Nutrient Uptake of Two Semidomesticated Jaltomata Schltdl. Species for Their Cultivation
Abstract
1. Introduction
2. Results
2.1. Analysis of Effects on the Nutrient Concentration and Total Dry Matter
2.2. Macronutrients
2.3. Micronutrients
2.4. Electrical Conductivity
2.5. Sps × EC Combination
2.6. Order of Macronutrients and Micronutrients Uptake
2.7. Total Dry Matter
2.8. Sps x EC Combination
2.9. Total Dry Matter Accumulation Order by Plant Structure
3. Discussion
4. Materials and Methods
4.1. Genetic Material
4.2. Experiment Management
4.3. Experimental Design
4.4. Variables Evaluated
4.4.1. Macro- and Micronutrients Analysis in Leaves
4.4.2. Plant Characteristics
4.4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- González, C.F.; Cabezas, G.M.; Ramírez-Gómez, M.; Ramírez, D.J. Curvas de absorción de macronutrientes en tres variedades de caña de azúcar (Saccharum officinarum L.) para panela, en la hoya del río Suárez. Rev. UDCA Actual. Divulg. Cient. 2018, 21, 395–404. [Google Scholar] [CrossRef]
- Gastelum-Osorio, D.A.; Sandoval-Villa, M.; Trejo-López, C.; Castro-Brindis, R. Fuerza iónica de la solución nutritiva y densidad de plantación sobre la producción y calidad de frutos de Physalis peruviana L. Rev. Chapingo Ser. Hortic. 2013, 19, 197–210. [Google Scholar] [CrossRef]
- Mione, T.; Leiva, G.S.; Yacher, L. Jaltomata sanmiguelina (Solanaceae), a new species from northern Peru. University Proceedings, Volga region. Nat. Sci. 2015, 3, 3–13. [Google Scholar]
- Flores-Sánchez, I.D.; Sandoval-Villa, M.; Uscanga-Mortera, E.; Silva-Rojas, H.V. Jaltomata tlaxcala, a new species of the genus Jaltomata (Solanaceae, Solanoideae, Solaneae). Phytotaxa 2024, 641, 85–98. [Google Scholar] [CrossRef]
- Fonteyne, S.; Castillo, C.J.B.; Lopez-Ridaura, S.; Van, L.J.; Espidio, B.J.; Osorio, A.L.; Martínez, H.F.; Odjo, S.; Verhulst, N. Review of agronomic research on the milpa, the traditional polyculture system of Mesoamerica. Front. Agron. 2023, 5, 1115490. [Google Scholar] [CrossRef]
- Ortiz, G.S.; Saynes, S.V.; Bunge, V.V.; Anglés-Hernández, M.; Pérez, M.E.; Prado, B. Soil governance and sustainable agriculture in Mexico. Soil Secur. 2022, 7, 100059. [Google Scholar] [CrossRef]
- Flores-Sánchez, I.D.; Sandoval-Villa, M.; Alcántar-González, E.G.; Sánchez-García, P.; Soto-Hernández, R.M.; Uscanga-Mortera, E. Caracteres agronómicos de dos poblaciones de Jaltomata procumbens (Cav.) J. L. Gentry. Agron. Mesoam. 2021, 32, 733–749. [Google Scholar] [CrossRef]
- Flores-Sánchez, I.D.; Sandoval-Villa, M.; Soto-Hernández, R.M. Effects of electrical conductivity and pruning on secondary metabolite contents in fruits of Jaltomata procumbens (Cav.) J. L. Gentry. Nat. Prod. Commun. 2023, 18, 1–11. Available online: https://journals.sagepub.com/doi/10.1177/1934578X221150547 (accessed on 12 May 2024). [CrossRef]
- Mera-Ovando, L.M.; Alvarado-Flores, R.; Basurto-peña, F.; Bye-Bottler, R.; Castro-Lara, D.; Evangelista, V.; Mapes-Sánchez, C.; Martínez-Alfaro, M.Ä.; Molina, N.; Saldívar, J. “De quelites me como un taco”. Experiencia en educación nutricional. Rev. Jardín Botánico Nac. 2003, 24, 45–49. Available online: https://www.jstor.org/stable/42597191 (accessed on 22 June 2021).
- Williams, D.E. Tres Arvenses Solanáceas Comestibles y su Proceso de Domesticación en el Estado de Tlaxcala, México. Master’s Thesis, Colegio de Postgraduados, Montecillo, Mexico, 1985. Available online: http://catalogo.colpos.mx/cgi-bin/koha/opac-detail.pl?biblionumber=16343 (accessed on 7 March 2022).
