Cardiovascular Disease: From Molecular and Cellular Mechanisms to Therapeutic Opportunities
A topical collection in Cells (ISSN 2073-4409). This collection belongs to the section "Cells of the Cardiovascular System".
Viewed by 158967Editor
Interests: cardiovascular research; regenerative medicine; stem cells; exosomes; inflammation; noncoding RNAs; diabetes; angiogenesis; signal transduction
Topical Collection Information
Dear Colleagues,
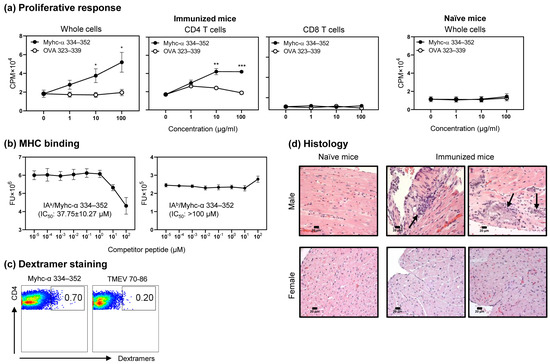
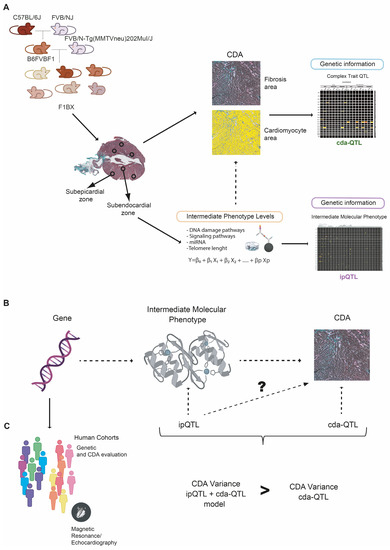
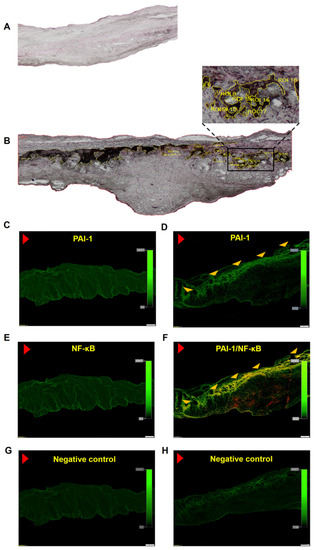
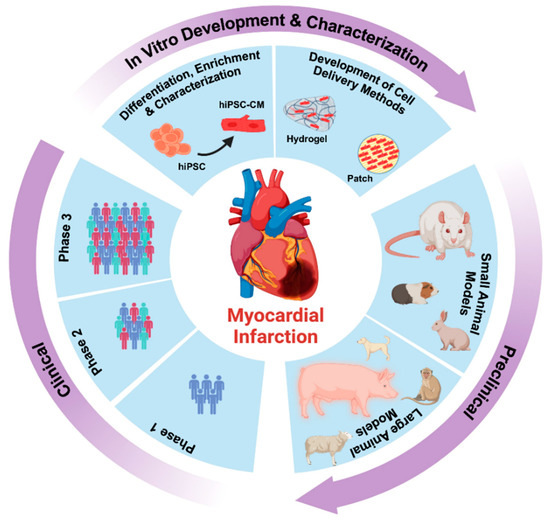
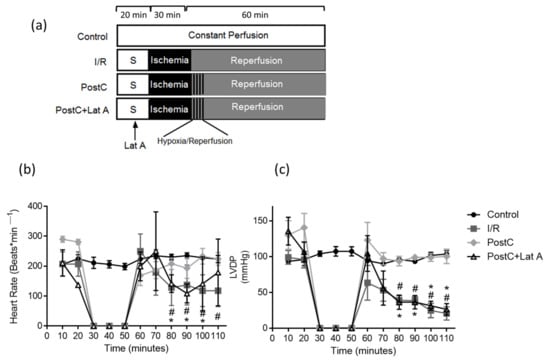
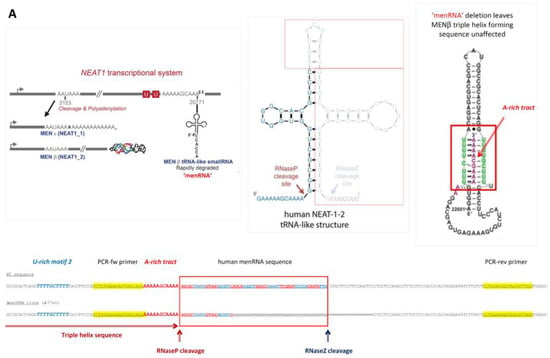
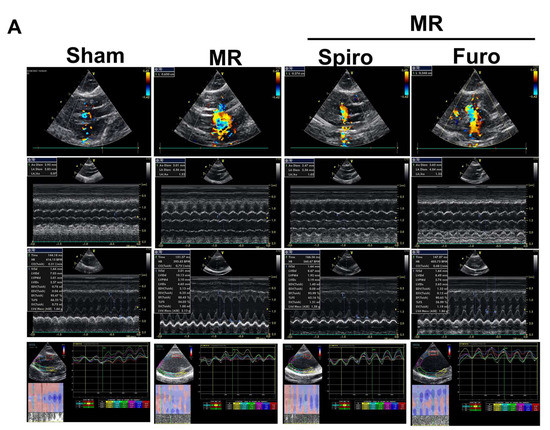
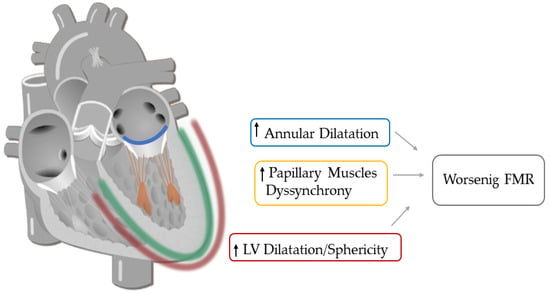
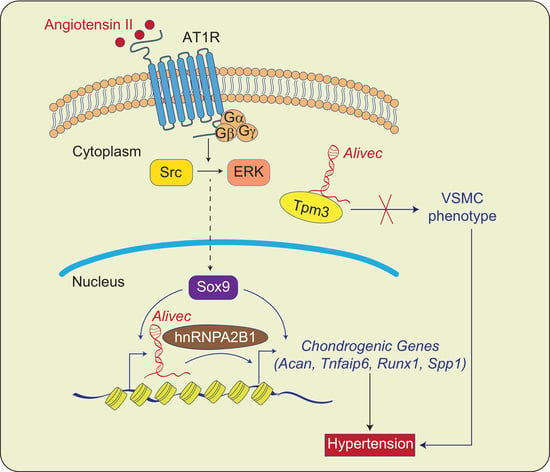
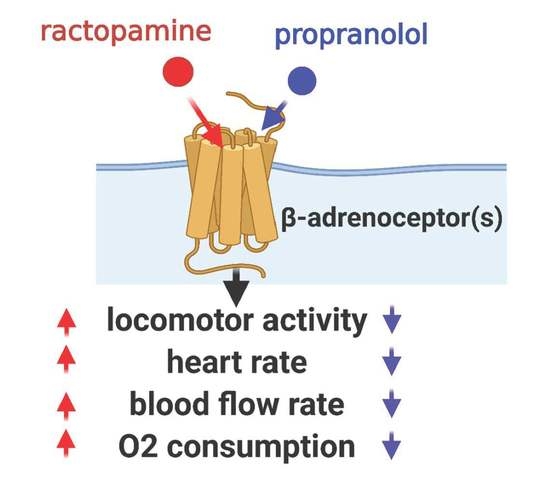
