Abstract
Hydrogen peroxide (H2O2) is relatively stable among ROS (reactive oxygen species) and could act as a signal in plant cells. In the present work, detached tomato leaves were treated with exogenous H2O2 at 10 mmol/L for 8 h to study the mechanism of how H2O2 regulates leaf senescence. The data indicated that H2O2 treatment significantly accelerated the degradation of chlorophyll and led to the upregulation of the expression of leaf senescence-related genes (NYC1, PAO, PPH, SGR1, SAG12 and SAG15) during leaf senescence. H2O2 treatment also induced the accumulation of H2O2 and malondialdehyde (MDA), decreased POD and SOD enzyme activities and inhibited H2S production by reducing the expression of LCD1/2 and DCD1/2. A correlation analysis indicated that H2O2 was significantly and negatively correlated with chlorophyll, the expression of leaf senescence−related genes, and LCD1/2 and DCD1/2. The principal component analysis (PCA) results show that H2S showed the highest load value followed by O2•−, H2O2, DCD1, SAG15, etc. Therefore, these findings provide a basis for studying the role of H2O2 in regulating detached tomato leaf senescence and demonstrated that H2O2 plays a positive role in the senescence of detached leaves by repressing antioxidant enzymes and H2S production.
1. Introduction
Senescence is the final stage of plant development, which includes two types of senescence: mitotic and post-mitotic senescence [1]. Leaves are organs that characterize plants as autotrophic organisms and leaf senescence is a kind of post-mitotic senescence. As leaves undergo senescence, chlorophyll degradation is initiated and leads to chloroplast degeneration. Additionally, macromolecules, including proteins, lipids and nucleic acids, are also catabolized to small molecules which are exported to other developing organs, such as new buds, flowers, fruits, etc. Chloroplasts constitute about 70% of the total proteins in leaves, and thus the components’ catabolism is critical for C/N mobilization. Chlorophyll degradation causes the first visible changes of leaf senescence. Chlorophyll breakdown is initiated from the conversion of chlorophyll b to chlorophyll a, which is catalyzed by two chlorophyll b reductases, NONYELLOW COLORING 1 (NYC1) and NYC1-like (NOL). Additionally, STAYGREEN (SGR), Pheophytin Pheophorbide Hydrolase (PPH) and Pheophorbide a Oxygenase (PAO) also participate in chlorophyll degradation [2]. Leaf senescence is a highly coordinated process regulated by hundreds of senescence-associated genes (SAGs) whose transcripts increase with leaf age [2]. Among them, SAG12 and SAG15 are the predominant SAGs that are up-regulated significantly during age-related senescence [3].
Many phytohormones function to integrate environmental or developmental signals to regulate plant senescence. Ethylene is a gaseous signal that could promote leaf senescence and fruit ripening. Additionally, abscisic acid (ABA), a plant hormone regulating plants’ responses to abiotic and biotic stresses, shows increases during leaf senescence and the exogenous application of ABA could trigger leaf senescence [4]. In addition to the traditional phytohormones, reactive oxygen species (ROS) are indispensable for plant senescence [5]. ROS are produced as byproducts of aerobic energy metabolism and the excessive accumulation of ROS can lead to oxidative stress and, consequently, to damage to macromolecules and membranes [6]. The ROS, including singlet oxygen (1O2), superoxide radical (O2•−), hydrogen peroxide (H2O2), etc., are extremely reactive and H2O2 can efficiently oxidize the active thiols and travel long distances by passing through the biological membranes via aquaporins; since H2O2 is more stable compared to other ROS and easily diffuses across membranes between different cellular compartments, it could act as a signaling molecule to transmit external signals to intracellular pathways [7]. H2O2 plays an important role during plant senescence, and it could be used as a signal to promote senescence in different plant species [8,9]. At high concentrations, it could lead to cell death during the final stages of senescence. Chloroplasts, an important source of ROS in photosynthetic plant cells, are also targets of ROS-induced damages [10]. H2O2 application was found to induce the expression of the NAC transcription factor ORS1, which triggers the expression of senescence-associated genes and accelerates senescence [11]. However, whether and how H2O2 application affects the senescence of detached tomato leaves is still unclear.
