Abstract
Pregnancy induces significant changes in oral health because of hormonal fluctuations, making it a crucial period for preventive measures. Dental stem cells (DSCs), particularly those derived from the dental pulp and periodontal ligaments, offer promising avenues for regenerative therapies and, possibly, preventive interventions. While the use of DSCs already includes various applications in regenerative dentistry in the general population, their use during pregnancy requires careful consideration. This review explores recent advancements, challenges, and prospects in using DSCs to address oral health issues, possibly during pregnancy. Critical aspects of the responsible use of DSCs in pregnant women are discussed, including safety, ethical issues, regulatory frameworks, and the need for interdisciplinary collaborations. We aimed to provide a comprehensive understanding of leveraging DSCs to improve maternal oral health.
1. Introduction
In recent years, attention has been focused on oral health during pregnancy [1,2]. Increasing evidence indicates that oral health plays a crucial role in maternal well-being and can affect the health of the developing fetus. Several authors have provided a relevant contribution to the significance of oral health maintenance during pregnancy, offering insights into preventive measures, management strategies, and collaborative efforts necessary among healthcare providers to ensure comprehensive care for expectant mothers [3,4,5]. Pregnancy is a unique stage of a woman’s life, marked by profound physiological changes, including fluctuations in hormone levels, which in turn exert significant effects on oral health [4,6]. These hormonal shifts, particularly elevated levels of estrogen, progesterone, and human chorionic gonadotropin (hCG), establish a setting favorable to oral health changes that, in turn, may favor the occurrence of gingivitis and periodontitis. Indeed, gingivitis, characterized by inflammation of the gums, is highly prevalent during pregnancy, affecting up to 60–75% of pregnant women [3,7]. Moreover, periodontitis, a more severe form of gum disease involving progressive destruction of periodontal tissues, is exacerbated during gestation [3,7,8]. Table 1 provides an overview of oral health indicators and their correlations with pregnancy-related complications. Each indicator is delineated alongside a description of its characteristics and factors contributing to its occurrence. Moreover, the potential risks of pregnancy-related complications associated with each indicator are indicated.

Table 1.
Correlation between oral health indicators and pregnancy-related complications.
The oral microbiota includes a dynamic and complex ecosystem, including bacteria, fungi, and viruses. Different authors delve into the specific microbial taxa that populate the oral cavity and their potential impact on maternal health during pregnancy [6,7,10,18]. These authors underline how hormonal fluctuations, immune system changes, and alterations in oral hygiene practices can influence the composition and diversity of oral microbiota, potentially leading to dysbiosis. The latter, characterized by disruptions in the balance of microbial communities and heightened pathogenic activity, has a significant impact on pregnancy outcomes. Notably, specific microbial species such as Porphyromonas gingivalis and Fusobacterium nucleatum, widely recognized for their involvement in periodontal disease, have emerged as relevant players during pregnancy. Indeed, they have been linked to adverse outcomes, particularly preterm birth and low birth weight [19]. Carrouel et al., specifically focused on the oral microbiota of pregnant women [20]. In particular, the presence and relative abundance of periodontal pathogens was assessed within the interdental microbiota of pregnant individuals with an intact periodontium, at 3 months of gestation. Through molecular and culture-based assays, several microbial species, including P. gingivalis, Tannerella forsythia, and Treponema denticola, among others, were detected. Given the well-known role of these pathogens in periodontal diseases, the authors highlight the potential implications of the presence of these microbial communities on maternal oral health during pregnancy and the potential risk of subsequent periodontal diseases.
Recently, the association between oral microbiota and hypertensive disorders during pregnancy, particularly preeclampsia, has become a focal point of investigation [21]. Preeclampsia, characterized by high blood pressure and organ dysfunction during pregnancy, represents a major health concern. The significance of maternal oral health extends beyond this scope, as emerging evidence suggests a potential link between periodontal diseases and several adverse pregnancy outcomes, including preterm birth and low birth weight [8,10,19,22]. This bidirectional relationship underscores the importance of proactive oral care interventions during pregnancy to safeguard maternal and fetal well-being. Maintaining optimal oral health during pregnancy is crucial for both maternal and fetal well-being. Nevertheless, there is considerable variability in the awareness and adherence to recommended oral hygiene practices among expectant mothers. In their study, Bushehab et al., offered valuable insights into the impact of oral hygiene practices and awareness levels of pregnant women [3]. Employing methodologies such as surveys, interviews, or questionnaires, these authors gathered data to assess the knowledge, attitudes, and behaviors of pregnant women concerning oral health during pregnancy and its potential implications for pregnancy outcomes.