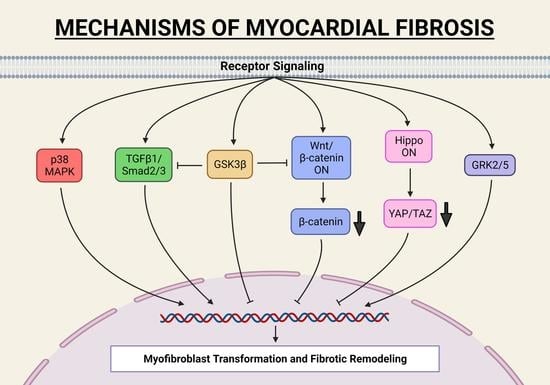
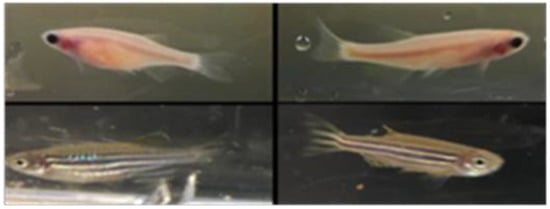
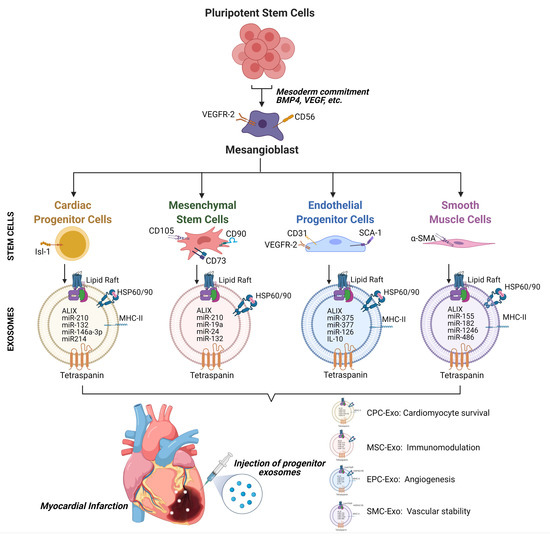
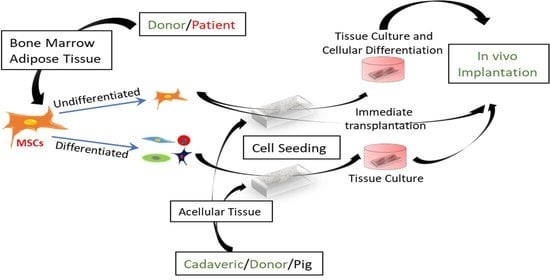
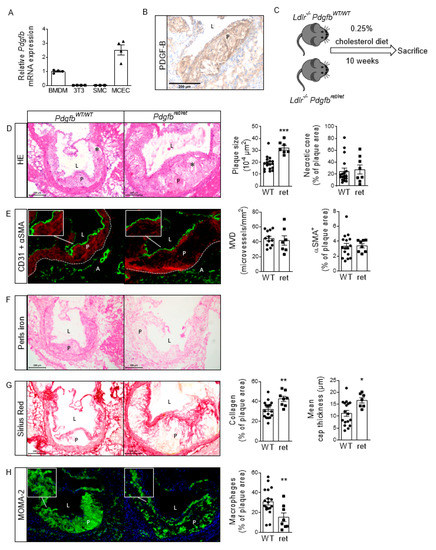
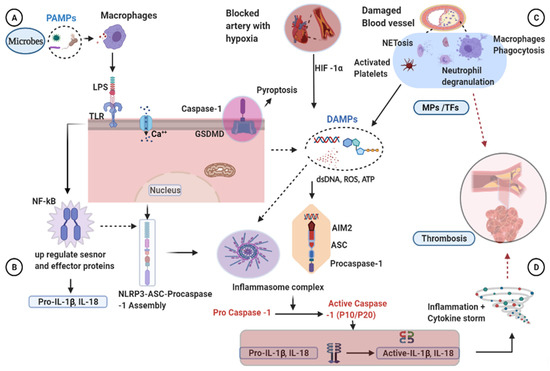
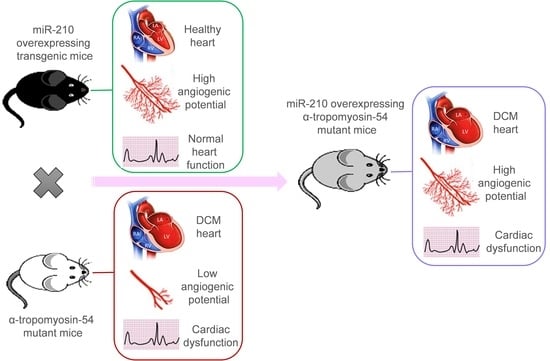
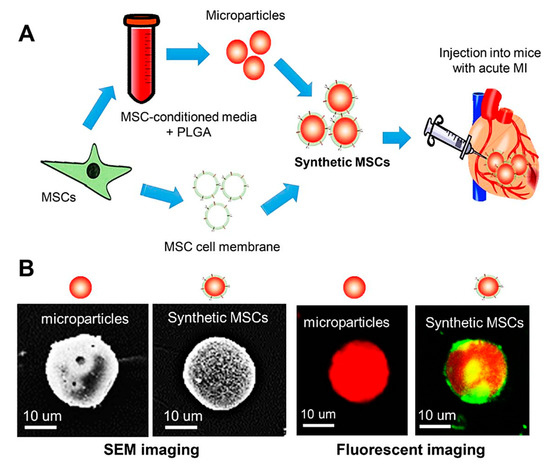
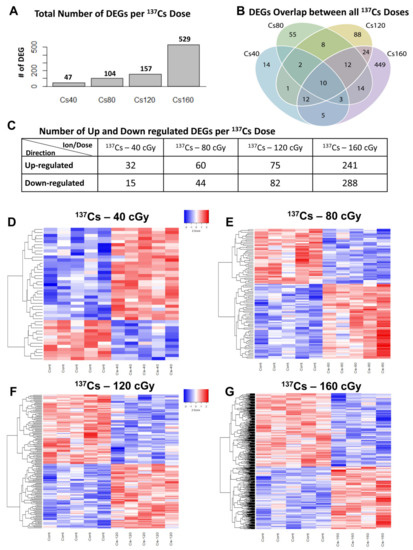
Despite the improvement shown in the prognosis of patients with myocardial infarction (MI) with the use of available therapies, including thrombolysis and urgent coronary revascularizations, a significant proportion of MI survivors are at risk of developing heart failure, which remains a major health concern worldwide. Surgical approaches such as cardiac transplants and artificial implants aside, current interventional strategies are geared toward providing symptomatic relief. It is imperative, therefore, to