Recreational Drug Overdose—Clinical Value of Toxicological Analysis
Abstract
1. Introduction
- To assess the differences in drug use between the patient history and analytical results;
- To describe the clinical characteristics, implications, and hospital management of drug-intoxicated patients based on their analytical results;
- To describe the drug use pattern over time.
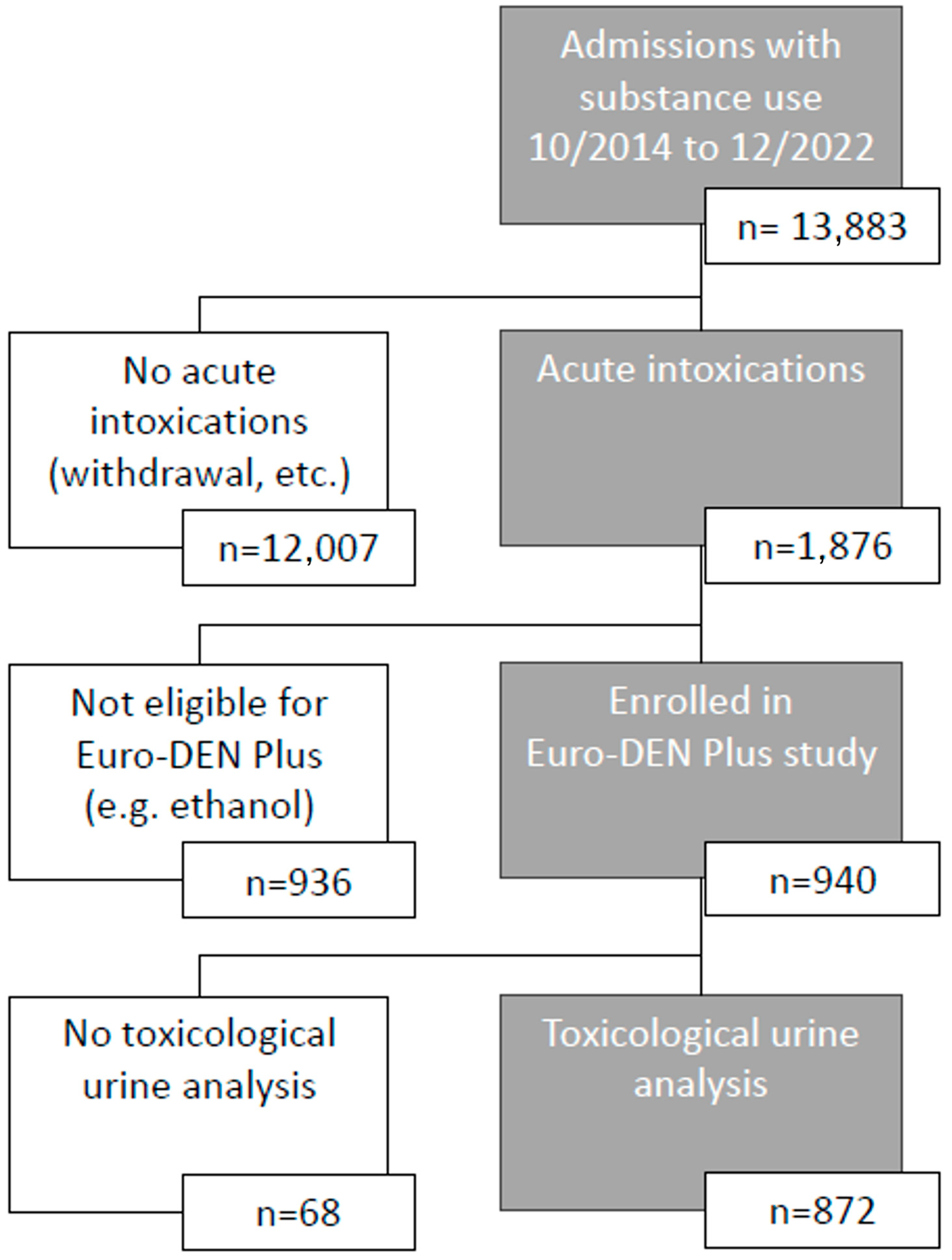
2. Materials and Methods
3. Results
3.1. Patient History vs. Analytics
3.2. Symptoms
3.3. Treatment
3.4. Outcome
3.5. Analytical Trends over Time
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Drug Agency. European Drug Report 2024: Trends and Developments. Available online: https://www.emcdda.europa.eu/publications/european-drug-report/2024_en (accessed on 12 June 2024).