- WHO. Guidance on Mainstreaming Biodiversity for Nutrition and Health; World Health Organization: Geneva, Switzerland, 2020; pp. 1–100. Available online: https://www.who.int/publications/i/item/9789240006690 (accessed on 18 September 2022).
- Li, X.; Siddique, K.H.M.; Akinnifesi, F.; Callens, K.; Broca, S.; Noorani, A.; Henrich, G.; Chikelu, M.; Bayasgalanbat, N. Introduction: Setting the scene. In Future Smart Food—Rediscovering Hidden Treasures of Neglected and Underutilized Species for Zero Hunger in Asia; Xuan, L., Kadambot, H.M.S., Eds.; FAO: Rome, Italy, 2018; pp. 15–32. Available online: http://www.fao.org/3/I8907EN/i8907en.pdf (accessed on 5 May 2022).
- FAO. Neglected and Underutilized Crops Species; COAG/2018/INF/7; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018; pp. 1–5. Available online: https://openknowledge.fao.org/items/3f2c9e30-d29c-4908-94d0-511fe4ffaeaf (accessed on 25 July 2023).
- N’Danikou, S.; Tchokponhoue, D.A. Plant domestication for enhanced food security. In Zero Hunger; Filho, W.L., Azul, A., Brandli, L., Özuyar, P., Wall, T., Eds.; Springer: Cham, Switzerland, 2019; pp. 1–11. [Google Scholar] [CrossRef]
- Li, X.; Yadav, R.; Siddique, K.H.M. Neglected and underutilized crop species: The key to improving dietary diversity and fighting hunger and malnutrition in Asia and the Pacific. Front. Nutr. 2020, 7, 593711. [Google Scholar] [CrossRef]
- Sarukhán, J.; (Universidad Nacional Autónoma de México, Ciudad de México, Mexico). Personal communication, 2020.
- Castro-Villarreal, J.A.; Villarreal-Núñez, J.E. Absorción de nutrientes en tomate industrial IDIAP–T7 en suelos de la región de Azuero. Cienc. Agropecu. 2020, 30, 27–44. Available online: http://www.revistacienciaagropecuaria.ac.pa/index.php/ciencia-agropecuaria/article/view/125 (accessed on 12 November 2022).
- Muratore, C.; Espen, L.; Prinsi, B. Nitrogen uptake in plants: The plasma membrane root transport systems from a physiological and proteomic perspective. Plants 2021, 10, 681. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Fan, X.; Miller, A.J.; Xu, G. Plant nitrogen uptake and assimilation: Regulation of cellular pH homeostasis. J. Exp. Bot. 2020, 71, 4380–4392. [Google Scholar] [CrossRef]
- Leghari, S.J.; Wahocho, N.A.; Laghari, G.M.; HafeezLaghari, A.; MustafaBhabhan, G.; HussainTalpur, K.; Bhutto, T.A.; Wahocho, S.A.; Lashari, A.A. Role of nitrogen for plant growth and development: A review. Adv. Environ. Biol. 2016, 10, 209–219. Available online: https://www.researchgate.net/publication/309704090_Role_of_Nitrogen_for_plant_Growth_and_Development_A_review#:~:text=All%20plants%20utilize%20nitrogen%20(N,and%20physiological%20functions%20of%20plant (accessed on 15 November 2021).
