1. Introduction
The soil seed bank is usually considered as the natural reserve of all viable seeds in a soil profile and on the soil surface [
1]. Many seeds may remain viable though dormant in the soil for years. According to persistence time in the soil, soil seed banks are categorized as transient seed banks (persist for less than 1 year), short persistent seed banks (persist for 1–5 years) and long-term persistent seed banks (persist for more than 5 years) [
2]. As a potential source of genetic diversity for plant populations, soil seed banks are vital for vegetation succession, vegetation maintenance, ecosystem restoration, species diversity conservation, and population management strategies [
3,
4,
5]. Therefore, it is of great value to study the species composition, and temporal and spatial dynamics, of soil seed banks to develop comprehensive weed management, sustainable agriculture, and conservation strategies.
Biological invasions have been become one of the main factors leading to biodiversity loss and environment change on a global scale [
6,
7,
8]. The presence of seed banks has been regarded as one of the key components of successful biological invasion for many species [
9]. The invasiveness of many exotic plant species is frequently associated with high proliferation and the production of seeds that may persist in the soil for long periods of time [
10]. The successful establishment of invasive plants is determined in many cases by the production of a persistent soil seed bank in newly invaded areas, given the role of seed banks as sources of propagules, genetic diversity, and in spreading the risk of germination failure over time [
9]. Therefore, studies on the soil seed bank of an invasive plant species can provide valuable information on the invasiveness of a species for planning restoration measures and for developing comprehensive prevention and control measures. This is especially important for invasive alien plant species that are new to a geographic area for which information on seed bank dynamics may be lacking.
Acmella radicans (Jacquin) R.K. Jansen is an annual erect herb in the Asteraceae family that originated in central America and Mexico [
11]. This plant has become broadly distributed in Colombia, Bangladesh, Cuba, Curacao, India, Tanzania, and Thailand [
11,
12,
13]. A naturalized population was first recorded in China in 2014 in Anhui Province [
14]. In 2017,
A. radicans was first found in Changning County, Baoshan City of Yunnan Province in southwestern China during a survey of invasive alien plant species. Currently,
A. radicans has become a serious invasive species and has invaded farmland, tea gardens, orchard land, roadsides, and ditches in Baoshan City, Lincang City and Puer City of Yunnan [
15].
Acmella radicans has become a dominant species in invaded regions, negatively affecting species richness, species diversity, and the evenness and soil nutrients of local communities. This plant can release allelochemicals such as (E, E)-2,4-decadienal, 2-tridecanone, and caryophyllene oxide to inhibit the seed germination and seedling growth of some major associated weeds and local vegetable crops such as
Ageratum conyzoides,
Bidens pilosa,
Brassica napus,
Chloris virgata, and
Digitaria sanguinalis [
15,
16]. Furthermore,
A. radicans demonstrates considerable ecological adaptability, occupies a wide range of habitat types, possesses a long flowering and fruiting period (November–March), and exhibits a prolific reproductive capacity (a single plant can produce up to 14,300 seeds) [
15]. However, little is known about the soil seed bank in communities invaded by
A. radicans.
In the current study, the composition, density, species richness, diversity indices, and evenness index of soil seed bank communities of A. radicans in two different habitats (wasteland and cultivated land) in Yunnan Province, China, were examined through field sampling and greenhouse tests. Seed bank characterization is important to better elucidate its invasion and spread mechanisms and provide a scientific basis for the forecasting, risk analysis, prevention, and control of A. radicans.