- Wood, D.M.; Heyerdahl, F.; Yates, C.B.; Dines, A.M.; Giraudon, I.; Hovda, K.E.; Dargan, P.I. The European Drug Emergencies Network (Euro-DEN). Clin. Toxicol. 2014, 52, 239–241. [Google Scholar] [CrossRef] [PubMed]
- European Monitoring Centre for Drugs and Drug Addiction. Drug-Related Hospital Emergency Presentations in Europe: Update from the Euro-DEN Plus Expert Network, Technical Report. Publications Office of the European Union: Luxembourg, 2020. [Google Scholar] [CrossRef]
- Heyerdahl, F.; Hovda, K.E.; Giraudon, I.; Yates, C.; Dines, A.M.; Sedefov, R.; Wood, D.M.; Dargan, P.I. Current European data collection on emergency department presentations with acute recreational drug toxicity: Gaps and national variations. Clin. Toxicol. 2014, 52, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Pulver, B.; Fischmann, S.; Westphal, F.; Schonberger, T.; Schaper, J.; Budach, D.; Jacobsen-Bauer, A.; Dreiseitel, W.; Zagermann, J.; Damm, A.; et al. The ADEBAR project: European and international provision of analytical data from structure elucidation and analytical characterization of NPS. Drug Test. Anal. 2022, 14, 1491–1502. [Google Scholar] [CrossRef]
- Crulli, B.; Dines, A.M.; Blanco, G.; Giraudon, I.; Eyer, F.; Liechti, M.E.; Miro, O.; Hovda, K.E.; Heyerdahl, F.; Yates, C.; et al. Novel psychoactive substances-related presentations to the emergency departments of the European drug emergencies network plus (Euro-DEN plus) over the six-year period 2014–2019. Clin. Toxicol. 2022, 60, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Noseda, R.; Schmid, Y.; Scholz, I.; Liakoni, E.; Liechti, M.E.; Dargan, P.I.; Wood, D.M.; Dines, A.M.; Yates, C.; Heyerdahl, F.; et al. MDMA-related presentations to the emergency departments of the European Drug Emergencies Network plus (Euro-DEN Plus) over the four-year period 2014–2017. Clin. Toxicol. 2021, 59, 131–137. [Google Scholar] [CrossRef]
- Lyphout, C.; Yates, C.; Margolin, Z.R.; Dargan, P.I.; Dines, A.M.; Heyerdahl, F.; Hovda, K.E.; Giraudon, I.; Bucher-Bartelson, B.; Green, J.L.; et al. Presentations to the emergency department with non-medical use of benzodiazepines and Z-drugs: Profiling and relation to sales data. Eur. J. Clin. Pharmacol. 2019, 75, 77–85. [Google Scholar] [CrossRef]
- Galicia, M.; Dargan, P.I.; Dines, A.M.; Yates, C.; Heyerdahl, F.; Hovda, K.E.; Giraudon, I.; Euro, D.E.N.P.R.G.; Wood, D.M.; Miro, O.; et al. Clinical relevance of ethanol coingestion in patients with GHB/GBL intoxication. Toxicol. Lett. 2019, 314, 37–42. [Google Scholar] [CrossRef]
- Heier, E.C.; Eyer, F.; Rabe, C.; Geith, S.; Dargan, P.I.; Wood, D.M.; Heyerdahl, F.; Dines, A.M.; Giraudon, I.; Erik Hovda, K.; et al. Clinical effect of ethanol co-use in patients with acute drug toxicity involving the use of central nervous system depressant recreational drugs. Eur. J. Emerg. Med. 2022, 29, 291–300. [Google Scholar] [CrossRef]
- Tenenbein, M. Do you really need that emergency drug screen? Clin. Toxicol. 2009, 47, 286–291. [Google Scholar] [CrossRef]
- Beckman Coulter. Available online: https://www.beckmancoulter.com/de/products/chemistry/au480 (accessed on 1 September 2024).