Given the evolving landscape of oral health and the dynamic physiological shifts that occur during pregnancy, there is growing interest in investigating minimally invasive approaches to manage various oral clinical conditions in pregnant women, with a focus on ensuring their comfort and well-being.
With an understanding of the pivotal roles of stem cells in both medicine and dentistry, this concise review considers the potential use of dental stem cells (DSCs) throughout pregnancy across diverse clinical contexts. The overarching goal is to mitigate concerns regarding fear, pain, trauma, and fetal risks associated with interventions. Despite the inherent challenges stemming from limited research in this domain, this initiative is poised to assist research entities in refining strategies for future employment of DSCs to enhance oral health outcomes in expectant mothers.
2. DSCs and Pregnancy
2.1. A Brief History of Stem Cells
A synthetic journey through history provides invaluable insights into the significance of DSCs, beginning with the discovery of stem cells themselves. Stem cells have long captivated scientists due to their extraordinary regenerative potential, with roots tracing back to Ernst Haeckel’s proposal of “Stammzellen” in 1868 [23]. The late 20th century witnessed pivotal moments in stem cell research, including Alexander Maksimov’s coining of the term “stem cell” in 1908 and the groundbreaking isolation of embryonic stem cells (ESCs) from mouse embryos in 1981 [24]. A monumental leap occurred in 1998 when James Thomson and his team successfully isolated human ESCs, marking a significant milestone [25]. Subsequently, in 2006, Shinya Yamanaka’s discovery of induced pluripotent stem cells (iPSCs) revolutionized the field by providing a method to reprogram adult cells into a pluripotent state, offering an ethically acceptable alternative to ESCs [26]. This breakthrough offered a priceless alternative to ESCs by circumventing the need for human embryos. Stem cell research expanded to encompass various types of adult stem cells, including mesenchymal stem cells (MSCs) found in bone marrow and adipose tissue, among other sources [27]. Next, DSCs emerged as promising candidates, when sources such as dental pulp, periodontal ligaments, and dental follicles provided an easily accessible reservoir of cells for future applications in regenerative therapies [28,29,30,31]. Through the exploration of stem cell origins and historical milestones, researchers have paved the way for advancements in regenerative medicine, offering potential solutions for treating a myriad of diseases and injuries.
2.2. What Do We Know about DSCs?
DSCs represent a valuable asset with vast therapeutic potential, not only within dentistry but also extending to a broader range of medical applications [28,30,31]. Undoubtedly, to fully harness their benefits, further research efforts are imperative to unravel their intricate mechanisms of action, refine isolation and culture methodologies, and bridge the gap between preclinical discoveries and clinical implementation. Delving into the complexity of DSCs unveils their unique characteristics, various sources, intrinsic properties, and promising applications. For researchers, these aspects represent novel pathways that can be utilized in both regenerative medicine and oral healthcare.
Table 2 provides a concise overview of various types of dental stem cells, including their sources, functions, and potential applications in regenerative medicine and oral healthcare. DSCs, derived from different tissues such as dental pulp, periodontal ligaments, and oral mucosa, exhibit unique properties such as self-renewal and multilineage differentiation potential. These cells hold significant promise for tissue regeneration and repair in dentistry, offering potential treatments for a range of oral and systemic diseases, including periodontal disease, dental caries, and craniofacial defects.

Table 2.
Comprehensive overview of the different DSCs, listed according to their characteristics, sources, functions, and potential applications.
2.3. Isolation of DSCs: Standardization of Protocols
The standardization of experimental protocols aimed at obtaining DSCs is essential for significant advancement in basic and clinical research. Table 3 provides a summary of the published protocols for the in vitro isolation of various types of stem cells from different anatomical niches in the oral cavity. For each stem cell type, step-by-step procedures are described, including tissue collection, dissociation, cell isolation, and culture [48,49,50,51,52,53]. Strict adherence to these standardized isolation protocols is essential and will undoubtedly enhance the reproducibility and scalability of stem cell-based therapies and research efforts in regenerative dentistry and beyond.

Table 3.