3. Discussion
Increasing numbers of studies have demonstrated that invasive alien plants can reduce species richness, species diversity indices, and evenness indices through strong ecological adaptability, competitiveness and allelopathic inhibition, altering underground and aboveground composition and the structure of plant communities in invaded habitats [
17,
18,
19,
20]. As a new invasive species in Yunnan province,
A. radicans is already widely distributed in Baoshan, Lincang and Puer cities, causing serious damage to local agricultural production and ecological environments [
15]. However, the effect of
A. radicans invasion on soil seed bank characteristics was previously unclear. Our current study found that
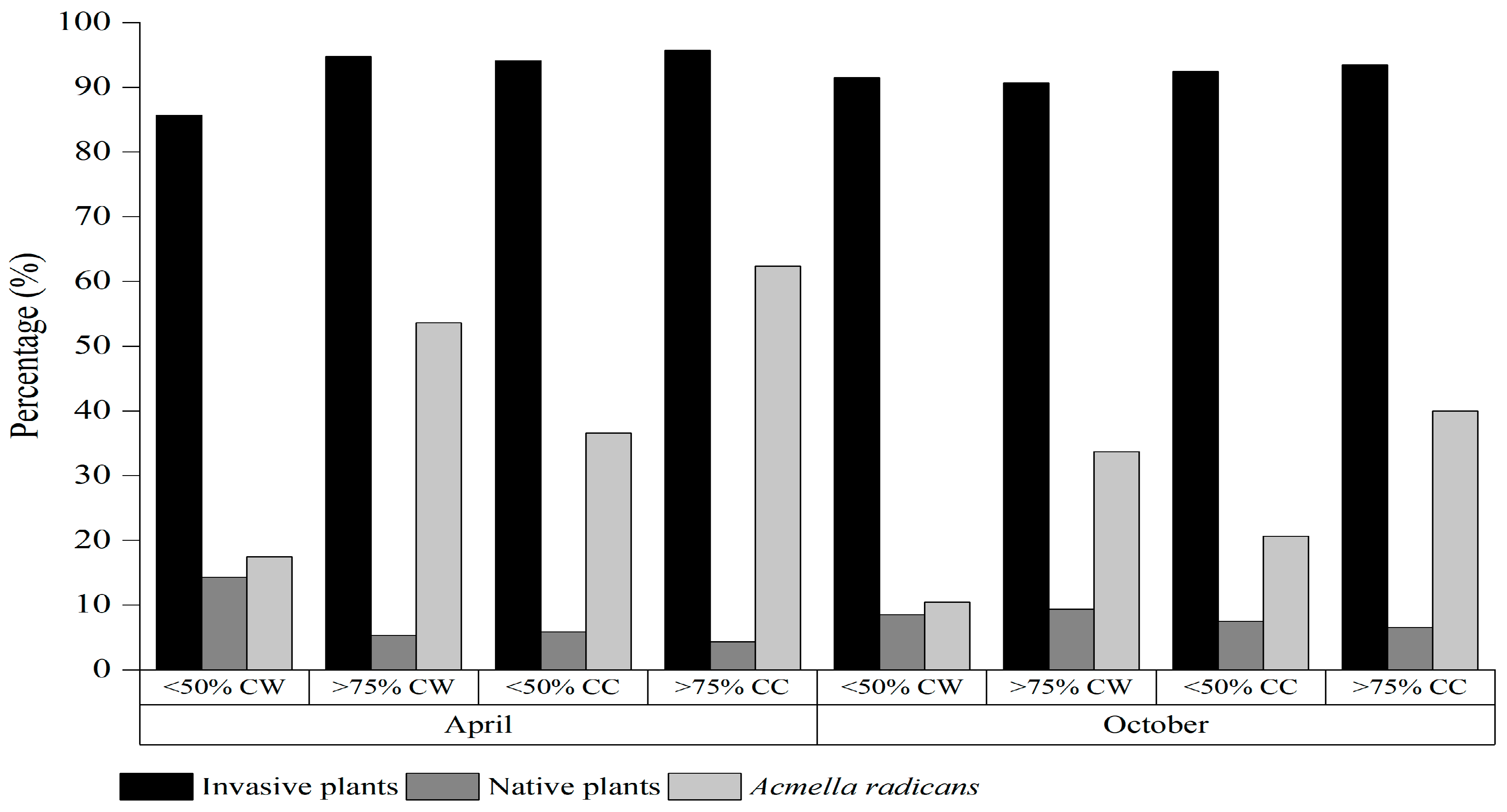
A. radicans has modified the species composition, population density and weed community diversity of the soil seed bank in the invaded area, and human disturbance in cultivated land has aggravated these effects.
The soil seed bank is generally considered to be an important potential seed source for the invasion and spread of invasive alien plants and plays a critical role in vegetation succession and species diversity maintenance [
17,
18]. Characterizing the seed bank of invasive alien plants informs an understanding of the type, magnitude and rate of change in the seed bank and predicts the recruitment potential of these species from the seed bank [
21]. The similarity between soil seed banks and the standing vegetation of invasive plants is usually high at the initial invasion stage and gradually declines over time [
17,
18]. Our current study found that the species richness of the soil seed bank is clearly higher than that of the aboveground vegetation, while the seed bank population is markedly smaller than the seed rain from the aboveground vegetation in
A. radicans invaded habitats. A major factor contributing to this disparity is that a large proportion of the seed rain was transported to other places by wind and other disturbances. A second major factor is related to the invasion history and the storage characteristics of other plant seeds. As the invasion duration and resulting damage increases, invasive alien plants will gradually come to occupy a dominant position in both the local vegetation and the soil seed bank, while reducing the population density of other plants in the community, especially native species [
22,
23,
24,
25]. The current study found that as the seed density of
A. radicans significantly increased, seed densities of many other dominant invasive plants and native plants clearly declined. The
A. radicans seed density corresponded to increases in
A. radicans cover, which was partly due to the strong competitiveness and allelopathic inhibition of
A. radicans [
15,
16]. Moreover, with increased cover of
A. radicans, the seed density of other invasive plants as well as
A. radicans in study plots was markedly increased. The proportion of invasive species in the seed bank was significantly higher in cultivated land than in wasteland under the same
A. radicans cover, indicating that
A. radicans had higher negative impacts on native species than the other invasive alien species present, and that the weed community in cultivated land was affected by both the stress of
A. radicans invasion and human disturbance. Local plant species and community structure are more vulnerable to disturbance than those of invasive alien plants under the same conditions because invasive species usually have greater morphological plasticity and competitiveness [
26]. Synergistic impacts may result when adventive invasive alien plant species and disturbances coincide that are conducive to both the invasion of the adventive species and expansion of other invasive species [
27].