Hydrogen sulfide (H2S) exposure at high concentrations is highly toxic to all animals. However, H2S was found to be produced endogenously in animal cells and plants [12]. In recent decades, H2S was found to regulate multiple aspects of plant growth and development, such as seed germination, root formation, stomatal movement, etc. [13,14]. Additionally, H2S could effectively delay fruit ripening and senescence by repressing ethylene synthesis and signaling pathways in strawberries, tomatoes, pears, etc. [15,16,17]. Moreover, H2S could activate the activities of antioxidant enzymes such as catalase (CAT), superoxide dismutase (SOD) and peroxidase (POD), which may help to alleviate ROS stress during fruit ripening and senescence [16]. However, there is no clue whether H2O2 interferes with the generation of H2S. Thus, in the present work, H2O2 was applied to detached tomato leaves and its effects on leaf senescence and H2S metabolism were explored.
2. Results
2.1. Effect of H2O2 Treatment on Detached Tomato Leaf Senescence
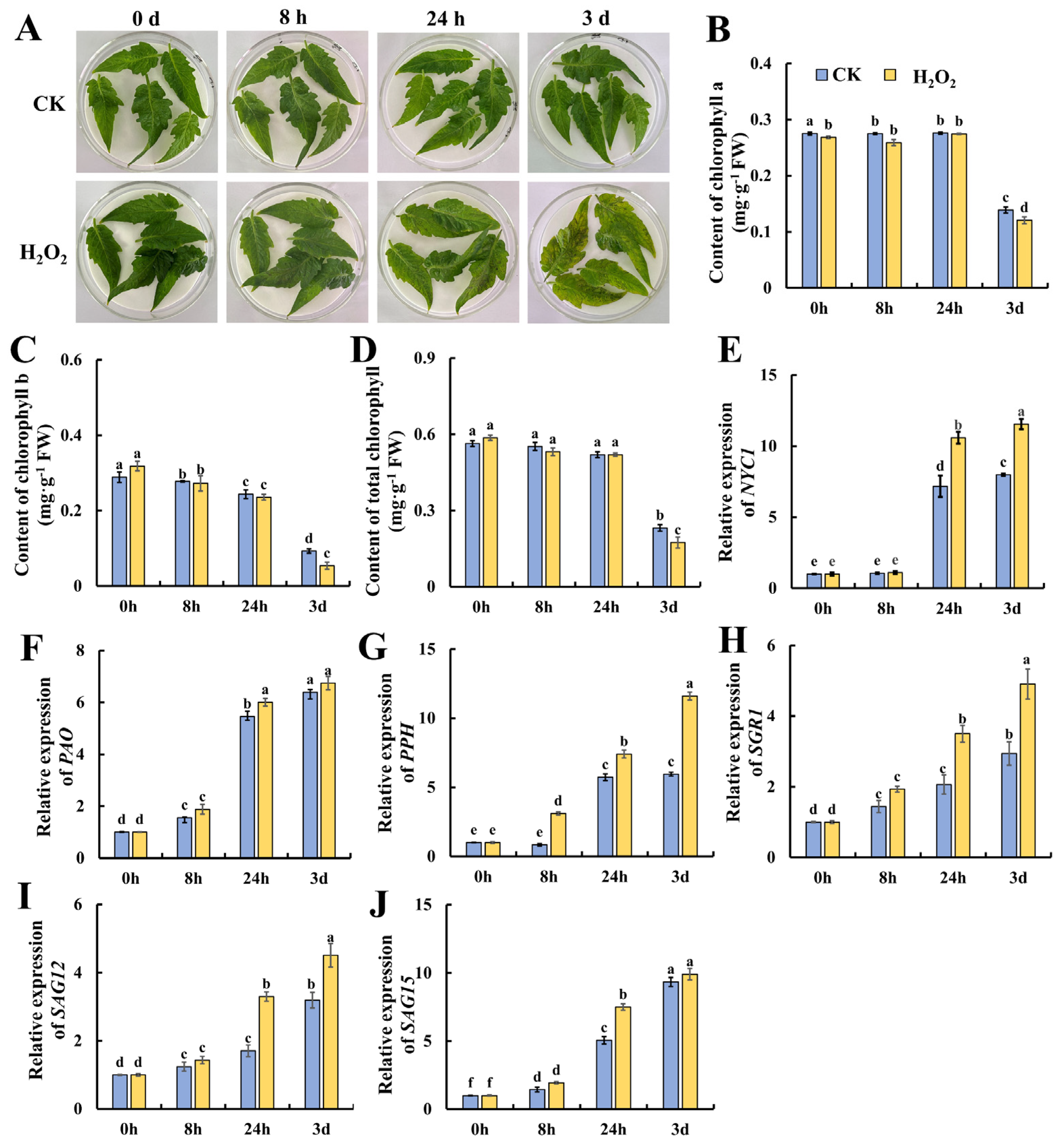
H2O2 is a potent alternative that is used as a sanitizing agent for horticultural products, whereas whether it could promote leaf senescence is still unclear. Thus, we treated mature tomato leaves with 10 mmol/L of H2O2 for 8 h, and subjected them to storage for another 3 days. As shown in Figure 1A, after 24 h of storage, H2O2 induced yellow and brown spots on the leaves which became more obvious compared with the control leaves on day 3. Then, the contents of chlorophyll a, b and total chlorophyll were determined. As shown in Figure 1B, chlorophyll a content was almost kept at a relative stable level till 24 h of storage, followed by a significant decrease on day 3 in both the H2O2-treated and control leaves. As shown in Figure 1C, the content of chlorophyll b decreased gradually till 24 h and dropped on day 3, suggesting that the degradation of chlorophyll b preceded that of chlorophyll a. Additionally, H2O2 treatment caused significantly lower chlorophyll b and total chlorophyll contents in contrast with the control, suggesting that H2O2 elicits tomato leaf senescence during storage. Then, the genes evolved in chlorophyll degradation, including NYC1, PAO, PPH, SGR1, SAG12 and SAG15, were determined at the transcriptional level. NYC1, PAO and PPH are key chlorophyll-degrading enzymes during leaf senescence. Additionally, the SGR (STAY GREEN) protein affects chlorophyll degradation by interacting with chlorophyll-degrading enzymes, and SGR1 in tomatoes promotes chlorophyll degradation. SAGs also play an important role in leaf senescence [2]. As shown in Figure 1E, NYC1 remained at a low level during the 8 h of H2O2 treatment and increased significantly at 24 h of storage, whereas H2O2 treatment induced significantly higher NYC1 expression at 24 h and 3 days of storage. The expression of PAO was also induced at 24 h of storage, but no significant changes were observed between H2O- and H2O2-treated leaves. The PPH expression in Figure 1G indicates that PPH increased in expression, and H2O2 treatment induced significantly higher expression at 8 h and 3 days. Similarly, increasing trends in the expression of SGR1, SAG12 and SAG15 were also observed during leaf storage. H2O2 could induce higher expression in PPH at 8 h, 24 h and 3 days; in SGR1 and SAG12 at 24 h and 3 days; and in SAG15 at 24 h. In sum, the above data suggest that H2O2 could act as a signal to elicit leaf senescence during storage by accelerating chlorophyll degradation and senescence-related gene expressions.
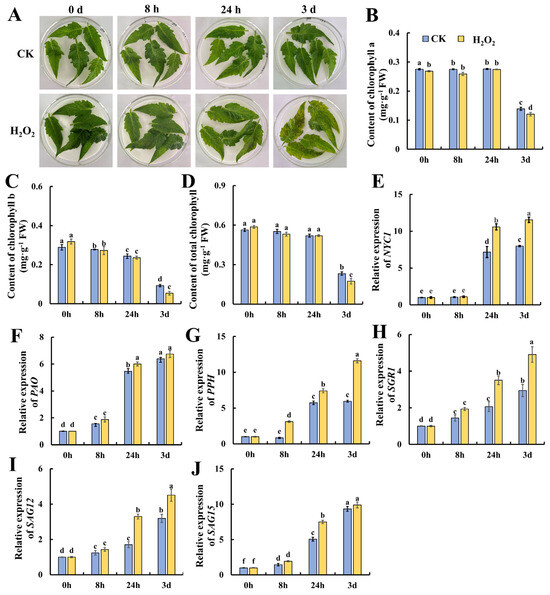
Figure 1.