- Ragel, P.; Raddatz, N.; Leidi, E.O.; Quintero, F.J.; Pardo, J.M. Regulation of K+ Nutrition in Plants. Front. Plant Sci. 2019, 10, 281. [Google Scholar] [CrossRef]
- Fan, X.; Naz, M.; Fan, X.; Xuan, W.; Miller, A.J.; Xu, G. Plant nitrate transporters: From gene function to application. J. Exp. Bot. 2017, 68, 2463–2475. [Google Scholar] [CrossRef]
- Malhotra, H.; Vandana; Sharma, S.; Pandey, R. Phosphorus nutrition: Plant growth in response to deficiency and excess. In Plant Nutrients and Abiotic Stress Tolerance, 1st ed.; Hasanuzzaman, M., Fujita, M., Oku, H., Nahar, K., Hawrylak-Nowak, B., Eds.; Springer: Singapore, 2018; Volume 1, pp. 171–190. [Google Scholar] [CrossRef]
- Jacquot, A.; Chaput, V.; Mauries, A.; Li, Z.; Tillard, P.; Fizames, C.; Bonillo, P.; Bellegarde, F.; Laugier, E.; Santoni, V.; et al. NRT2.1 C-terminus phosphorylation prevents root high affinity nitrate uptake activity in Arabidopsis thaliana. New Phytol. 2020, 228, 1038–1054. [Google Scholar] [CrossRef]
- Gilliham, M.; Dayod, M.; Hocking, B.J.; Xu, B.; Conn, S.J.; Kaiser, B.N.; Leigh, R.A.; Tyerman, S.D. Calcium delivery and storage in plant leaves: Exploring the link with water flow. J. Exp. Bot. 2011, 62, 2233–2250. [Google Scholar] [CrossRef]
- González-Fontes, A.; Navarro-Gochicoa, M.T.; Ceacero, C.J.; Herrera-Rodríguez, M.B.; Camacho-Cristóbal, J.J.; Rexach, J. Understanding calcium transport and signaling, and its use efficiency in vascular plants. In Plant Macronutrient Use Efficiency, 1st ed.; Hossain, M.A., Kamiya, T., Burrit, D., Tran, L.-S.P., Fujiwara, T., Eds.; Elsevier: London, UK, 2017; Volume 1, pp. 165–180. [Google Scholar] [CrossRef]
- Ribeiro, L.R.; Leonel, S.; Azevedo, J.M.S.; García, E.L.; Leonel, M.; Honorato, M.L.N.; Silva, M.S.; Ferreira, R.B. Improving the nutritional value and extending shelf life of red guava by adding calcium chloride. LWT—Food Sci. Technol. 2020, 130, e109655. [Google Scholar] [CrossRef]
- Guo, W.; Nazim, H.; Liang, Z.; Yang, D. Magnesium deficiency in plants: An urgent problem. Crop J. 2016, 4, 83–91. [Google Scholar] [CrossRef]
- Farhat, N.; Elkhouni, A.; Zorrig, W.; Smaoui, A.; Abdelly, C.; Rabhi, M. Effects of magnesium deficiency on photosynthesis and carbohydrate partitioning. Acta Physiol. Plant. 2016, 38, 145. [Google Scholar] [CrossRef]
- Tang, R.-J.; Luan, S. Regulation of calcium and magnesium homeostasis in plants: From transporters to signaling network. Curr. Opin. Plant Biol. 2017, 39, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Yoshinari, A.; Takano, J. Insights into the mechanisms underlying boron homeostasis in plants. Front. Plant Sci. 2017, 8, 1951. [Google Scholar] [CrossRef]
- Karthika, K.S.; Rashmi, I.; Parvathi, M.S. Biological functions, uptake, and transport of essential nutrients in relation to plant growth. In Plant Nutrients and Abiotic Stress Tolerance, 1st ed.; Hasanuzzaman, M., Fujita, M., Oku, H., Nahar, K., Hawrylak-Nowak, B., Eds.; Springer: Singapore, 2018; Volume 1, pp. 1–49. [Google Scholar] [CrossRef]
- Krohling, C.A.; Eutrópio, F.J.; Azevedo, B.A.; Barros, D.L.; Campostrini, E.; Dias, T.; Coutinho, R.A. Ecophysiology of iron homeostasis in plants. Soil Sci. Plant Nutr. 2016, 62, 39–47. [Google Scholar] [CrossRef]
- Anjum, N.A.; Singh, H.P.; Khan, M.I.R.; Masood, A.; Per, T.S.; Negi, A.; Batish, D.R.; Khan, N.A.; Duarte, A.C.; Pereira, E.; et al. Too much is bad—An appraisal of phytotoxicity of elevated plant-beneficial heavy metal ions. Environ. Sci. Pollut. Res. 2015, 22, 3361–3382. [Google Scholar] [CrossRef] [PubMed]