Protocols available in the literature for stem cell isolation from the oral cavity.
3. Clinical Implications and Applications of DSCs in Oral Health
Minimizing oral disease-related complications during pregnancy remains a widely recognized key point to be pursued. Monitoring oral health indicators during prenatal care allows for early intervention, potentially mitigating the risk of pregnancy-related complications [56]. Educating pregnant women about the importance of oral hygiene practices and regular dental check-ups is crucial in preventing potential complications [57]. Implementing comprehensive oral health education programs can empower women to adopt preventive measures and enhance their overall well-being during pregnancy [58]. In this scenario, the clinical applications of DSCs during pregnancy represent a novel and highly challenging tool to work with. The following are some of the most likely possibilities for DSCs’ use in pregnant women:
- Prevention and Treatment of Dental Caries: DSCs can be employed for the development of novel strategies for the remineralization of dental enamel and dentin, offering a non-invasive approach to prevent and treat dental caries in pregnant women [59].
- Pulp Regeneration: Pulpal diseases, including pulpitis and pulp necrosis, can pose significant challenges during pregnancy due to limited treatment options that are safe for both the mother and fetus [34]. DSC-mediated pulp regeneration involves the transplantation of DSCs, such as stem cells from the apical papilla (SCAPs) or DPSCs, into the pulp chamber to promote pulp tissue regeneration and repair [30,31,32]. This approach holds promise for preserving the vitality of compromised teeth and avoiding invasive procedures during pregnancy.
- Periodontal Disease Management: Preclinical studies have demonstrated the efficacy of DSC-based therapies in promoting periodontal tissue regeneration and reducing inflammation, offering a potential treatment modality for pregnant women with periodontal disease [37,52,56,60].
- Salivary Gland Regeneration: DSC-based approaches for salivary gland regeneration offer a potential solution for restoring salivary gland function and alleviating xerostomia in pregnant women [61,62].
- Oral-Origin Organoid Transplantation: Recent advancements in stem cell research have led to the development of oral-origin organoids, including tooth germ organoids, salivary gland organoids, taste bud organoids, and lingual epithelial organoids [40,42]. These organoids hold promise for regenerative therapies in maternal oral health enhancement. Transplantation of organoids derived from oral tissues may offer innovative approaches for repairing damaged oral structures and restoring oral function during pregnancy.
- Neural Regeneration: Pregnancy-related neuropathies and nerve injuries in the oral and maxillofacial region can lead to significant discomfort and functional impairment. DSCs have shown promise in promoting neural regeneration and may offer novel therapeutic avenues for managing pregnancy-related neuropathic pain and sensory disturbances [35,45,63].
These clinical implications underscore the potential of DSCs as versatile tools for enhancing maternal oral health during pregnancy. Further research and clinical studies are needed to validate the safety and efficacy of DSC-based therapies in pregnant women.
4. Limitations for DSC Use in Pregnancy
Currently, no studies specifically address the use of DSCs to counteract oral lesions during pregnancy. The most likely explanations might be the potential risks and ethical considerations associated with such interventions, including the ones indicated below.
Safety Concerns: Pregnancy is a sensitive period during which any intervention, including the use of stem cells, may pose risks to the mother and developing fetus. Stem cell therapies carry inherent risks such as immune rejection, infection, and tumorigenicity [56,64]. Additionally, the effects of stem cell therapy on fetal development are not well understood, raising concerns about potential adverse outcomes.
Ethical Considerations: Conducting research involving pregnant women raises complex ethical issues, including the need to ensure the safety and well-being of both the mother and fetus [65]. Researchers must adhere to strict ethical guidelines and obtain informed consent from participants, considering the potential risks and benefits of the intervention.
Regulatory Hurdles: Stem cell therapies, particularly those involving novel applications, such as during pregnancy, may face regulatory challenges and require extensive preclinical and clinical testing to ensure safety and efficacy [66]. The lack of established protocols and regulatory frameworks for using stem cells during pregnancy may hinder research in this area [67].
Limited Evidence: Despite the potential therapeutic benefits of stem cell therapy, including DSC treatment of oral lesions, there is still limited evidence of their safety and efficacy, specifically in pregnant women [68]. The absence of robust clinical data may deter researchers and clinicians from further exploring this application.