- ThermoFisher. Available online: https://www.thermofisher.com/de/de/home/industrial/chromatography/liquid-chromatography-lc/hplc-uhplc-systems/ultimate-3000-hplc-uhplc-systems.html (accessed on 1 September 2024).
- Agilent. 09/2024. Available online: https://www.agilent.com/en/product/gas-chromatography/gc-systems/8860-gc-system (accessed on 1 September 2024).
- Hunt, D.E.; Kling, R.; Almozlino, Y.; Jalbert, S.; Chapman, M.T.; Rhodes, W. Telling the Truth About Drug Use:How Much Does It Matter? J. Drug Issues 2015, 45, 314–329. [Google Scholar] [CrossRef]
- Monte, A.A.; Heard, K.J.; Hoppe, J.A.; Vasiliou, V.; Gonzalez, F.J. The accuracy of self-reported drug ingestion histories in emergency department patients. J. Clin. Pharmacol. 2015, 55, 33–38. [Google Scholar] [CrossRef]
- Brandt, S.D.; Kavanagh, P.V. Addressing the challenges in forensic drug chemistry. Drug Test. Anal. 2017, 9, 342–346. [Google Scholar] [CrossRef]
- Verdu, G.; Rodriguez Garcia, M.; Martinez, E.; Mares, L.; Martin, E.; To-Figueras, J.; Salgado, E.; Brunet, M.; Parra-Robert, M. Dipentylone: A novel synthetic cathinone in Spain’s illicit drug landscape. Findings in a clinical toxicology laboratory. Clin. Toxicol. 2024, 62, 209–211. [Google Scholar] [CrossRef]
- Liakoni, E.; Yates, C.; Dines, A.M.; Dargan, P.I.; Heyerdahl, F.; Hovda, K.E.; Wood, D.M.; Eyer, F.; Liechti, M.E.; Euro-DEN Plus Research Group. Acute recreational drug toxicity: Comparison of self-reports and results of immunoassay and additional analytical methods in a multicenter European case series. Medicine 2018, 97, e9784. [Google Scholar] [CrossRef]
- Bharat, C.; Webb, P.; Wilkinson, Z.; McKetin, R.; Grebely, J.; Farrell, M.; Holland, A.; Hickman, M.; Tran, L.T.; Clark, B.; et al. Agreement between self-reported illicit drug use and biological samples: A systematic review and meta-analysis. Addiction 2023, 118, 1624–1648. [Google Scholar] [CrossRef]
- European Union Drugs Agency. European Drug Emergencies Network (Euro-DEN Plus): Data and Analysis. Available online: https://www.euda.europa.eu/publications/data-factsheet/european-drug-emergencies-network-euro-den-plus-data-and-analysis_en (accessed on 1 September 2024).