- Connorton, J.M.; Balk, J.; Rodríguez-Celma, J. Iron homeostasis in plants—A brief overview. Metallomics 2017, 9, 813–823. [Google Scholar] [CrossRef]
- Rai, S.; Singh, P.K.; Mankotia, S.; Swain, J.; Satbhai, S.B. Iron homeostasis in plants and its crosstalk with copper, zinc, and manganese. Plant Stress 2021, 1, 100008. [Google Scholar] [CrossRef]
- Li, J.; Jia, Y.; Dong, R.; Huang, R.; Liu, P.; Li, X.; Wang, Z.; Liu, G.; Chen, Z. Advances in the mechanisms of plant tolerance to manganese toxicity. Int. J. Mol. Sci. 2019, 20, 5096. [Google Scholar] [CrossRef]
- Noulas, C.; Tziouvalekas, M.; Karyotis, T. Zinc in soils, water, and food crops. J. Trace Elem. Med. Biol. 2018, 49, 252–260. [Google Scholar] [CrossRef]
- Alejandro, S.; Höller, S.; Meier, B.; Peiter, E. Manganese in plants: From acquisition to subcellular allocation. Front. Plant Sci. 2020, 11, 300. [Google Scholar] [CrossRef]
- Hale, K.L.; McGrath, S.P.; Lombi, E.; Stack, S.M.; Terry, N.; Pickering, I.J.; George, G.N.; Pilon-Smits, E.A.H. Molybdenum sequestration in Brassica species. A role for anthocyanins? Plant Physiol. 2001, 126, 1391–1402. [Google Scholar] [CrossRef] [PubMed]
- Rana, M.S.; Bhantana, P.; Imran, M.; Saleem, M.H.; Moussa, M.G.; Khan, Z.; Khan, I.; Alam, M.D.; Abbas, M.; Binyamin, R.; et al. Molybdenum potential vital role in plants metabolism for optimizing the growth and development. Ann. Environ. Sci. Toxicol. 2020, 4, 32–44. [Google Scholar] [CrossRef]
- Stefanov, M.; Yotsova, E.; Rashkov, G.; Ivanova, K.; Markovska, Y.; Apostolova, E.L. Effects of salinity on the photosynthetic apparatus of two Paulownia lines. Plant Physiol. Biochem. 2016, 101, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Muchate, N.S.; Nikalje, G.C.; Rajurkar, N.S.; Suprasanna, P.; Nikam, T.D. Plant salt stress: Adaptive responses, tolerance mechanism and bioengineering for salt tolerance. Bot. Rev. 2016, 82, 371–406. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; Zhao, H.; Guo, D.; He, L.; Liu, F.; Zhou, Q.; Nandwani, D.; Hui, D.; Yu, J. Electrical conductivity of nutrient solution influenced photosynthesis, quality, and antioxidant enzyme activity of pakchoi (Brassica campestris L. ssp. Chinensis) in a hydroponic system. PLoS ONE 2018, 13, e0202090. [Google Scholar] [CrossRef]
- Gandica, O.H.; Peña, H. Acumulación de materia seca y balance de nutrientes en tomate (Solanum lycopersicum L.) cultivado en ambiente protegido. Bioagro 2015, 27, 111–120. Available online: https://ve.scielo.org/pdf/ba/v27n2/art07.pdf (accessed on 8 March 2022).
- Pérez-Labrada, F.; Benavides-Mendoza, A.; Valdez-Aguilar, L.A.; Robledo-Torres, V. Citric acid in the nutrient solution increases the mineral absorption in potted tomato grown in calcareous soil. Pak. J. Bot. 2016, 48, 67–74. Available online: https://www.researchgate.net/publication/295086648_Citric_acid_in_the_nutrient_solution_increases_the_mineral_absorption_in_potted_tomato_grown_in_calcareous_soil (accessed on 15 July 2022).
- Steiner, A.A. The universal nutrient solution. In Proceedings of the Sixth International Congress on Soilless Culture, ISOSC, Lunteren, The Netherlands, 29 April–5 May 1984; pp. 633–650. [Google Scholar]
- Horneck, D.A.; Miller, R.O. Determination of total nitrogen in plant tissue. In Handbook of Reference Methods for Plant Analysis, 1st ed.; Kalra, Y.P., Ed.; Taylor & Francis Group: New York, NY, USA, 1997; pp. 75–83. Available online: https://scholar.google.com/scholar_lookup?hl=en&publication_year=1998&author=DA+Horneck&author=RO+Miller%26&title=Determination+of+total+nitrogen+in+plant+tissue (accessed on 21 February 2025).