Table 4 shows the critical points currently affecting DSCs’ employment in pregnancy. Overcoming such limitations through further research, interdisciplinary collaboration, long-term monitoring, and adherence to ethical and regulatory standards will enhance the credibility and applicability of leveraging DSCs in pregnancy. Overall, leveraging DSCs during pregnancy to enhance oral health remains an objective to be pursued.

Table 4.
Parameters that may influence the research development in the DSC field.
5. Future Trajectories of DSCs during Pregnancy
Extensive research should focus on the therapeutic potential of DSCs in pregnancy-associated oral diseases, particularly elucidating the biomolecular mechanisms responsible for the beneficial effects. Furthermore, efforts should be directed toward optimizing the delivery methods and dosing regimens of DSC-based therapies to maximize their therapeutic efficacy. Scheme 1 outlines key issues that may deserve special attention and should ideally become priority topics for assessment by both basic and clinical researchers. The ability of DSCs to proliferate and differentiate during pregnancy (even at different stages of fetal development), their capacity to migrate and regulate immune responses in response to hormonal changes, and the potential alterations in their interaction with the oral cavity microbiota influenced by pregnancy are all crucial aspects that need clarification. A deep understanding of these issues will promote the clinical use of DSCs during pregnancy, especially regarding their role in maternal oral healing, tissue regeneration, and fetal wellness.
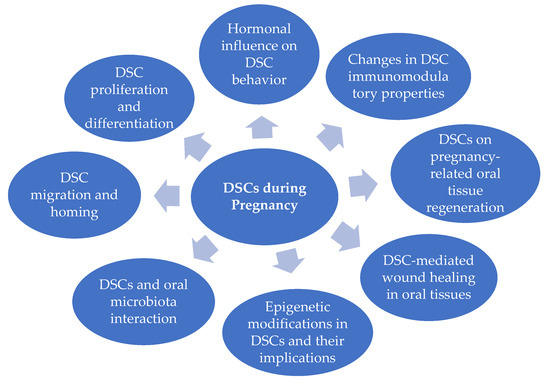
Scheme 1.
Potential impact of pregnancy on DSCs. Different lines of research will have to be developed to expand the knowledge on each specific issue.
6. Conclusions
DSCs stand as a promising source of multipotent cells with substantial potential for advancing regenerative dentistry and oral health enhancement. These cells can be isolated from various oral tissues, primarily including the dental pulp, periodontal ligament, and dental follicles. DSCs possess notable versatility, demonstrating the ability to differentiate into multiple cell lineages, such as osteoblasts, odontoblasts, adipocytes, and neural cells. This adaptability makes DSCs attractive candidates for diverse clinical applications, ranging from tissue engineering to interventions addressing pregnancy-related oral health challenges. Despite significant progress in DSC research, numerous challenges persist. These encompass the need for standardization in isolation protocols, the enhancement of cell proliferation and differentiation, the optimization of scaffold design, rigorous evaluation of safety and efficacy, and the attainment of regulatory approval for clinical implementation. Overcoming these obstacles is crucial to fully harness the therapeutic potential of DSC-based therapies and propel the field of regenerative dentistry forward. In this pursuit, establishing interdisciplinary collaborations among healthcare practitioners is pivotal. Such collaborative efforts will drive further exploration in basic and preclinical research, facilitating the seamless integration of DSC therapies into specialized clinical contexts, including those relevant to pregnancy and prenatal care.