Accelerated senescence of tomato leaves by hydrogen peroxide treatment leads to reduced chlorophyll levels and the up-regulation of the expression of genes related to chlorophyll degradation. (A) Phenotypic changes in tomato leaves at 0 h, 8 h, 24 h and 3 d after treatment. Changes in (B) chlorophyll a, (C) chlorophyll b and (D) chlorophyll content, and the gene expressions of (E) NYC1, (F) PAO, (G) PPH, (H) SGR1, (I) SAG12 and (J) SAG15 at different treatment times and in different treatment groups. Data are expressed as mean ± SD (n = 3), and significant differences at the level of p < 0.05 are indicated with different letters.
2.2. Effect of H2O2 Treatment on Reactive Oxygen Species in Tomato Leaves during Storage
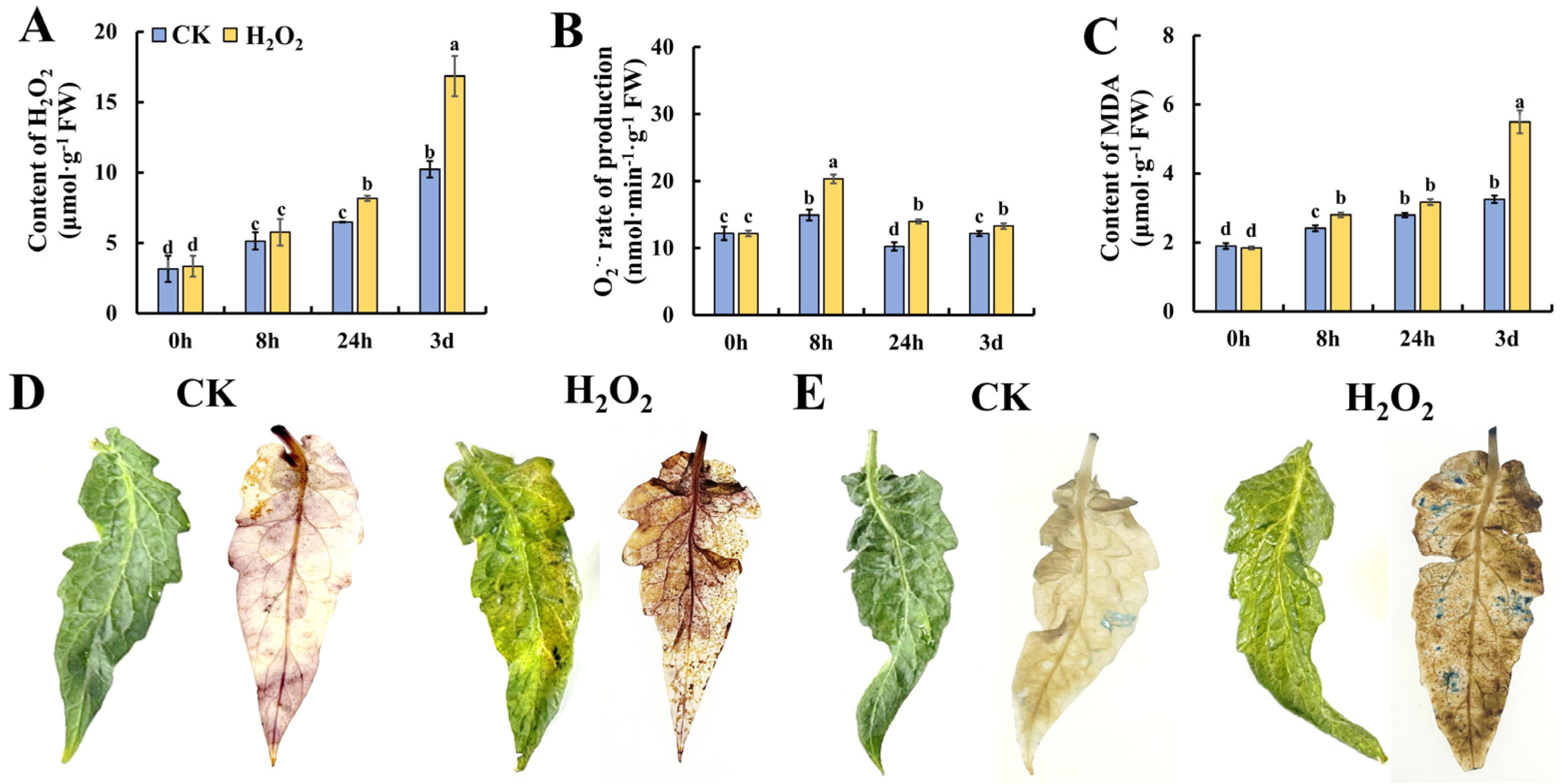
ROS have been demonstrated as critical initiators of plant senescence. Thus, we determined the content of ROS-related metabolites in tomato leaves during storage. As shown in Figure 2A, the content of H2O2 increased gradually during the storage of leaves, and the content in H2O2-treated tomato leaves was significantly higher than that in the control. Figure 2B shows the changes in O2•− in tomato leaves during storage, and H2O2 treatment was able to induce a significantly higher level of O2•− at 8 h and 24 h of storage. Similar to the change pattern of H2O2, the content of MDA increased during leaf storage and H2O2 treatment caused a significantly higher accumulation of MDA in tomato leaves during 3 days of storage. To obtain visible evidence of ROS metabolism in tomato leaves, Figure 2D shows the phenotype of tomato leaves on day 3 stained by DAB which indicate the content of H2O2. It was found that H2O2 treatment caused a deeper level of browning, suggesting that exogenous H2O2 treatment induced the accumulation of H2O2 in tomato leaves. Additionally, the dead cells in tomato leaves after storage for 3 days were visualized by trypan blue staining. And the image in Figure 2E indicates that more cells underwent cell necrosis during storage. These results suggest that H2O2 caused an imbalanced ROS metabolism in leaves, which may contribute to accelerated leaf senescence in tomato leaves during storage.
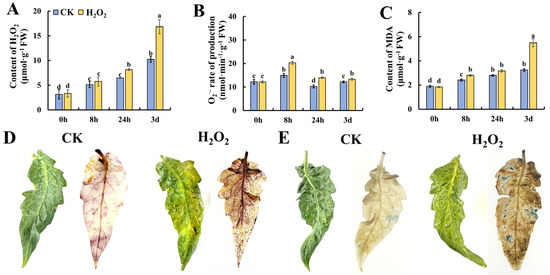
Figure 2.
Effect of H2O2 treatment on ROS homeostasis in tomato leaves. (A–C) Content of H2O2 (A), production rate of O2•− (B) and content of MDA (C) in tomato leaves. DAB (D) and trypan blue (E) staining of leaves on day 3 after H2O2 treatment. Data are expressed as mean ± SD (n = 3), and significant differences at the level of p < 0.05 are indicated with different letters.
2.3. Effect of H2O2 Treatment on the Activities of Antioxidant Enzymes
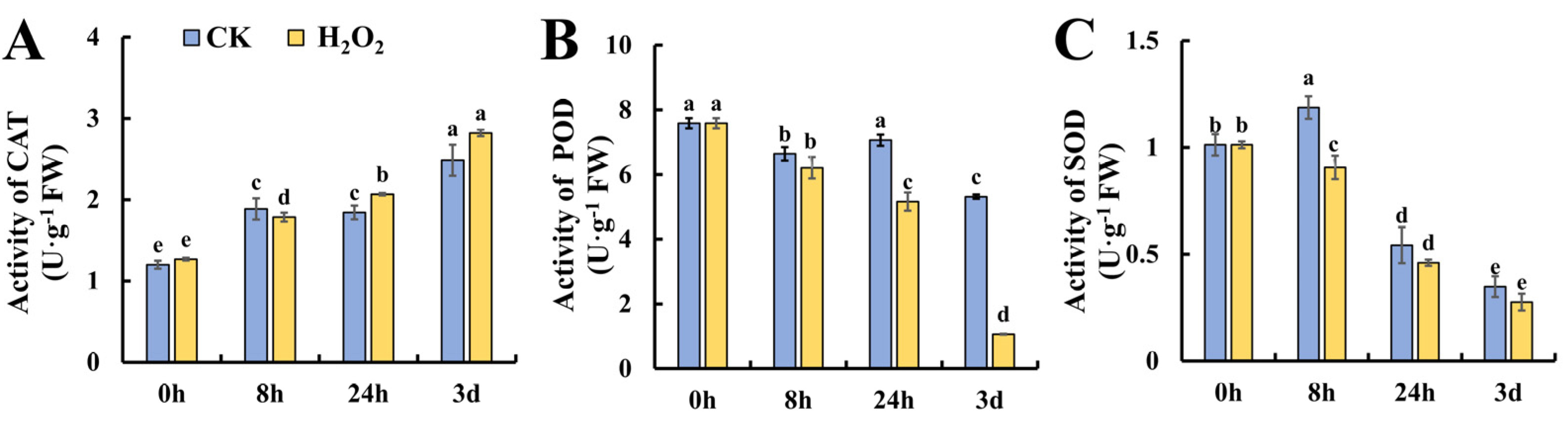
To further investigate the changes in ROS metabolism under H2O2 treatment, the enzymes responsible for ROS metabolism were studied at the activity level. The first line of defense against ROS in plants is SOD, which converts O2•− to H2O2. In addition, POD and CAT are actively involved in the decomposition of H2O2 into H2O and O2•− [18]. As shown in Figure 3A, the activity of CAT increased gradually during leaf storage and no significant difference was found between the control and H2O2 treatment. Figure 3B shows the changes in POD activity during leaf senescence; it was found that POD exhibited a decreasing trend and H2O2 treatment caused significantly lower POD activity at 24 h and 3 days of storage. Figure 3C shows that the SOD activity increased slightly at 8 h in the control leaves, whereas the increase was attenuated in H2O2-treated leaves followed by a decrease in both the control and H2O2 treatment. Generally, H2O2 caused the decreased activity of POD and SOD during tomato leaf senescence, which may contribute to earlier senescence in H2O2-treated leaves.
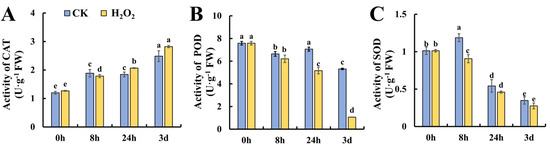
Figure 3.
Effect of H2O2 treatment on antioxidant enzyme activities in tomato leaves. (A–C) Activities of CAT (A), POD (B) and SOD (C) in tomato leaves under H2O2 and H2O treatment. Data are expressed as mean ± SD (n = 3), and significant differences at the level of p < 0.05 are indicated with different letters.