- SAS Institute. User’s Guide Version 9.2; SAS Institute Inc.: Cary, NC, USA, 2008. [Google Scholar]



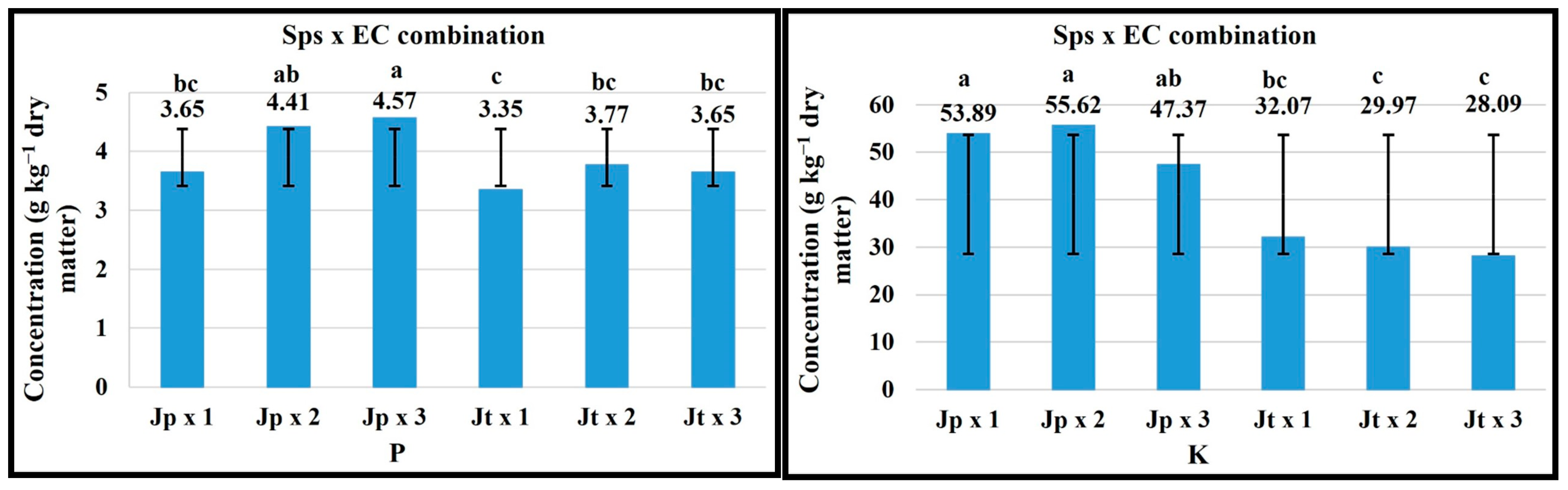
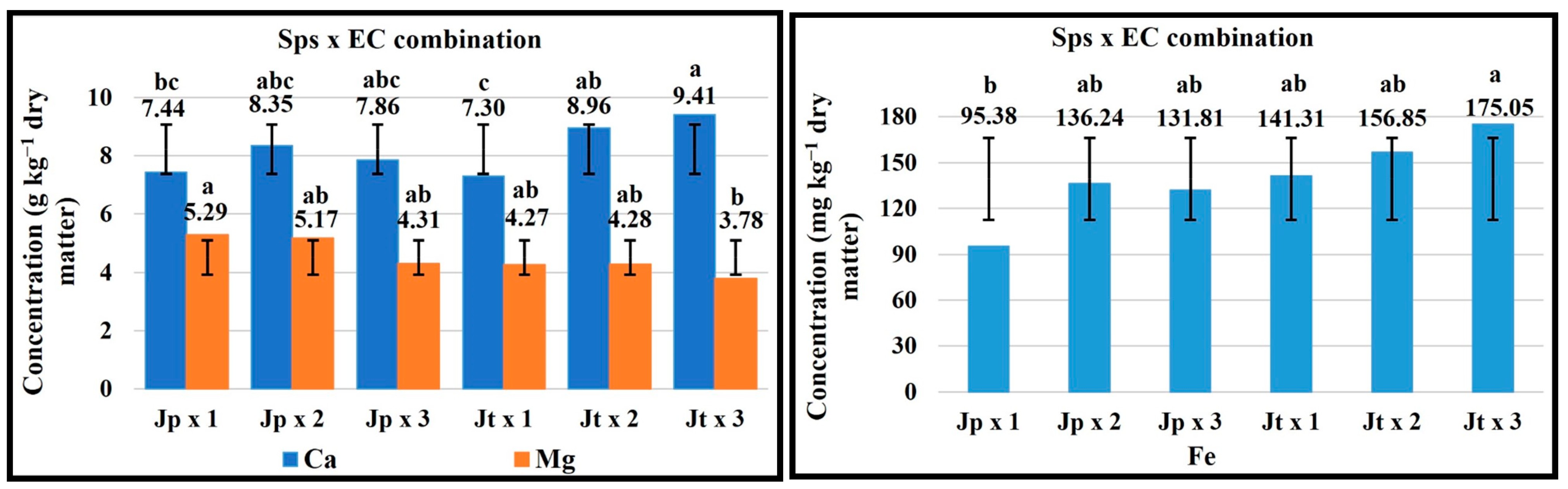