Author Contributions
Conceptualization, A.M. (Aida Meto), A.S. and E.B.; writing—original draft preparation, A.M. (Aida Meto), S.P. and A.M. (Agron Meto); writing—review and editing, A.S., S.P., A.M. (Agron Meto) and E.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hartnett, E.; Haber, J.; Krainovich-Miller, B.; Bella, A.; Vasilyeva, A.; Lange Kessler, J. Oral Health in Pregnancy. J. Obstet. Gynecol. Neonatal Nurs. 2016, 45, 565–573. [Google Scholar] [CrossRef]
- Pecci-Lloret, M.P.; Linares-Pérez, C.; Pecci-Lloret, M.R.; Rodríguez-Lozano, F.J.; Oñate-Sánchez, R.E. Oral Manifestations in Pregnant Women: A Systematic Review. J. Clin. Med. 2024, 13, 707. [Google Scholar] [CrossRef]
- Bushehab, N.M.E.; Sreedharan, J.; Reddy, S.; D’souza, J.; Abdelmagyd, H. Oral Hygiene Practices and Awareness of Pregnant Women about the Effects of Periodontal Disease on Pregnancy Outcomes. Int. J. Dent. 2022, 2022, 5195278. [Google Scholar] [CrossRef]
- Chawłowska, E.; Karasiewicz, M.; Lipiak, A.; Staszewski, R.; Cofta, M.; Biskupska, M.; Giernaś, B.; Zawiejska, A. Oral Health Behaviours, Knowledge, and Literacy of Expectant Mothers: A Cross-Sectional Study among Maternity Ward Patients. Int. J. Environ. Res. Public Health 2022, 19, 11762. [Google Scholar] [CrossRef] [PubMed]
- Przeklasa-Bierowiec, A.; Jakubik, A.; Szczeklik, K.; Majewska, I.; Marcinek, A.; Pytko-Polończyk, J. Awareness of oral health prophylaxis in pregnant women. Folia Med. Crac. 2020, 60, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Han, Y.W. Oral bacteria, oral health, and adverse pregnancy outcomes. Periodontol 2000 2022, 89, 181–189. [Google Scholar] [CrossRef]
- Balan, P.; Chong, Y.S.; Umashankar, S.; Swarup, S.; Loke, W.M.; Lopez, V.; He, H.G.; Seneviratne, C.J. Keystone Species in Pregnancy Gingivitis: A Snapshot of Oral Microbiome during Pregnancy and Postpartum Period. Front. Microbiol. 2018, 9, 2360. [Google Scholar] [CrossRef]
- Boggess, K.A. Choosing the left fork: Steven Offenbacher and understanding maternal periodontal disease and adverse pregnancy outcomes. J. Periodontol. 2020, 91, S40–S44. [Google Scholar] [CrossRef]
- Offenbacher, S.; Lieff, S.; Boggess, K.A.; Murtha, A.P.; Madianos, P.N.; Champagne, C.M.E.; McKaig, R.G.; Jared, H.L.; Mauriello, S.M.; Auten, R.L., Jr.; et al. Maternal periodontitis and prematurity. Part I: Obstetric outcome of prematurity and growth restriction. Ann. Periodontol. 2001, 6, 164–174. [Google Scholar] [CrossRef]
- Vidmar Šimic, M.; Maver, A.; Zimani, A.N.; Hočevar, K.; Peterlin, B.; Kovanda, A.; Premru-Sršen, T. Oral microbiome and preterm birth. Front. Med. 2023, 10, 1177990. [Google Scholar] [CrossRef]
- Cho, G.J.; Kim, S.Y.; Lee, H.C.; Kim, H.Y.; Lee, K.M.; Han, S.W.; Oh, M.J. Association between dental caries and adverse pregnancy outcomes. Sci. Rep. 2020, 10, 5309. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Fogarty, C.; Wu, T.T.; Alkhers, N.; Zeng, Y.; Thomas, M.; Youssef, M.; Wang, L.; Cowen, L.; Abdelsalam, H.; et al. Oral health and Candida carriage in socioeconomically disadvantaged US pregnant women. BMC Pregnancy Childbirth 2019, 19, 480. [Google Scholar] [CrossRef]
- Duarte da Silva, K.; Vargas-Ferreira, F.; Dâmaso Bertoldi, A.; Celso Lopes Fernandes de Barros, F.; Fernando Demarco, F.; Britto Correa, M.; Beatriz Chaves Tarquinio, S. Oral mucosal lesions in pregnant women: A population-based study. Oral Dis. 2022, 28, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Bett, J.V.S.; Batistella, E.Â.; Melo, G.; Munhoz, E.A.; Silva, C.A.B.; Guerra, E.N.D.S.; Porporatti, A.L.; De Luca Canto, G. Prevalence of oral mucosal disorders during pregnancy: A systematic review and meta-analysis. J. Oral Pathol. Med. 2019, 48, 270–277. [Google Scholar] [CrossRef]