- Romanek, K.; Stenzel, J.; Schmoll, S.; Schrettl, V.; Geith, S.; Eyer, F.; Rabe, C. Synthetic cathinones in Southern Germany—Characteristics of users, substance-patterns, co-ingestions, and complications. Clin. Toxicol. 2017, 55, 573–578. [Google Scholar] [CrossRef]
- Schmoll, S.; Romanek, K.; Stich, R.; Bekka, E.; Stenzel, J.; Geith, S.; Eyer, F.; Rabe, C. An internet-based survey of 96 German-speaking users of “bath salts”: Frequent complications, risky sexual behavior, violence, and delinquency. Clin. Toxicol. 2018, 56, 219–222. [Google Scholar] [CrossRef]
- Vallersnes, O.M.; Dines, A.M.; Wood, D.M.; Yates, C.; Heyerdahl, F.; Hovda, K.E.; Giraudon, I.; Euro, D.E.N.R.G.; Dargan, P.I. Psychosis associated with acute recreational drug toxicity: A European case series. BMC Psychiatry 2016, 16, 293. [Google Scholar] [CrossRef]
- Waters, M.L.; Dargan, P.I.; Yates, C.; Dines, A.M.; Eyer, F.; Giraudon, I.; Heyerdahl, F.; Hovda, K.E.; Liechti, M.E.; Miró, Ò.; et al. Clinical effects of cannabis compared to synthetic cannabinoid receptor agonists (SCRAs): A retrospective cohort study of presentations with acute toxicity to European hospitals between 2013 and 2020. Clin. Toxicol. 2024, 62, 378–384. [Google Scholar] [CrossRef]
- Noseda, R.; Franchi, M.; Pagnamenta, A.; Muller, L.; Dines, A.M.; Giraudon, I.; Heyerdahl, F.; Eyer, F.; Hovda, K.E.; Liechti, M.E.; et al. Determinants of Admission to Critical Care Following Acute Recreational Drug Toxicity: A Euro-DEN Plus Study. J. Clin. Med. 2023, 12, 5970. [Google Scholar] [CrossRef]
- Bundesministerium für Gesundheit. Das Neue-psychoaktive-Stoffe-Gesetz (NpSG). Available online: https://www.bundesgesundheitsministerium.de/service/begriffe-von-a-z/n/npsg (accessed on 1 September 2024).
- Penninga, E.I.; Graudal, N.; Ladekarl, M.B.; Jurgens, G. Adverse Events Associated with Flumazenil Treatment for the Management of Suspected Benzodiazepine Intoxication--A Systematic Review with Meta-Analyses of Randomised Trials. Basic Clin. Pharmacol. Toxicol. 2016, 118, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Pergolizzi, J., Jr.; Raffa, R.; LeQuang, J.A.K.; Breve, F.; Varrassi, G. Old Drugs and New Challenges: A Narrative Review of Nitazenes. Cureus 2023, 15, e40736. [Google Scholar] [CrossRef] [PubMed]
- Vallersnes, O.M.; Dines, A.M.; Wood, D.M.; Heyerdahl, F.; Hovda, K.E.; Yates, C.; Giraudon, I.; Caganova, B.; Ceschi, A.; Galicia, M.; et al. Self-discharge during treatment for acute recreational drug toxicity: An observational study from emergency departments in seven European countries. Int. J. Emerg. Med. 2023, 16, 86. [Google Scholar] [CrossRef]