identify new mechanisms and to develop novel treatment strategies to minimize subsequent cardiac remodeling that can adversely affect heart function. In this context, identifying new targets to improve tissue repair, including preservation of the cardiac microvasculature, clearance of apoptotic cells, and tissue regeneration, are of great interest.
Cells is planning to publish a Topical Collection in current year titled “Cardiovascular Disease: From Molecular and Cellular Mechanisms to Therapeutic Opportunities” that will solicit original research articles, short communications, and brief reviews highlighting new exciting findings in the areas of cardiovascular biology, disease, and new therapeutic target identification and validation. Our goal is to invite submissions that reflect novel, exciting research on broad topics in cardiovascular disease and mechanisms, including but not limited to stem cell biology, signaling mechanisms, cardiac protection, non-coding RNAs, and epigenetic and genetic approaches related to cardiovascular disease mechanisms and therapies. Each submitted manuscript will go through a rigorous and peer-review process. Submitted manuscripts must not have been published previously nor be under consideration at other journals.
Prof. Dr. Raj Kishore
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as short communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cells is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2700 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- cardiovascular injury and repair
- cardioprotection
- signaling mechanisms
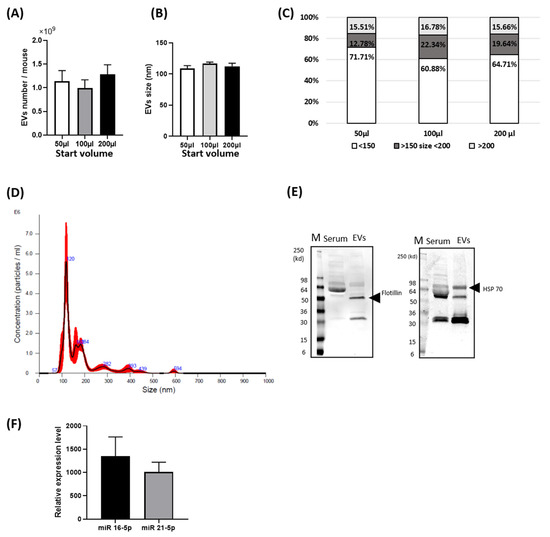
- stem cells and extracellular vesicles
- noncoding RNAs
- angiogenesis
- therapeutics