- Cardoso, J.A.; Spanemberg, J.C.; Cherubini, K.; Figueiredo, M.A.; Salum, F.G. Oral granuloma gravidarum: A retrospective study of 41 cases in Southern Brazil. J. Appl. Oral Sci. 2013, 21, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Lomeli Martinez, S.M.; Carrillo Contreras, N.G.; Gómez Sandoval, J.R.; Zepeda Nuño, J.S.; Gomez Mireles, J.C.; Varela Hernández, J.J.; Mercado-González, A.E.; Bayardo González, R.A.; Gutiérrez-Maldonado, A.F. Oral Pyogenic Granuloma: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 16885. [Google Scholar] [CrossRef]
- Crusell, M.K.W.; Brink, L.R.; Nielsen, T.; Allin, K.H.; Hansen, T.; Damm, P.; Lauenborg, J.; Hansen, T.H.; Pedersen, O. Gestational diabetes and the human salivary microbiota: A longitudinal study during pregnancy and postpartum. BMC Pregnancy Childbirth 2020, 20, 69. [Google Scholar] [CrossRef] [PubMed]
- Saadaoui, M.; Singh, P.; Al Khodor, S. Oral microbiome and pregnancy: A bidirectional relationship. J. Reprod. Immunol. 2021, 145, 103293. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.W.; Redline, R.W.; Li, M.; Yin, L.; Hill, G.B.; McCormick, T.S. Fusobacterium nucleatum induces premature and term stillbirths in pregnant mice: Implication of oral bacteria in preterm birth. Infect. Immun. 2004, 72, 2272–2279. [Google Scholar] [CrossRef]
- Carrouel, F.; Kanoute, A.; Lvovschi, V.E.; Bourgeois, D. Periodontal pathogens of the interdental microbiota in a 3 months pregnant population with an intact periodontium. Front. Microbiol. 2023, 14, 1275180. [Google Scholar] [CrossRef]
- Beckers, K.F.; Sones, J.L. Maternal microbiome and the hypertensive disorder of pregnancy, preeclampsia. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H1–H10. [Google Scholar] [CrossRef]
- Ye, C.; Kapila, Y. Oral microbiome shifts during pregnancy and adverse pregnancy outcomes: Hormonal and Immunologic changes at play. Periodontol 2000 2021, 87, 276–281. [Google Scholar] [CrossRef]
- Armstrong, L.; Lako, M.; Buckley, N.; Lappin, T.R.; Murphy, M.J.; Nolta, J.A.; Pittenger, M.; Stojkovic, M. Editorial: Our top 10 developments in stem cell biology over the last 30 years. Stem Cells 2012, 30, 2–9. [Google Scholar] [CrossRef]
- Evans, M.J.; Kaufman, M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature 1981, 292, 154–156. [Google Scholar] [CrossRef]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Pittenger, M.F.; Mackay, A.M.; Beck, S.C.; Jaiswal, R.K.; Douglas, R.; Mosca, J.D.; Moorman, M.A.; Simonetti, D.W.; Craig, S.; Marshak, D.R. Multilineage potential of adult human mesenchymal stem cells. Science 1999, 284, 143–147. [Google Scholar] [CrossRef]
- Tatullo, M.; Marrelli, M.; Shakesheff, K.M.; White, L.J. Dental pulp stem cells: Function, isolation and applications in regenerative medicine. J. Tissue Eng. Regen. Med. 2015, 9, 1205–1216. [Google Scholar] [CrossRef]
- Sun, L.; Du, X.; Kuang, H.; Sun, H.; Luo, W.; Yang, C. Stem cell-based therapy in periodontal regeneration: A systematic review and meta-analysis of clinical studies. BMC Oral Health 2023, 23, 492. [Google Scholar] [CrossRef]
- Zhang, W.; Yelick, P.C. Tooth Repair and Regeneration: Potential of Dental Stem Cells. Trends Mol. Med. 2021, 27, 501–511. [Google Scholar] [CrossRef]
- Botelho, J.; Cavacas, M.A.; Machado, V.; Mendes, J.J. Dental stem cells: Recent progresses in tissue engineering and regenerative medicine. Ann. Med. 2017, 49, 644–651. [Google Scholar] [CrossRef]
- Nagata, M.; Ono, N.; Ono, W. Unveiling diversity of stem cells in dental pulp and apical papilla using mouse genetic models: A literature review. Cell Tissue Res. 2021, 383, 603–616. [Google Scholar] [CrossRef]
- Shi, X.; Mao, J.; Liu, Y. Pulp stem cells derived from human permanent and deciduous teeth: Biological characteristics and therapeutic applications. Stem Cells Transl. Med. 2020, 9, 445–464. [Google Scholar] [CrossRef]