- Grafinger, K.E.; Liechti, M.E.; Liakoni, E. Clinical value of analytical testing in patients presenting with new psychoactive substances intoxication. Br. J. Clin. Pharmacol. 2020, 86, 429–436. [Google Scholar] [CrossRef]
- Dufayet, L.; Bargel, S.; Bonnet, A.; Boukerma, A.K.; Chevallier, C.; Evrard, M.; Guillotin, S.; Loeuillet, E.; Paradis, C.; Pouget, A.M.; et al. Gamma-hydroxybutyrate (GHB), 1,4-butanediol (1,4BD), and gamma-butyrolactone (GBL) intoxication: A state-of-the-art review. Regul. Toxicol. Pharmacol. 2023, 142, 105435. [Google Scholar] [CrossRef]

| Substance Class | History | Analytics | PPV | NPV | |
|---|---|---|---|---|---|
| All opiates and opioids | Yes | 322 (36.9%) | 499 (57.2%) | 93.8% (90.6–95.9%) | 64.2% (60.1–68.1%) |
| No | 550 (63.1%) | 373 (42.8%) | |||
| Buprenorphine | Yes | 51 (5.8%) | 157 (18.0%) | 82.4% (69.7–90.4%) | 86.0% (83.5–88.2%) |
| No | 821 (94.2%) | 715 (82.0%) | |||
| Methadone | Yes | 109 (12.5%) | 194 (22.2%) | 88.1% (80.7–92.9%) | 87.2% (84.6–89.4%) |
| No | 763 (87.5%) | 678 (77.8%) | |||
| Opiates | Yes | 162 (18.6%) | 252 (28.9%) | 78.4% (71.4–84.0%) | 82.4% (79.4–85.1%) |
| No | 710 (81.4%) | 620 (71.1%) | |||
| Opioids | Yes | 35 (4.0%) | 67 (7.7%) | 51.4% (35.6–67.0%) | 94.1% (92.4–95.6%) |
| No | 837 (96.0%) | 805 (92.3%) | |||
| Amphetamines and MDMA | Yes | 116 (13.3%) | 186 (21.3%) | 73.3% (64.6–80.5%) | 86.6% (84.0–88.9%) |
| No | 756 (86.7%) | 686 (78.7%) | |||
| Benzodiazepines and Z-drugs | Yes | 181 (20.8%) | 467 (53.6%) | 96.1% (92.2–98.1%) | 57.6% (53.4–60.7%) |
| No | 691 (79.2%) | 405 (46.4%) | |||
| Cathinones and phenethylamines | Yes | 92 (10.6%) | 70 (8.0%) | 41.3% (31.8–51.5%) | 95.9% (94.3–97.1%) |
| No | 780 (89.4%) | 802 (92.0%) | |||
| Cocaine | Yes | 76 (8.7%) | 146 (16.7%) | 78.9% (68.5–86.6%) | 89.2% (86.9–91.2%) |
| No | 796 (91.3%) | 726 (83.3%) | |||
| GBL/GHB | Yes | 17 (1.9%) | 32 (3.7%) | 94.1% (73.0–99.0%) | 98.1% (97.0–98.9%) |
| No | 855 (98.1%) | 840 (93.3%) | |||
| Ketamine | Yes | 6 (0.7%) | 4 (0.5%) | 50% (18.8–81.2%) | 99.9% (99.3–100%) |
| No | 866 (99.3%) | 868 (99.5%) | |||
| LSD | Yes | 17 (1.9%) | 9 (1.0%) | 47.1% (26.2–69.0%) | 99.9% (99.4–100%) |
| No | 855 (98.1%) | 863 (99.0%) | |||
| Pregabalin | Yes | 131 (15%) | 387 (44.4%) | 90.1% (83.8–94.1%) | 63.7% (60.2–67.1%) |
| No | 741 (85.0%) | 485 (55.6%) | |||
| SCRA | Yes | 71 (8.1%) | 53 (6.1%) | 50.7% (39.3–62.0%) | 97.9% (96.6–98.7%) |