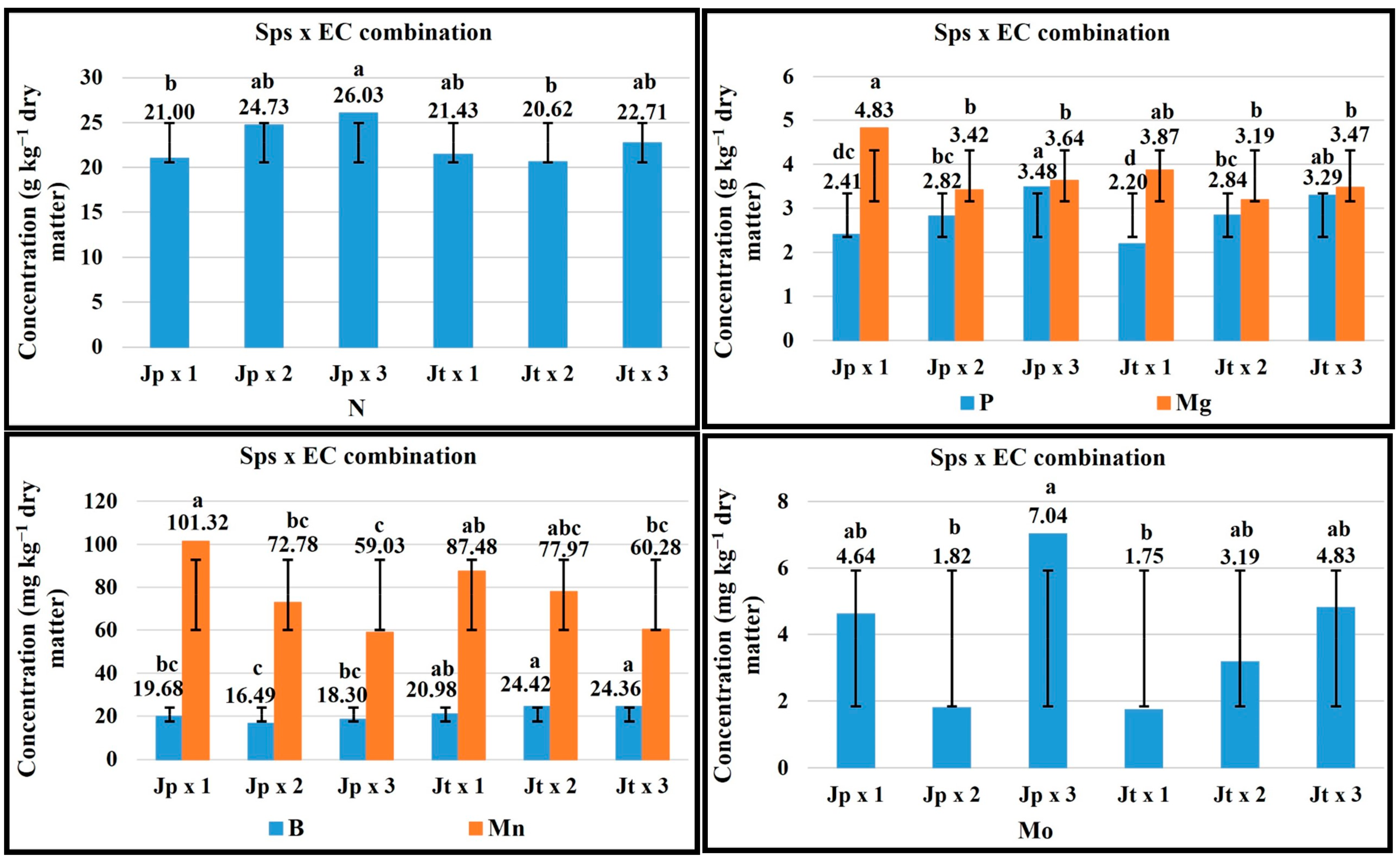
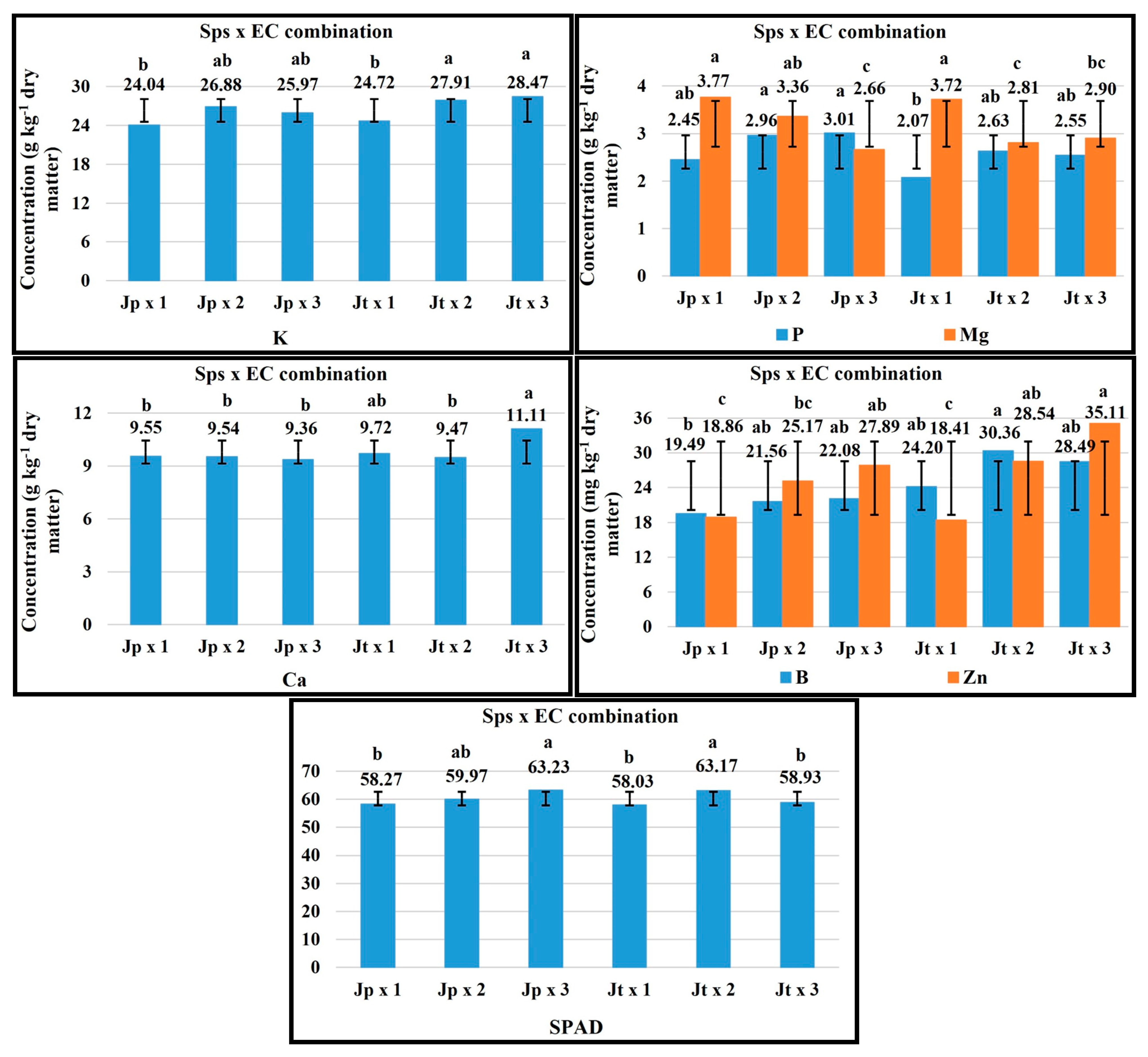