- Nakashima, M.; Iohara, K.; Murakami, M.; Nakamura, H.; Sato, Y.; Ariji, Y.; Matsushita, K. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: A pilot clinical study. Stem Cell Res. Ther. 2017, 8, 61. [Google Scholar] [CrossRef]
- Vaswani, B.K.; Mundada, B.P.; Bhola, N.; Paul, P.; Reche, A.; Ahuja, K.P. Stem-Cell Therapy: Filling Gaps in Oro-Maxillofacial Region. Cureus 2023, 15, e47171. [Google Scholar] [CrossRef]
- Miura, M.; Gronthos, S.; Zhao, M.; Lu, B.; Fisher, L.W.; Robey, P.G.; Shi, S. SHED: Stem cells from human exfoliated deciduous teeth. Proc. Natl. Acad. Sci. USA 2003, 100, 5807–5812. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, Z.; Yang, K.; Bai, Y. Research progress in cell therapy for oral diseases: Focus on cell sources and strategies to optimize cell function. Front. Bioeng. Biotechnol. 2024, 12, 1340728. [Google Scholar] [CrossRef]
- López, S.; Hoz, L.; Tenorio, E.P.; Buentello, B.; Magaña, F.S.; Wintergerst, A.; Navas, A.; Garfias, Y.; Arzate, H. Can Human Oral Mucosa Stem Cells Differentiate to Corneal Epithelia? Int. J. Mol. Sci. 2021, 22, 5976. [Google Scholar] [CrossRef]
- Calenic, B.; Greabu, M.; Caruntu, C.; Tanase, C.; Battino, M. Oral keratinocyte stem/progenitor cells: Specific markers, molecular signaling pathways and potential uses. Periodontology 2000 2015, 69, 68–82. [Google Scholar] [CrossRef]
- Hisha, H.; Tanaka, T.; Ueno, H. Lingual Epithelial Stem Cells and Organoid Culture of Them. Int. J. Mol. Sci. 2016, 17, 168. [Google Scholar] [CrossRef] [PubMed]
- Barlow, L.A. The sense of taste: Development, regeneration, and dysfunction. WIREs Mech. Dis. 2022, 14, e1547. [Google Scholar] [CrossRef] [PubMed]
- Barlow, L.A. Progress and renewal in gustation: New insights into taste bud development. Development 2015, 142, 3620–3629. [Google Scholar] [CrossRef] [PubMed]
- Tolouei, A.E.; Oruji, F.; Tehrani, S.; Rezaei, S.; Mozaffari, A.; Jahri, M.; Nasiri, K. Gingival mesenchymal stem cell therapy, immune cells, and immunoinflammatory application. Mol. Biol. Rep. 2023, 50, 10461–10469. [Google Scholar] [CrossRef]
- Kim, D.; Lee, A.E.; Xu, Q.; Zhang, Q.; Le, A.D. Gingiva-Derived Mesenchymal Stem Cells: Potential Application in Tissue Engineering and Regenerative Medicine—A Comprehensive Review. Front. Immunol. 2021, 12, 667221. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Cao, Z. Gingiva-derived Mesenchymal Stem Cells and Their Potential Applications in Oral and Maxillofacial Diseases. Curr. Stem Cell Res. Ther. 2020, 15, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Grawish, M.E. Gingival-derived mesenchymal stem cells: An endless resource for regenerative dentistry. World J. Stem Cells 2018, 10, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Fonticoli, L.; Della Rocca, Y.; Rajan, T.S.; Murmura, G.; Trubiani, O.; Oliva, S.; Pizzicannella, J.; Marconi, G.D.; Diomede, F. A Narrative Review: Gingival Stem Cells as a Limitless Reservoir for Regenerative Medicine. Int. J. Mol. Sci. 2022, 23, 4135. [Google Scholar] [CrossRef] [PubMed]
- Morsczeck, C.; Götz, W.; Schierholz, J.; Zeilhofer, F.; Kühn, U.; Möhl, C.; Sippel, C.; Hoffmann, K.H. Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol. 2005, 24, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Pisciotta, A.; Carnevale, G.; Meloni, S.; Riccio, M.; De Biasi, S.; Gibellini, L.; Ferrari, A.; Bruzzesi, G.; De Pol, A. Human dental pulp stem cells (hDPSCs): Isolation, enrichment and comparative differentiation of two sub-populations. BMC Dev. Biol. 2015, 15, 14. [Google Scholar] [CrossRef]