| No | 801 (91.9%) | 819 (93.9%) | |||
| THC | Yes | 86 (9.9%) | 329 (37.7%) | 80.2% (70.6–87.3%) | 66.9% (63.6–70.1%) |
| No | 786 (90.1%) | 543 (62.3%) | |||
| Symptoms | All n = 872 | All Opiates and Opioids n = 499 | p | BZD and Z-Drugs n = 467 | p | Cathinones and PA n = 70 | p | SCRA n = 53 | p | |
|---|---|---|---|---|---|---|---|---|---|---|
| Coma on admission | Yes | 222 (25.5%) | 150 (30.1%) | <0.001 * | 140 (30.0%) | 0.001 * | 18 (25.7%) | 0.959 | 9 (17.0%) | 0.144 |
| No | 650 (74.5%) | 349 (69.9%) | 327 (70.0%) | 52 (74.3%) | 44 (83.0%) | |||||
| Vomiting | Yes | 40 (4.6%) | 18 (3.6%) | 0.110 | 15 (3.2%) | 0.037 * | 5 (7.1%) | 0.287 | 8 (15.1%) | <0.001 * |
| No | 832 (95.4%) | 481 (96.4%) | 452 (96.8%) | 65 (92.9%) | 45 (84.9%) | |||||
| Hyperthermia | Yes | 32 (3.7%) | 22 (4.4%) | 0.179 | 19 (4.1%) | 0.501 | 7 (10.0%) | 0.003 * | 3 (5.7%) | 0.426 |
| No | 840 (96.3%) | 477 (95.6%) | 448 (95.9%) | 63 (90.0%) | 50 (94.3%) | |||||
| Headache | Yes | 17 (1.9%) | 5 (1.0%) | 0.019 * | 6 (1.3%) | 0.127 | 1 (1.4%) | 0.742 | 2 (3.8%) | 0.322 |
| No | 855 (98.1%) | 494 (99.0%) | 461 (98.7%) | 69 (98.6%) | 51 (96.2%) | |||||
| Anxiety | Yes | 176 (20.2%) | 51 (10.2%) | <0.001 * | 51 (10.9%) | <0.001 * | 20 (28.6%) | 0.068 | 12 (22.6%) | 0.645 |
| No | 696 (79.8%) | 448 (89.8%) | 416 (89.1%) | 50 (71.4%) | 41 (77.4%) | |||||
| Hallucinations | Yes | 156 (17.9%) | 50 (10.0%) | <0.001 * | 50 (10.7%) | <0.001 * | 27 (38.6%) | <0.001 * | 14 (26.4%) | 0.095 |
| No | 716 (82.1%) | 449 (90.0%) | 417 (89.3%) | 43 (61.4%) | 39 (73.6%) | |||||
| Agitation and aggression | Yes | 330 (37.8%) | 119 (23.8%) | <0.001 * | 125 (26.8%) | <0.001 * | 47 (67.1%) | <0.001 * | 26 (49.1%) | 0.082 |
| No | 542 (62.2%) | 380 (76.2%) | 342 (73.2%) | 23 (32.9%) | 27 (50.9%) | |||||
| Psychosis | Yes | 79 (9.1%) | 26 (5.2%) | <0.001 * | 25 (5.4%) | <0.001 * | 10 (14.3%) | 0.112 | 6 (11.3%) | 0.554 |
| No | 793 (90.9%) | 473 (94.8%) | 442 (94.6%) | 60 (85.7%) | 47 (88.7%) | |||||
| Seizures | Yes | 58 (9.1%) | 25 (5.0%) | 0.024 * | 20 (4.3%) | 0.003 * | 6 (8.6%) | 0.501 | 7 (13.2%) | 0.048 * |
| No | 793 (90.9%) | 474 (95.0%) | 447 (95.7%) | 64 (91.4%) | 46 (86.8%) | |||||
| Palpitations | Yes | 36 (4.1%) | 8 (1.6%) | <0.001 * | 6 (1.3%) | <0.001 * | 4 (5.7%) | 0.487 | 1 (1.9%) | 0.397 |
| No | 836 (95.9%) | 491 (98.4%) | 461 (98.7%) | 66 (94.3%) | 52 (98.1%) | |||||
| Chest pain | Yes | 24 (2.8%) | 11 (2.2%) | 0.253 | 7 (1.5%) | 0.015 * | 3 (4.3%) | 0.414 | 1 (1.9%) | 0.691 |
| No | 848 (97.2%) | 488 (97.8%) | 460 (98.5%) | 67 (95.7%) | 52 (98.1%) | |||||