2.4. Effect of H2O2 on Hydrogen Sulfide Metabolism in Tomato Leaves
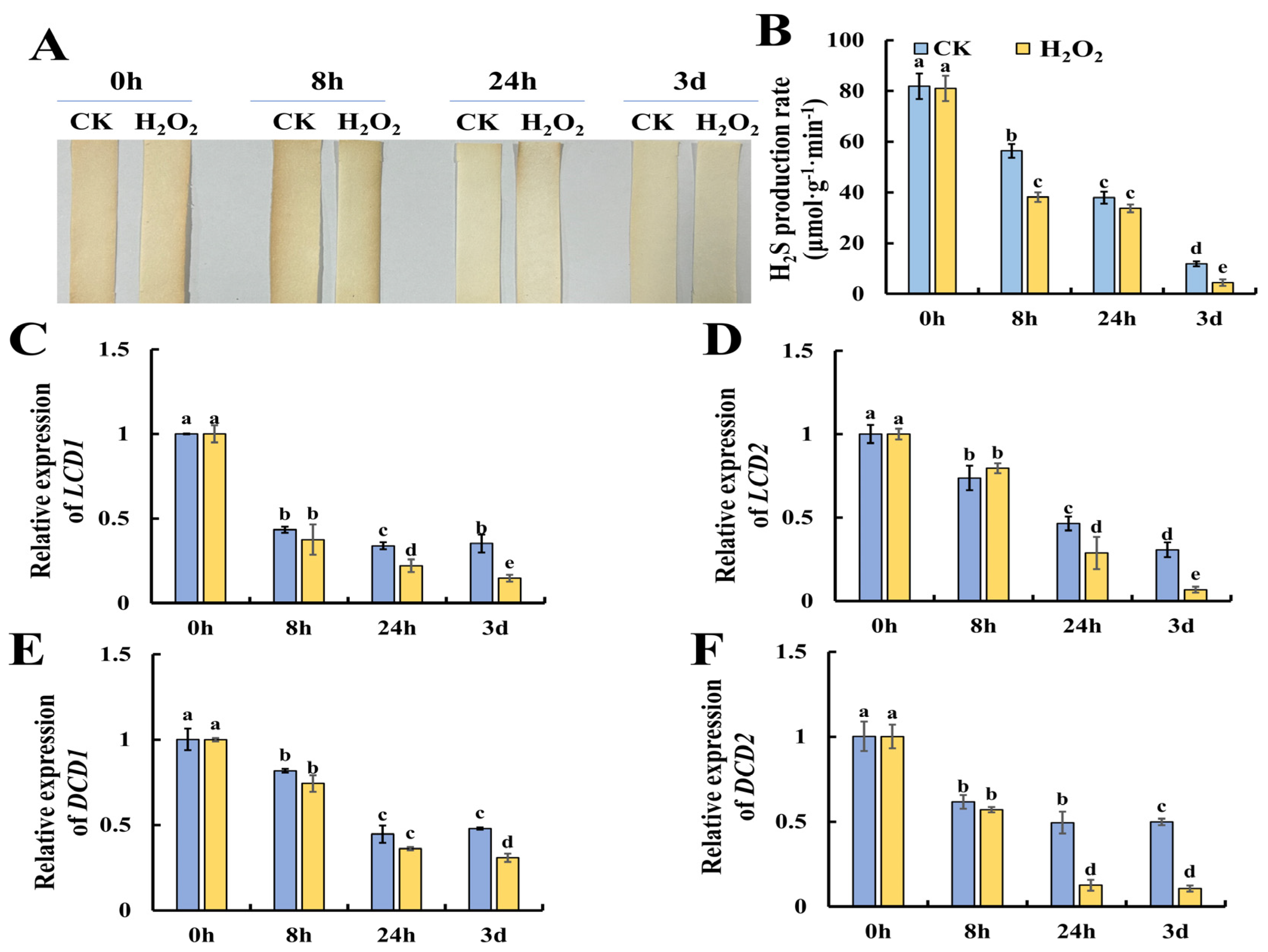
Previous research suggests that H2S could alleviate postharvest senescence in horticultural products by repressing the accumulation of ROS [16]. However, whether H2O2 could modulate H2S metabolism is still not clear. Thus, we evaluated the changes in the content of H2S in H2O2-treated tomato leaves during storage. Figure 4A shows the results of H2S production evaluated by lead acetate H2S detection strips, and the results showed that H2O2 treatment caused lower H2S contents especially at 8 h of H2O2 treatment. Furthermore, Figure 4B shows that H2O2 treatment resulted in a significantly lower rate of H2S production compared to the control. DCD is a key enzyme in the production of H2S using D-Cys as the substrate, while LCD catalyzes the decomposition of L-Cys to H2S, ammonia (NH4+) and pyruvate [19,20]. Then, we determined the expression of H2S-related metabolism genes, including LCD1, LCD2, DCD1 and DCD2. As shown in Figure 4C, the expression of LCD1 and LCD2 showed a decreasing trend during leaf senescence and H2O2 treatment caused significantly lower expression at 3 days of storage. Additionally, the expression of DCD1 and DCD2, which catalyze the production of H2S with D-cysteine as the substrate, were also determined. As shown in Figure 4E, F, a similar decreasing trend in the expression of DCD1 and DCD2 was observed, and H2O2 was found to reduce DCD1 expression at 3 days and DCD2 expression at 24 h and 3 days. Thus, we concluded that H2O2 could repress the generation of H2S by reducing the expression of LCD1/2 and DCD1/2.
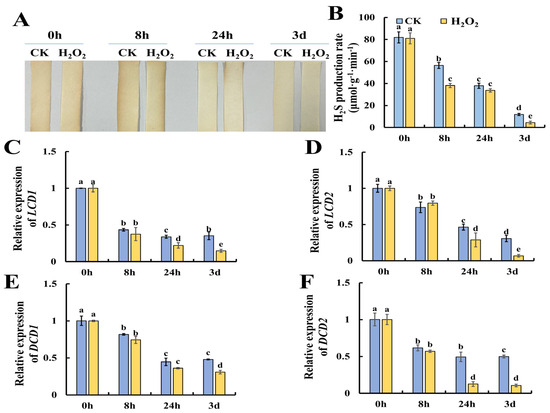
Figure 4.
Reduction in endogenous hydrogen sulfide production in tomato leaves by H2O2 treatment. (A,B) Tomato leaves with cysteine as substrates were assayed by lead acetate test paper and a hydrochloric acid absorption well method. (C–F) Expression patterns of LCD1 (C), LCD2 (D), DCD1 (E) and DCD2 (F) in tomato leaves under H2O2 and H2O treatment. Data are expressed as mean ± SD (n = 3), and significant differences at the level of p < 0.05 are indicated with different letters.