- Gronthos, S.; Arthur, A.; Bartold, P.M.; Shi, S. A method to isolate and culture expand human dental pulp stem cells. Methods Mol. Biol. 2011, 698, 107–121. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.C.; Krause, D.S.; Deans, R.J.; Keating, A.; Prockop, D.J.; Horwitz, E.M. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Seo, B.M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.Y.; Shi, S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Jin, S.H.; Lee, J.E.; Yun, J.H.; Kim, I.; Ko, Y.; Park, J.B. Isolation and characterization of human mesenchymal stem cells from gingival connective tissue. J. Periodontal Res. 2015, 50, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Sonoyama, W.; Liu, Y.; Yamaza, T.; Tuan, R.S.; Wang, S.; Shi, S.; Huang, G.T. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: A pilot study. J. Endod. 2008, 34, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.T.; Sonoyama, W.; Liu, Y.; Liu, H.; Wang, S.; Shi, S. The hidden treasure in apical papilla: The potential role in pulp/dentin regeneration and bioroot engineering. J. Endod. 2008, 34, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Iida, H. Oral Health Interventions during Pregnancy. Dent. Clin. N. Am. 2017, 61, 467–481. [Google Scholar] [CrossRef]
- Schröter, U.; Ziebolz, D.; Stepan, H.; Schmalz, G. Oral hygiene and oral health behavior, periodontal complaints and oral health-related quality of life in pregnant women. BMC Oral Health 2022, 22, 476. [Google Scholar] [CrossRef]
- Al Agili, D.E.; Khalaf, Z.I. The role of oral and prenatal healthcare providers in the promotion of oral health for pregnant women. BMC Pregnancy Childbirth 2023, 23, 313. [Google Scholar] [CrossRef] [PubMed]
- Popovici, D.; Crauciuc, E.; Socolov, R.; Balan, R.; Hurjui, L.; Scripcariu, I.; Pavaleanu, I. Early Diagnosis and Treatment of Dental Caries in Pregnancy. Maedica 2018, 13, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, Y.; Ding, G.; Fang, D.; Zhang, C.; Bartold, P.M.; Gronthos, S.; Shi, S.; Wang, S. Periodontal ligament stem cell-mediated treatment for periodontitis in miniature swine. Stem Cells 2008, 26, 106–114. [Google Scholar] [CrossRef]
- Pringle, S.; Van Os, R.; Coppes, R.P. Concise review: Adult salivary gland stem cells and a potential therapy for xerostomia. Stem Cells 2013, 31, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Shinmura, Y.; Tsuchiya, S.; Hata, K.I.; Honda, M.J.; Qu, H.C.; Takamoto, M.; Uno, K.; Hirose, Y.; Kamisaki, Y.; Ide, Y. Human salivary gland stem/progenitor cells remain dormant even after irradiation. Int. J. Mol. Med. 2008, 21, 209–215. [Google Scholar] [CrossRef][Green Version]
- Jing, J.; Feng, J.; Yuan, Y.; Guo, T.; Lei, J.; Pei, F.; Ho, T.V.; Chai, Y. Spatiotemporal single-cell regulatory atlas reveals neural crest lineage diversification and cellular function during tooth morphogenesis. Nat. Commun. 2022, 13, 4803. [Google Scholar] [CrossRef] [PubMed]
- Trounson, A.; McDonald, C. Stem Cell Therapies in Clinical Trials: Progress and Challenges. Cell Stem Cell 2015, 17, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Lo, B.; Parham, L. Ethical issues in stem cell research. Endocr. Rev. 2009, 30, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Miran, S.; Mitsiadis, T.A.; Pagella, P. Innovative Dental Stem Cell-Based Research Approaches: The Future of Dentistry. Stem Cells Int. 2016, 2016, 7231038. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.A.B.; Haque, A. Pregnancy-related dental problems: A review. Heliyon 2024, 10, e24259. [Google Scholar] [CrossRef]
- Shahbazi, M.N.; Jedrusik, A.; Vuoristo, S.; Recher, G.; Hupalowska, A.; Bolton, V.; Fogarty, N.N.M.; Campbell, A.; Devito, L.; Ilic, D.; et al. Self-organization of the human embryo in the absence of maternal tissues. Nat. Cell Biol. 2016, 18, 700–708. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).