| Hypertension | Yes | 35 (4.0%) | 13 (2.6%) | 0.014 * | 9 (1.9%) | <0.001 * | 1 (1.4%) | 0.251 | 2 (3.8%) | 0.927 |
| No | 837 (96.0%) | 486 (97.4%) | 458 (98.1%) | 69 (98.6%) | 51 (96.2%) | |||||
| Hypotension | Yes | 50 (5.7%) | 34 (6.8%) | 0.113 | 31 (6.6%) | 0.217 | 4 (5.7%) | 0.994 | 6 (11.3%) | 0.071 |
| No | 822 (94.3%) | 465 (93.2%) | 436 (93.4%) | 66 (94.3%) | 47 (88.7%) | |||||
| Arrhythmias | Yes | 8 (0.9%) | 5 (1.0%) | 0.762 | 3 (0.6%) | 0.360 | 0 (0%) | 0.401 | 3 (5.7%) | <0.001 * |
| No | 864 (99.1%) | 494 (99.0%) | 464 (99.4%) | 70 (100%) | 50 (94.3%) | |||||
| Coma during treatment | Yes | 249 (28.6%) | 166 (33.3%) | <0.001 * | 155 (33.2%) | 0.001 * | 25 (36.2%) | 0.145 | 11 (21.2%) | 0.219 |
| No | 621 (71.2%) | 332 (66.7%) | 312 (66.8%) | 44 (63.8%) | 41 (78.8%) | |||||
| Treatment | All n = 872 | All Opiates and Opioids n = 499 | p | BZD and Z-Drugs n = 467 | p | Cathinones and PA n = 70 | p | SCRA n = 53 | p | |
|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | Yes | 846 (97.0%) | 492 (98.6%) | 0.002 * | 461 (98.7%) | 0.002 * | 69 (98.6%) | 0.426 | 46 (86.8%) | <0.001 * |
| No | 26 (3.0%) | 7 (1.4%) | 6 (1.3%) | 1 (1.4%) | 7 (13.2%) | |||||
| Intubation | Pre-hospital | 37 (4.2%) | 25 (5.0%) | 0.057 | 21 (4.5%) | 0.723 | 3 (4.3%) | 0.304 | 1 (1.9%) | 0.694 |
| Hospital | 24 (2.8%) | 17 (3.4%) | 13 (2.8%) | 4 (5.7%) | 2 (3.8%) | |||||
| No | 811 (93.0%) | 457 (91.6%) | 433 (92.7%) | 63 (90.0%) | 50 (94.3%) | |||||
| Naloxone | Pre-hospital | 44 (5.0%) | 39 (7.8%) | <0.001 * | 26 (5.6%) | 0.011 * | 2 (2.9%) | 0.638 | 0 (0%) | 0.011 |
| Hospital | 38 (4.4%) | 32 (6.4%) | 29 (6.2%) | 4 (5.7%) | 0 (0%) | |||||
| Both | 7 (0.8%) | 7 (1.4%) | 4 (0.9%) | 0 (0%) | 0 (0%) | |||||
| No | 783 (89.8%) | 421 (84.4%) | 408 (87.4%) | 64 (91.4%) | 53 (100%) | |||||
| Flumazenil | Pre-hospital | 18 (2.1%) | 13 (2.6%) | 0.002 * | 14 (3.0%) | <0.001 * | 0 (0%) | 0.878 | 0 (0%) | 0.076 |
| Hospital | 28 (3.2%) | 23 (4.6%) | 24 (5.1%) | 4 (5.8%) | 0 (0%) | |||||
| Both | 1 (0.1%) | 1 (0.2%) | 1 (0.2%) | 0 (0%) | 0 (0%) | |||||
| No | 824 (94.5%) | 462 (92.6%) | 428 (91.6%) | 65 (94.2%) | 52 (100%) | |||||
| Sedation | Pre-hospital | 53 (6.1%) | 22 (4.4%) | <0.001 * | 11 (2.4%) | <0.001 * | 5 (7.1%) | <0.001 * | 10 (18.9%) | <0.001 * |
| Hospital | 219 (25.1%) | 90 (18.1%) | 95 (20.3%) | 27 (38.6%) | 9 (17.0%) | |||||
| Both | 70 (8.0%) | 30 (6.0%) | 27 (5.8%) | 11 (15.7%) | 3 (5.7%) | |||||
| No | 529 (60.7%) | 356 (71.5%) | 334 (71.5%) | 27 (38.6%) | 31 (58.5%) | |||||
