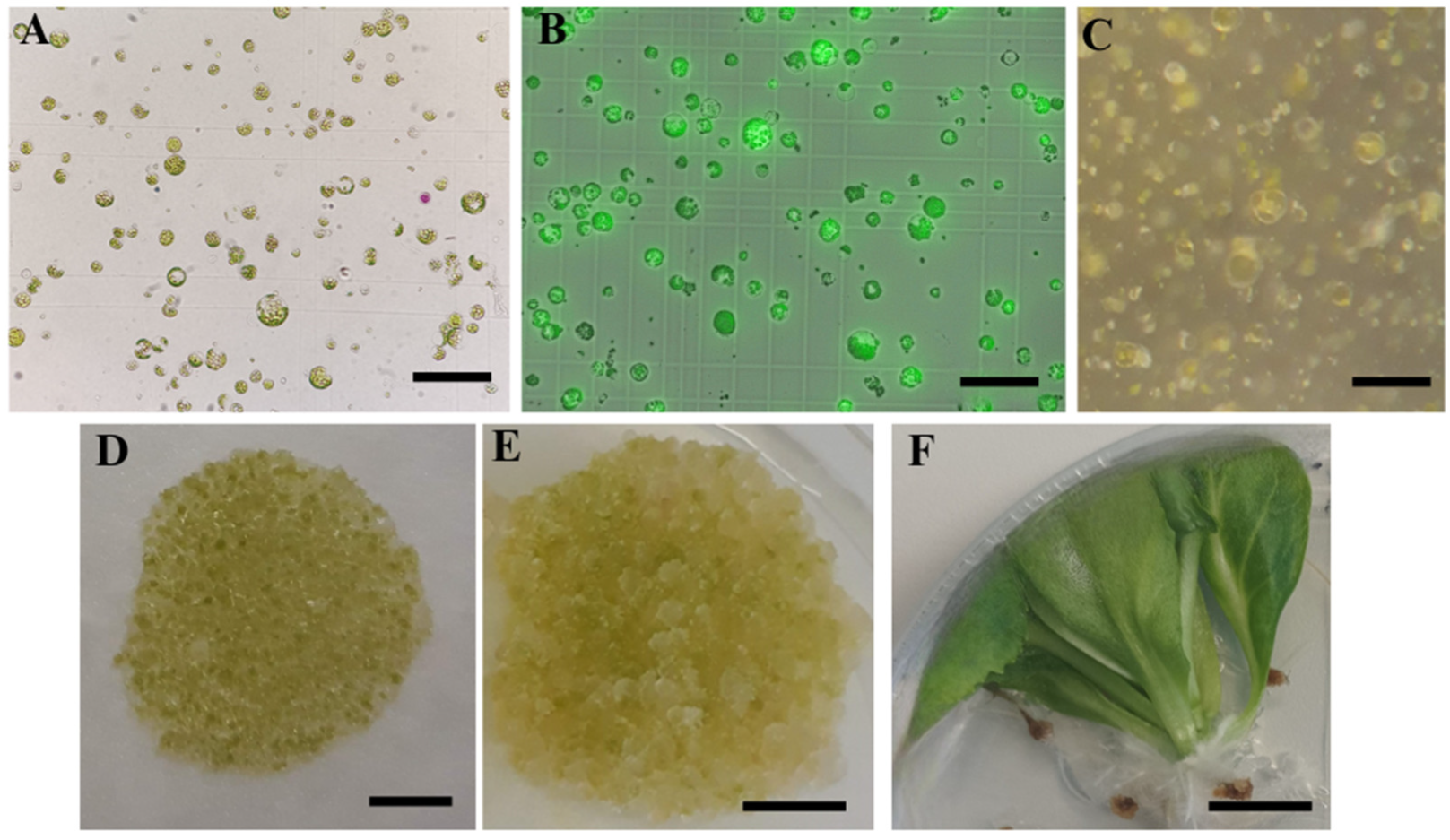
33] as an osmotic regulator in the culture of cabbage protoplasts, but proved to be unsuitable in our experiments as only a low-density micro-calli was obtained, which did not proliferate further. In Protocol 2, the osmotic and plant growth regulators were gradually changed during cultivation, but no differences in the efficiency of micro-calli induction could be observed compared to Protocol 1.
It has been shown several times that the genotype plays a significant role in the response of
B. oleracea protoplasts [
23,
29,
30,
31]. In this study, all of the tested cultivars produced micro-calli, but only protoplasts of ‘Krautman F1’ formed shoots. It has been reported that the optimal protoplast culture medium may vary among cultivars [
19]. Surprisingly, in our experiments, no shoots were obtained from the protoplasts of ‘Reball F1’, which is considered one of the responsive cabbage cultivars [
23,
30]. One of the possible reasons could be the too high density of protoplasts. Andersson et al. [
35] and Moon et al. [
43] reported that too low or too high protoplast density can inhibit shoot regeneration. This hypothesis could be confirmed since ‘Reball F1’ formed micro-calli with high density but no shoots, while shoots were obtained from the cultivar ‘Krautman F1’, which formed micro-calli with lower density. Micro-calli obtained from ‘Reball F1’ and ‘Krautman F1’ also differed in size, which was inversely proportional to their number. In cauliflower protoplasts, micro-calli of at least 2 mm in size transferred to a solid medium for successful shoot regeneration [
51].
Shoot regeneration usually requires lower auxin and higher cytokinin levels in Brassicaceae protoplasts [
26], while Kiełkowska and Adamus [
23] used only cytokinin or even a medium without growth regulators [
31]. The addition of auxin (0.2 mg/L NAA) rather than using only cytokinin BAP had a better effect on shoot formation in our experiments when protoplasts were cultured according to Protocol 1. To further optimize the shoot regeneration from cabbage protoplasts, different growth regulators could be tested. Li et al. [
26] used ten different combinations and obtained plants in four of them with the highest shoot regeneration of 45.0% on medium supplemented with 2.2 mg/L of TDZ and 1.0 mg/L NAA.