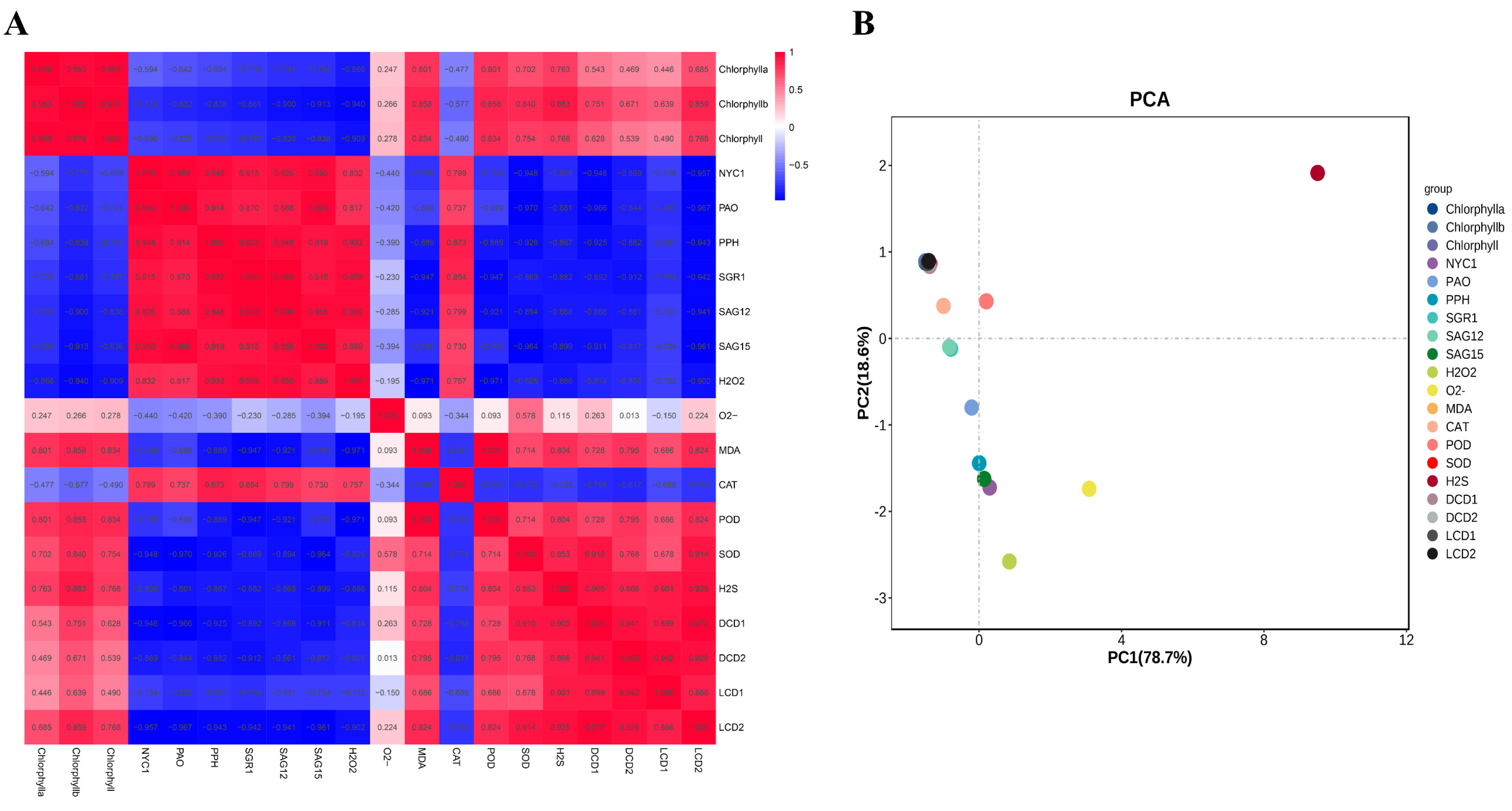
2.5. Correlation and Principal Component Analysis of Physiological Indexes and Senescence-Related Gene Expression
The correlation among the contents of H2O2 and MDA, H2S and the O2•− production rate; the contents of total chlorophyll, chlorophyll a and chlorophyll b; the gene expression of NYC1, PAO, PPH, SGR1, SAG12, SAG15, LCD1, LCD2, DCD1 and DCD2; and the activities of POD, SOD and CAT were analyzed. Figure 5A shows that the content of H2O2 showed higher negative correlations with total chlorophyll, chlorophyll a and chlorophyll b with correlation values of −0.909, −0.866 and −0.940, respectively. Additionally, total chlorophyll, chlorophyll a and chlorophyll b also showed negative correlations with the gene expression of NYC1, PAO, PPH, SGR1, SAG12 and SAG15, while they showed positive correlations with the gene expression of LCD1/2 and DCD1/2. H2S showed positive correlations with total chlorophyll, chlorophyll a, chlorophyll b, suggesting the role of H2S in alleviating chlorophyll degradation during leaf senescence. In all, the correlation data suggest that H2O2 may accelerate chlorophyll degradation, while H2S showed a protective role.
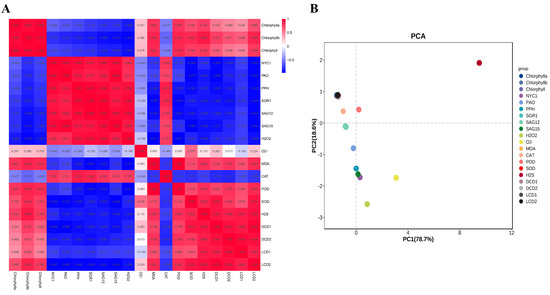
Figure 5.
(A) Correlation analysis among the parameters of chlorophyll a, chlorophyll b, chlorophyll, H2O2, MDA, the production of O2•− and H2S, and the gene expressions of NYC1, PAO, PPH, SGR1, SAG12, SAG15, DCD1, DCD2, LCD1 and LCD2 in tomato leaves at 0, 8, 24 h and 3 d after H2O2 and H2O treatments. The correlation indexes are as follows: “+” indicates a positive correlation, “−” indicates a negative correlation, 0.8–1 indicates a high correlation, 0.6–0.8 indicates a strong correlation, 0.4–0.6 indicates a moderate correlation and 0.2–0.4 indicates a moderate correlation. (B) PCA of the testing indicators in tomato leaves at 0, 8, 24 h and 3 d after H2O2 and H2O treatments. PC1 and PC2, respectively, represent the contribution rate of principal components.
Principal component analysis (PCA) was carried out based on the data of H2O2; MDA and H2S content; the O2•− production rate; total chlorophyll; chlorophyll a; chlorophyll b; the gene expression of NYC1, PAO, PPH, SGR1, SAG12, SAG15, LCD1, LCD2, DCD1 and DCD2; and the activity of POD, SOD and CAT. As shown in Figure 5B, PC1 and 2 contributed to 78.7% and 18.6% of the variability in the data, respectively. The data indicated that H2S showed the highest load value followed by O2•−, H2O2, DCD1, SAG15, etc. Therefore, it could be concluded that H2S and ROS metabolites contribute to the difference in leaf senescence caused by H2O2 treatment.
3. Discussion
During O2 metabolism in aerobic organisms, a variety of ROS are produced along with cell respiration, and the incomplete reduction of O2 leads to the generation of O2•−, hydroxyl radical (OH•) and H2O2. Excessive ROS are capable of inducing cellular damage by the oxidation of proteins, membranes and the mutation of DNA sequences [5,21]. However, appropriate concentrations of ROS could act as signaling molecules [22]. Among the ROS, H2O2 has been shown to regulate plant growth, development and stress resistance because it can react with the thiol (-SH) group on cysteine residues in proteins, which can regulate the function of target proteins [23].