Invasive alien plants not only change species composition and population density of weed communities in invaded habitats but also affect the species diversity. Zheng et al. [
28] reported that the negative correlation between the importance value of
Spermacoce latifolia and the species diversity of the community indicated that the invasion and spreading of
S. latifolia may have a negative impact on the species diversity of the community. Nan et al. [
18] found that the importance value of
Oenothera laciniata in the community had a positive correlation with the species diversity of the community but this was not significant. Robertson and Hickman [
29] found that as
Bothriochloa spp. invasion increases, the native plant community decreased in diversity and abundance, but no effect on native seed bank diversity and density was observed. Our previous study found that
A. radicans had negative impacts on species richness, species diversity, and evenness and soil nutrients of local communities [
15]. Furthermore, we found that the species richness, Simpson index, Shannon–Wiener index, and Pielou index of the weed community usually increased as the
A. radicans cover declined, and most diversity indices of weed communities in cultivated land were lower than in wasteland under the same cover of
A. radicans, showing that the invasion of
A. radicans has a negative impact on the diversity of local weed communities and that human disturbance in cultivated land may aggravate this trend. In our study area, there were few human activities (never grazed and/or cultivated) since 2002 in the wasteland habitat, indicating that the decline in species diversity of the weed community was directly caused by
A. radicans invasion. The more pronounced decline in weed community species diversity in cultivated land was caused by the combination of
A. radicans invasion and human disturbance. Other studies have reported that the invasion of exotic plant species and native species suppression were increased by numerous disturbances [
30,
31].
Invasive alien plants adapt to different environmental conditions and changes with various reproductive strategies during their life history [
32]. Soil seed banks may be the most tolerant to a wide range of environmental conditions by comparison to other life history elements because many seeds may be dormant or remain viable in the soil over the long term [
2,
33]. The soil seed bank is heterogeneously distributed in soil wherein the topsoil layer is usually transient seed and has the highest proportion of the seed bank, while the proportion of the seed bank gradually decreases and becomes more of a persistent seed bank with increases in soil depth [
34,
35,
36].
Acmella radicans is an annual herb, with a five-month flowering and fruiting period, producing up to 14,300 seeds per plant [
15]. Its high fecundity is critical to the growth and spread of its populations. The present study showed that
A. radicans seeds in the 0–5 cm, 5–10 cm and 10–20 cm layers of cultivated land accounted for 56.8–64.9%, 26.7–31.8% and 8.1–13.5% of the total seed density, respectively. In wasteland, most
A. radicans seeds were stored in the 0–5 cm layer, accounting for 80.7–90.6% of the seed bank. The seed densities of
A. radicans substantially increased with increases in the cover of
A. radicans in both habitats, and the seed density of
A. radicans in cultivated land was significantly higher than in wasteland under the same
A. radicans cover. It is clear that
A. radicans forms a large proportion of its persistent soil seed bank in deep soil in cultivated land, potentially contributing to future recruitment, especially if facilitated by human disturbance. Abundant transient and persistent seed banks of
A. radicans are conducive to maintaining relatively high populations or even undergoing large outbreaks under the right conditions. Moreover, the persistent seed bank ensures
A. radicans is not threatened with extirpation under adverse environment and human disturbance.
Considering the plant growth, reproduction, and soil seed bank characteristics of A. radicans under natural conditions, it is critical to adopt a long-term and comprehensive plan for its prevention, control and management. As an annual herb, A. radicans completes its life history and sexual reproduction within a single year. Thus, in order to reduce or avoid the seed dispersal of A. radicans, the optimum time for prevention and control is before flowering or at least prior to seed maturation with efficient herbicides or elimination by physical control techniques. In addition, for managing the transient and/or persistent soil seed bank A. radicans, a reduction in human activities or the use of soil herbicides to limit the seed germination after crop planting could be effective. Finally, prevention and control measures such as no-tillage mulching, plastic film mulching, intercropping, and replacement techniques could be combined, which would serve not only to inhibit the number of A. radicans effectively but also produce excellent ecological and economic benefits. Therefore, comprehensive measures must be considered for the prevention and control of A. radicans, incorporating chemical herbicides supplemented by physical control, ecological restoration, and replacement control. It is possible to control or reduce the extent of invasion by A. radicans, but this would require several years of continuous prevention and control measures.