| Substance Class | Cardiac Arrest n = 12 | p | Death n = 4 | p | |
|---|---|---|---|---|---|
| All opiates and opioids | Yes | 12 (100%) | 0.003 * | 3 (75.0%) | 0.471 |
| No | 0 (0%) | 1 (25.0%) | |||
| Buprenorphine | Yes | 3 (25.0%) | 0.525 | 1 (25.0%) | 0.715 |
| No | 9 (75.0%) | 3 (75.0%) | |||
| Methadone | Yes | 2 (16.7%) | 0.640 | 0 (0%) | 0.284 |
| No | 10 (83.3%) | 4 (100%) | |||
| Opiates | Yes | 10 (83.3%) | <0.001 * | 3 (75.0%) | 0.041 * |
| No | 2 (16.7%) | 1 (25.0%) | |||
| Opioids | Yes | 2 (16.7%) | 0.239 | 0 (0%) | 0.563 |
| No | 10 (83.3%) | 4 (100%) | |||
| Amphetamines and MDMA | Yes | 0 (0%) | 0.069 | 1 (25.0%) | 0.862 |
| No | 12 (100%) | 3 (75.0%) | |||
| Benzodiazepines and Z-drugs | Yes | 9 (75.0%) | 0.134 | 3 (75.0%) | 0.389 |
| No | 3 (25.0%) | 1 (25.0%) | |||
| Cathinones and phenethylamines | Yes | 0 (0%) | 0.303 | 1 (25.0%) | 0.211 |
| No | 12 (100%) | 3 (75.0%) | |||
| Cocaine | Yes | 2 (16.7%) | 0.994 | 0 (0%) | 0.369 |
| No | 10 (83.3%) | 4 (100%) | |||
| Ethanol | Yes | 5 (41.7%) | 0.985 | 1 (25.0%) | 0.505 |
| No | 7 (58.3%) | 3 (75.0%) | |||
| GBL/GHB | Yes | 0 (0%) | 0.496 | 0 (0%) | 0.696 |
| No | 12 (100%) | 4 (100%) | |||
| Ketamine | Yes | 0 (0%) | 0.813 | 0 (0%) | 0.892 |
| No | 12 (100%) | 4 (100%) | |||
| LSD | Yes | 0 (0%) | 0.722 | 0 (0%) | 0.838 |
| No | 12 (100%) | 4 (100%) | |||
| Pregabalin | Yes | 9 (75.0%) | 0.032 * | 2 (50.0%) | 0.817 |
| No | 3 (25.0%) | 2 (50.0%) | |||
| SCRA | Yes | 0 (0%) | 0.375 | 0 (0%) | 0.610 |
| No | 12 (100%) | 4 (100%) | |||
| THC | Yes | 3 (25.0%) | 0.360 | 0 (0%) | 0.119 |
| No | 9 (75.0%) | 4 (100%) | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zellner, T.; Eyer, F.; Rabe, C.; Geith, S.; Haberl, B.; Schmoll, S. Recreational Drug Overdose—Clinical Value of Toxicological Analysis. Toxics 2024, 12, 662. https://doi.org/10.3390/toxics12090662
Zellner T, Eyer F, Rabe C, Geith S, Haberl B, Schmoll S. Recreational Drug Overdose—Clinical Value of Toxicological Analysis. Toxics. 2024; 12(9):662. https://doi.org/10.3390/toxics12090662
Chicago/Turabian StyleZellner, Tobias, Florian Eyer, Christian Rabe, Stefanie Geith, Bettina Haberl, and Sabrina Schmoll. 2024. "Recreational Drug Overdose—Clinical Value of Toxicological Analysis" Toxics 12, no. 9: 662. https://doi.org/10.3390/toxics12090662
APA StyleZellner, T., Eyer, F., Rabe, C., Geith, S., Haberl, B., & Schmoll, S. (2024). Recreational Drug Overdose—Clinical Value of Toxicological Analysis. Toxics, 12(9), 662. https://doi.org/10.3390/toxics12090662