H2O2 was increased during fruit senescence, suggesting that endogenous H2O2 showed a role in promoting fruit senescence [24]. To investigate whether exogenous H2O2 regulates tomato leaf senescence, we treated detached tomato leaves with 10 mmol/L of H2O2 for 8 h, and subjected them to storage for 3 days. The data indicated that H2O2 treatment caused significantly lower chlorophyll contents in contrast with the control, accompanied with the increased gene expression of chlorophyll degradation genes, including NYC1, PAO, PPH and SGR1, and senescence-related genes such as SAG12 and SAG15. The increased gene expression of chlorophyll degradation genes may contribute to lower chlorophyll contents in H2O2-treated leaves. SAG12 and SAG15 may function together to decompose proteins in leaves during senescence. Exogenous H2O2 also causes increases in H2O2, O2•− and MDA, which is a byproduct of lipid peroxidation, and thus the disturbed ROS homeostasis may lead to ROS stress and, finally, leaf senescence. Similarly, we previously reported that the depletion of DCD2, which encodes a D-cysteine desulfhydrase, causes decreased H2S release and increased H2O2 and MDA accumulation in fruits, which leads to an imbalance in ROS metabolism [24]. It has been shown that ROS production is closely related to plant senescence metabolism, and that excessive H2O2 accumulation is an important promoter of leaf senescence [25]. H2O2, due its role of deactivating microbes, has been applied to postharvest horticultural products. For instance, H2O2 treatment on harvest longan fruit causes a higher browning index and an increased rate of O2•− in the pericarp due to the reducing capacity of active oxygen scavenging [26]. Additionally, H2O2 treatment accelerated the accumulation of endogenous H2O2 by activating NADPH oxidase in bamboo shoots and up-regulated DNase, RNase and caspase 3-like activities, leading to the acceleration of the programmed cell death process [27]. It has also been shown that most senescence-related and ROS scavenging genes are up-regulated in the ls1 mutant, that LS1 may regulate leaf development and function, and that the disruption of LS1 function promotes ROS accumulation and accelerates leaf senescence and cell death in rice [28]. Similarly, we also observed increased cell death in tomato leaves treated with H2O2 (Figure 2E). The balance between SOD and different H2O2 scavenging enzymes such as POD and CAT in the cell is thought to be critical in determining the level of ROS homeostasis [29]. To further investigate the increase in ROS metabolism under H2O2 treatment, the enzymes responsible for ROS metabolism were studied and H2O2 was found to decrease the activity of POD and SOD during tomato leaf senescence, which may contribute to earlier senescence in H2O2-treated leaves. Therefore, H2O2 could accelerate leaf senescence by increasing endogenous ROS production and promoting chlorophyll degradation through up-regulating chlorophyll decomposing genes.
H2S could effectively delay fruit ripening and senescence by repressing ethylene synthesis and signaling pathways or by activating the activities of antioxidant enzymes such as CAT, SOD and POD, which may help to alleviate ROS stress during fruit ripening and senescence [16]. Additionally, compared with wild-type fruits, the mutation of D-cysteine desulfhydrase SlDCD2 induced H2O2 and MDA accumulation in fruits, which led to an imbalance in ROS metabolism. All the above evidence suggests that H2S could alleviate plant senescence by repressing ROS accumulation. However, there is no clue whether H2O2 interferes with the generation of H2S. We hypothesis that H2O2 may attenuate the production of H2S. To test this hypothesis, endogenous H2S production and the gene expression of H2S-releasing enzymes were analyzed. The data indicate that H2O2-treatment caused lower H2S contents, especially at 8 h of H2O2 treatment on detached tomato leaves. LCD (L-cysteine desulfhydrase, with L-Cys as the substrate) has also been shown to catalyze the degradation of cysteine to H2S, ammonia and pyruvate, and DCD uses D-cysteine as the substrate [30]. In addition, LCD1 mutation accelerated leaf senescence, whereas LCD1 overexpression significantly delayed leaf senescence, which may be due to endogenous H2S caused by LCD1 mutation [31]. The gene expression data indicate that H2O2 could repress the generation of H2S by reducing the expression of LCD1/2 and DCD1/2, which may contribute to decreased endogenous H2S in H2O2-treated leaves.
To study the relations between H2O2 and other parameters, the correlation among H2O2, MDA and H2S content; the O2•− production rate; total chlorophyll; chlorophyll a; chlorophyll b; the gene expression of NYC1, PAO, PPH, SGR1, SAG12, SAG15, LCD1, LCD2, DCD1 and DCD2; and the activity of POD, SOD and CAT were analyzed. The data revealed that H2O2 showed higher negative correlations with total chlorophyll, chlorophyll a and chlorophyll b. Additionally, chlorophyll also showed negative correlations with the gene expression of NYC1, PAO, PPH, SGR1, SAG12 and SAG15, whereas it showed positive correlations with the gene expression of LCD1, LCD2, DCD1 and DCD2. H2S showed positive correlations with total chlorophyll, chlorophyll a and chlorophyll b, suggesting the role of H2S in alleviating chlorophyll degradation during leaf senescence, but a negative correlation with H2O2. In all, the correlation data suggest that H2O2 may accelerate chlorophyll degradation, while H2S showed a protective role, and there is an antagonizing relation between H2O2 and H2S.
In summary, the role of H2O2 in regulating detached tomato leaf senescence was explored. Exogenous H2O2 application could promote chlorophyll degradation by increasing the expression of chlorophyll degradation-related genes. Additionally, H2O2 causes excessive ROS accumulation through repressing the activities of the antioxidant enzymes POD and SOD. By analyzing endogenous H2S content and the expression of LCDs/DCDs, H2S production was attenuated by H2O2 treatment, which may contribute to accelerated leaf senescence, suggesting the antagonizing relation between H2O2 and H2S. Thus, we provide solid evidence that H2O2 plays a positive role in detached leaf senescence, and H2O2 exhibits an antagonizing relation with H2S.
Controlling senescence can greatly improve crop yields and other plant characteristics, such as extending shelf life, especially in light of potential future food shortages and the use of plants as bioenergy sources [32]. Understanding how leaf senescence is regulated will help control senescence in the future, either through genetic modification or the manipulation of key environmental triggers [33]. In the present work, we found that H2O2 could act as a signal to promote leaf senescence by inhibiting H2S production. Thus, genetic manipulation on H2O2 metabolism-related genes such SOD, CAT and POD may provide a valuable tool to delay leaf senescence in crops or horticulture plants, thereby improving the yield.
4. Materials and Methods
4.1. Plant Material and Growing Conditions
The wild-type tomatoes (Solanum lycopersicum cv. “Micro-Tom”) used in this study were grown in a growth chamber (23 ± 2 °C; 50–70% relative humidity) under 16 h of light/8 h of darkness. Plants were grown to 6 weeks of age and tomato leaves in the same leaf position were removed for subsequent treatments.
4.2. Hydrogen Peroxide Treatment of Tomato Leaves
According to a previous study [34], we soaked tomato leaves in the same leaf position and growth period using 10 mmol/L of H2O2 for 8 h, and set the H2O treatment as a control. For the selection of the appropriate concentration of H2O2 and the treatment time, tomato leaves were treated with H2O2 at concentrations of 500 µmol/L, 1 mmol/L, 10 mmol/L and 100 mmol/L; the leaf phenotypes were recorded for 5 days; and thus the treatment time of 8 h and 10 mmol/L were selected as the treatment procedure that promoted leaf senescence obviously. Samples collected after 0 h, 8 h, 24 h and 3 d of treatment were used for mRNA extraction and qPCR.
4.3. RNA Extraction and RT–qPCR
RNA was extracted from a 0.5 g leaf and the first strand cDNA was synthesized following the method reported previously [31]. Tomato Tubulin was used as an internal reference.
4.4. Determination of the Levels of Chlorophyll in Tomato Leaves
The tomato fruit samples without seeds were uniformly ground into powder under freezing conditions, mixed with 5 mL of acetone and hexane (2:3), centrifuged at 4000 rpm for 5 min to collect the supernatant, and the absorbance values were determined by an enzyme meter at 663 nm, 645 nm, 505 nm and 453 nm. The contents of chlorophyll a, b and total chlorophyll were calculated by the formula described by Nagata et al. [35].
4.5. MDA Content
Based on the methodology reported by Li [36], 0.5 g of leaf samples was homogenized with 5% trichloroacetic acid, incubated and then centrifuged to collect the supernatant. The absorbance was measured at 450, 532 and 600 nm.
4.6. H2O2 Content
For the determination of the H2O2 content, 0.5 g of tomato leaves was taken, homogenized in 3 mL of pre-cooled acetone, centrifuged at 12,000 rpm for 30 min and the absorbance value at 508 nm was measured to determine the H2O2 content [37].
4.7. Production Rate of O2•−
As mentioned previously [38], the reaction buffer was composed of 50 mM phosphate buffer (pH 7.8) containing 17 mM sulfanilic acid, 1 mM hydroxylamine hydrochloride, 7 mM 1-naphthylamine and 50 µL sample solution. The absorbance of the mixture was measured at 530 nm, and the production rate of O2•− was calculated using previously described formulas.
4.8. Antioxidant Enzyme Assay
Tomato tissue (0.5 g) was extracted using 10 mL of 50 mM phosphate buffer (pH 7.8) at 4 °C. Then, samples were centrifuged at 10,000× g and 4 °C for 15 min. The supernatant was the crude antioxidant enzyme solution. CAT, SOD and POD activities were measured and calculated as described previously [39,40,41]. An increase in absorbance of 1.0 × 10−5 OD470 nm·min−1 was considered 1 U of POD activity, a decrease in absorbance of 1.0 × 10−3 at OD240 nm·min−1 was considered 1 U of CAT activity and the amount used to inhibit 5% of the photochemical reduction of NBT was considered 1 U of SOD activity. The results are expressed on an FW (Fresh Weight) basis as U·g−1.
4.9. H2S Production in Tomato Leaves
H2S production was determined using the lead sulfide method [42]. The leaf samples were ground to a powder in liquid nitrogen, homogenized in 5 mL of buffer (containing 100 mM potassium dihydrogen phosphate buffer pH 7.4, 10 mM Cys and 2 mM pyridoxine 5′-phosphate) and centrifuged to obtain a supernatant. Five sheets of lead acetate H2S test paper were placed in an airtight container above the liquid phase and incubated at 37 °C, protected from light, for several hours until the paper darkened. We soaked the test paper with 1 mL of 1 M HCl and it further reacted with N, Ndimethyl-phenylenediamine (DPD) with FeCl3, which was detected colorimetrically at 670 nm.
4.10. Statistical Analysis
The data were analyzed by SPSS statistics 25 software (SPSS version 25.0; Armonk, NY, USA), and the experimental data were plotted and analyzed by Excel software (Microsoft Office 2016). The correlation and the principal component analysis (PCA) of the data above were analyzed by the tools on the OmicShare (omicshare.com, accessed on 16 September 2023) platform.
Author Contributions
Conceptualization, K.H., Y.Y. and H.Z.; data curation, K.H. and Y.Y.; formal analysis, K.H., Y.Y., S.W. and W.G.; investigation, K.H., Y.Y., S.W. and G.Y.; validation, K.H., G.Y., D.Z. and W.L.; visualization, K.H.; writing original draft, K.H. and Y.Y.; writing review and editing, K.H., Y.Y., S.W., W.G., M.G., Y.S., D.Z., W.L. and H.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Natural Science Foundation of China (31970200, 31970312), the Fundamental Research Funds for the Central Universities (JZ2021HGPA0063), the Anhui Provincial Key Research and Development Plan (2022i01020001), and the Natural Science Foundations of Anhui Province (1908085MC72).
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interests.
References
- Guo, Y.; Gan, S. Leaf senescence: Signals, execution, and regulation. Curr. Top. Dev. Biol. 2005, 71, 83–112. [Google Scholar] [PubMed]
- Guo, Y.; Ren, G.; Zhang, K.; Li, Z.; Miao, Y.; Guo, H. Leaf senescence: Progression, regulation, and application. Mol. Hortic. 2021, 1, 1–25. [Google Scholar] [CrossRef]
- Álvarez, C.; García, I.; Moreno, I.; Pérez-Pérez, M.E.; Crespo, J.L.; Romero, L.C.; Gotor, C. Cysteine-generated sulfide in the cytosol negatively regulates autophagy and modulates the transcriptional profile in Arabidopsis. Plant Cell 2012, 24, 4621–4634. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-D.; Seo, P.J.; Yoon, H.-K.; Park, C.-M. The Arabidopsis NAC transcription factor VNI2 integrates abscisic acid signals into leaf senescence via the COR/RD genes. Plant Cell 2011, 23, 2155–2168. [Google Scholar] [CrossRef]
- Jajic, I.; Sarna, T.; Strzalka, K. Senescence, stress, and reactive oxygen species. Plants 2015, 4, 393–411. [Google Scholar] [CrossRef] [PubMed]
- Rogers, H.J. Is there an important role for reactive oxygen species and redox regulation during floral senescence? Plant Cell Environ. 2012, 35, 217–233. [Google Scholar] [CrossRef]
- Bienert, G.P.; Chaumont, F. Aquaporin-facilitated transmembrane diffusion of hydrogen peroxide. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2014, 1840, 1596–1604. [Google Scholar] [CrossRef]
- Bieker, S.; Riester, L.; Stahl, M.; Franzaring, J.; Zentgraf, U. Senescence-specific Alteration of Hydrogen Peroxide Levels in Arabidopsis thaliana and Oilseed Rape Spring Variety Brassica napus L. cv. Mozart F. J. Integr. Plant Biol. 2012, 54, 540–554. [Google Scholar] [CrossRef]
- Rogers, H.; Munné-Bosch, S. Production and scavenging of reactive oxygen species and redox signaling during leaf and flower senescence: Similar but different. Plant Physiol. 2016, 171, 1560–1568. [Google Scholar] [CrossRef]
- Domínguez, F.; Cejudo, F.J. Chloroplast dismantling in leaf senescence. J. Exp. Bot. 2021, 72, 5905–5918. [Google Scholar] [CrossRef]
- Balazadeh, S.; Kwasniewski, M.; Caldana, C.; Mehrnia, M.; Zanor, M.I.; Xue, G.-P.; Mueller-Roeber, B. ORS1, an H2O2-responsive NAC transcription factor, controls senescence in Arabidopsis thaliana. Mol. Plant 2011, 4, 346–360. [Google Scholar] [CrossRef]
- Fang, H.; Liu, Z.; Long, Y.; Liang, Y.; Jin, Z.; Zhang, L.; Liu, D.; Li, H.; Zhai, J.; Pei, Y. The Ca2+/calmodulin2-binding transcription factor TGA3 elevates LCD expression and H2S production to bolster Cr6+ tolerance in Arabidopsis. Plant J. 2017, 91, 1038–1050. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tang, J.; Liu, X.P.; Wang, Y.; Yu, W.; Peng, W.Y.; Fang, F.; Ma, D.F.; Wei, Z.J.; Hu, L.Y. Hydrogen sulfide promotes root organogenesis in Ipomoea batatas, Salix matsudana and Glycine max. J. Integr. Plant Biol. 2009, 51, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Liu, D.; Li, P.; Shen, W. Hydrogen sulfide delays leaf yellowing of stored water spinach (Ipomoea aquatica) during dark-induced senescence by delaying chlorophyll breakdown, maintaining energy status and increasing antioxidative capacity. Postharvest Biol. Technol. 2015, 108, 8–20. [Google Scholar] [CrossRef]
- Hu, L.-Y.; Hu, S.-L.; Wu, J.; Li, Y.-H.; Zheng, J.-L.; Wei, Z.-J.; Liu, J.; Wang, H.-L.; Liu, Y.-S.; Zhang, H. Hydrogen sulfide prolongs postharvest shelf life of strawberry and plays an antioxidative role in fruits. J. Agric. Food Chem. 2012, 60, 8684–8693. [Google Scholar] [CrossRef] [PubMed]
- Yao, G.-F.; Wei, Z.-Z.; Li, T.-T.; Tang, J.; Huang, Z.-Q.; Yang, F.; Li, Y.-H.; Han, Z.; Hu, F.; Hu, L.-Y. Modulation of enhanced antioxidant activity by hydrogen sulfide antagonization of ethylene in tomato fruit ripening. J. Agric. Food Chem. 2018, 66, 10380–10387. [Google Scholar] [CrossRef] [PubMed]
- Yao, G.-F.; Li, C.; Sun, K.-K.; Tang, J.; Huang, Z.-Q.; Yang, F.; Huang, G.-G.; Hu, L.-Y.; Jin, P.; Hu, K.-D. Hydrogen sulfide maintained the good appearance and nutrition in post-harvest tomato fruits by antagonizing the effect of ethylene. Front. Plant Sci. 2020, 11, 584. [Google Scholar] [CrossRef]
- Tan, X.-L.; Zhao, Y.-T.; Shan, W.; Kuang, J.-F.; Lu, W.-J.; Su, X.-G.; Tao, N.-G.; Lakshmanan, P.; Chen, J.-Y. Melatonin delays leaf senescence of postharvest Chinese flowering cabbage through ROS homeostasis. Food Res. Int. 2020, 138, 109790. [Google Scholar] [CrossRef]
- Schmidt, A. A cysteine desulfhydrase from spinach leaves specific for D-cysteine. Z. Pflanzenphysiol. 1982, 107, 301–312. [Google Scholar] [CrossRef]
- Romero, L.C.; Aroca, M.Á.; Laureano-Marín, A.M.; Moreno, I.; García, I.; Gotor, C. Cysteine and cysteine-related signaling pathways in Arabidopsis thaliana. Mol. Plant 2014, 7, 264–276. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van Breusegem, F. Reactive oxygen species signalling in plant stress responses. Nat. Rev. Mol. Cell Biol. 2022, 23, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.S.; Park, J.H.; Wi, S.D.; Kang, C.H.; Chi, Y.H.; Chae, H.B.; Paeng, S.K.; Ji, M.G.; Kim, W.-Y.; Kim, M.G. Redox-dependent structural switch and CBF activation confer freezing tolerance in plants. Nat. Plants 2021, 7, 914–922. [Google Scholar] [CrossRef]
- Zhao, Y.-Q.; Hu, K.-D.; Yao, G.-F.; Wang, S.-Y.; Peng, X.-J.; Zhang, H. A D-cysteine desulfhydrase, SlDCD2, participates in tomato fruit ripening by modulating ROS homoeostasis and ethylene biosynthesis. Hortic. Res. 2023, 10, uhad014. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Li, Z.; Huang, P.; Li, B.; Fang, S.; Chu, J.; Guo, H. A tripartite amplification loop involving the transcription factor WRKY75, salicylic acid, and reactive oxygen species accelerates leaf senescence. Plant Cell 2017, 29, 2854–2870. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lin, H.; Zhang, S.; Chen, Y.; Chen, M.; Lin, Y. The role of active oxygen metabolism in hydrogen peroxide-induced pericarp browning of harvested longan fruit. Postharvest Biol. Technol. 2014, 96, 42–48. [Google Scholar] [CrossRef]
- Li, D.; Limwachiranon, J.; Li, L.; Zhang, L.; Xu, Y.; Fu, M.; Luo, Z. Hydrogen peroxide accelerated the lignification process of bamboo shoots by activating the phenylpropanoid pathway and programmed cell death in postharvest storage. Postharvest Biol. Technol. 2019, 153, 79–86. [Google Scholar] [CrossRef]
- Zhang, C.; Li, N.; Hu, Z.; Liu, H.; Hu, Y.; Tan, Y.; Sun, Q.; Liu, X.; Xiao, L.; Wang, W. Mutation of leaf senescence 1 encoding a C2H2 zinc finger protein induces ROS accumulation and accelerates leaf senescence in rice. Int. J. Mol. Sci. 2022, 23, 14464. [Google Scholar] [CrossRef]
- Quan, L.J.; Zhang, B.; Shi, W.W.; Li, H.Y. Hydrogen peroxide in plants: A versatile molecule of the reactive oxygen species network. J. Integr. Plant Biol. 2008, 50, 2–18. [Google Scholar] [CrossRef]
- Papenbrock, J.; Riemenschneider, A.; Kamp, A.; Schulz-Vogt, H.; Schmidt, A. Characterization of cysteine-degrading and H2S-releasing enzymes of higher plants-from the field to the test tube and back. Plant Biol. 2007, 9, 582–588. [Google Scholar] [CrossRef]
- Hu, K.; Peng, X.; Yao, G.; Zhou, Z.; Yang, F.; Li, W.; Zhao, Y.; Li, Y.; Han, Z.; Chen, X. Roles of a cysteine desulfhydrase LCD1 in regulating leaf senescence in tomato. Int. J. Mol. Sci. 2021, 22, 13078. [Google Scholar] [CrossRef]
- Buchanan-Wollaston, V.; Earl, S.; Harrison, E.; Mathas, E.; Navabpour, S.; Page, T.; Pink, D. The molecular analysis of leaf senescence–a genomics approach. Plant Biotechnol. J. 2003, 1, 3–22. [Google Scholar] [CrossRef]
- Masood, J.; Zhu, W.; Fu, Y.; Li, Z.; Zhou, Y.; Zhang, D.; Han, H.; Yan, Y.; Wen, X.; Guo, H. Scaffold protein RACK1A positively regulates leaf senescence by coordinating the EIN3-miR164-ORE1 transcriptional cascade in Arabidopsis. J. Integr. Plant Biol. 2023. [Google Scholar] [CrossRef]
- Ortiz-Espín, A.; Locato, V.; Camejo, D.; Schiermeyer, A.; De Gara, L.; Sevilla, F.; Jiménez, A. Over-expression of Trxo1 increases the viability of tobacco BY-2 cells under H2O2 treatment. Ann. Bot. 2015, 116, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Nagata, M.; Yamashita, I. Simple method for simultaneous determination of chlorophyll and carotenoids in tomato fruit. Nippon. Shokuhin Kogyo Gakkaishi 1992, 39, 925–928. [Google Scholar] [CrossRef]
- Li, S.-P.; Hu, K.-D.; Hu, L.-Y.; Li, Y.-H.; Jiang, A.-M.; Xiao, F.; Han, Y.; Liu, Y.-S.; Zhang, H. Hydrogen sulfide alleviates postharvest senescence of broccoli by modulating antioxidant defense and senescence-related gene expression. J. Agric. Food Chem. 2014, 62, 1119–1129. [Google Scholar] [CrossRef]
- Bellincampi, D.; Dipierro, N.; Salvi, G.; Cervone, F.; De Lorenzo, G. Extracellular H2O2 induced by oligogalacturonides is not involved in the inhibition of the auxin-regulated rolB gene expression in tobacco leaf explants. Plant Physiol. 2000, 122, 1379–1386. [Google Scholar] [CrossRef]
- Yi, S.Y.; Lee, D.J.; Yeom, S.I.; Yoon, J.; Kim, Y.H.; Kwon, S.Y.; Choi, D. A novel pepper (Capsicum annuum) receptor-like kinase functions as a negative regulator of plant cell death via accumulation of superoxide anions. New Phytol. 2010, 185, 701–715. [Google Scholar] [CrossRef]
- Aebi, H. [13] Catalase in vitro. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1984; Volume 105, pp. 121–126. [Google Scholar]
- Im, Y.J.; Ji, M.; Lee, A.; Killens, R.; Grunden, A.M.; Boss, W.F. Expression of Pyrococcus furiosus superoxide reductase in Arabidopsis enhances heat tolerance. Plant Physiol. 2009, 151, 893–904. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Castillo, F.J.; Penel, C.; Greppin, H. Peroxidase release induced by ozone in Sedum album leaves: Involvement of Ca2+. Plant Physiol. 1984, 74, 846–851. [Google Scholar] [CrossRef]
- Hine, C.; Harputlugil, E.; Zhang, Y.; Ruckenstuhl, C.; Lee, B.C.; Brace, L.; Longchamp, A.; Treviño-Villarreal, J.H.; Mejia, P.; Ozaki, C.K. Endogenous hydrogen sulfide production is essential for dietary restriction benefits. Cell 2015, 160, 132–144. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).