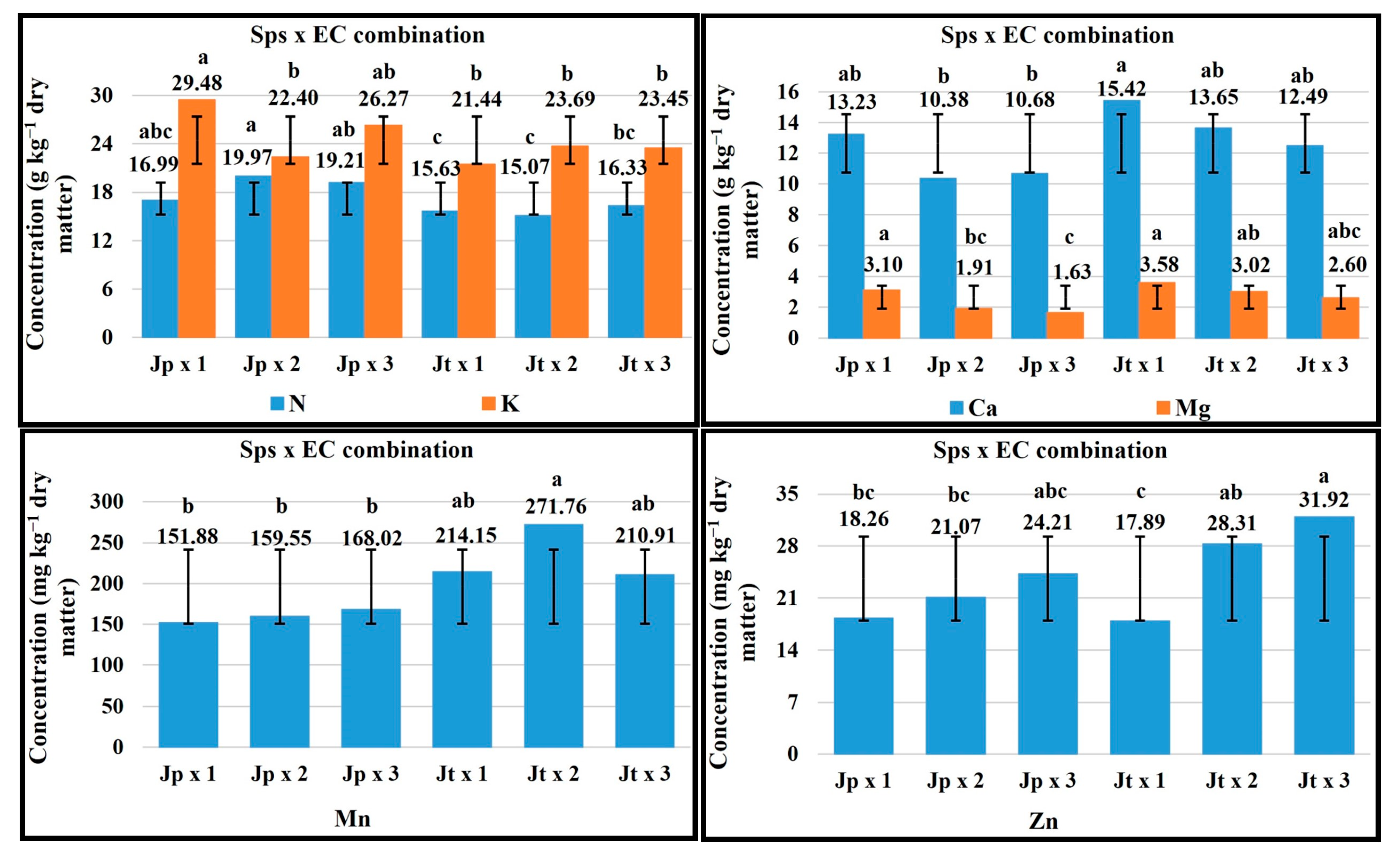
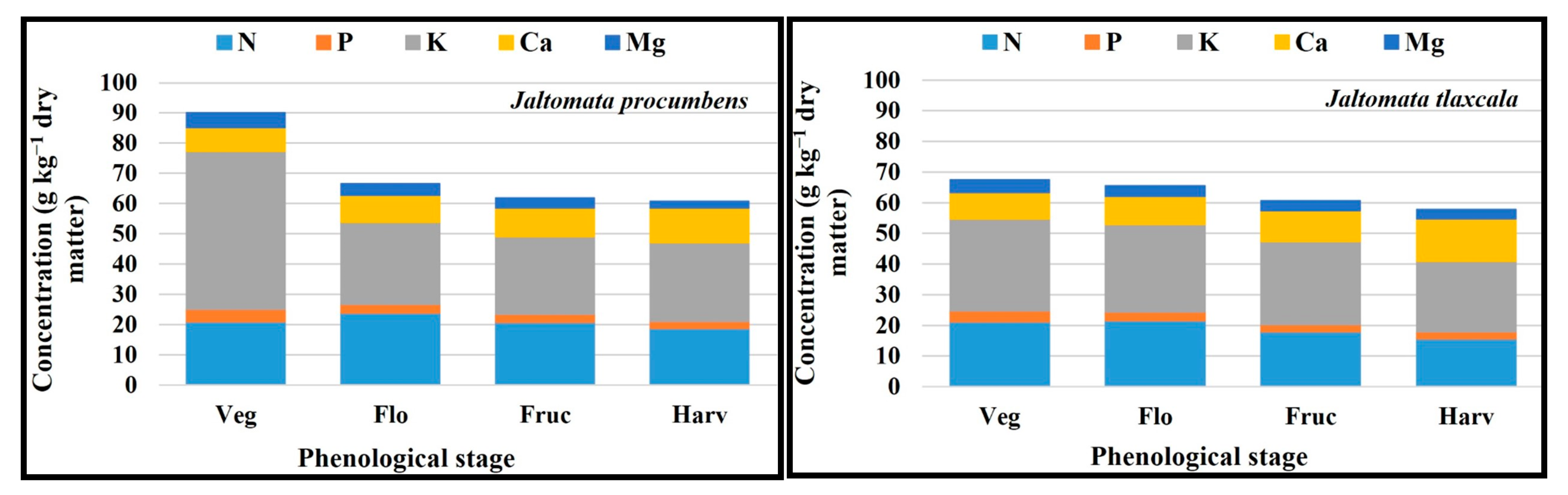

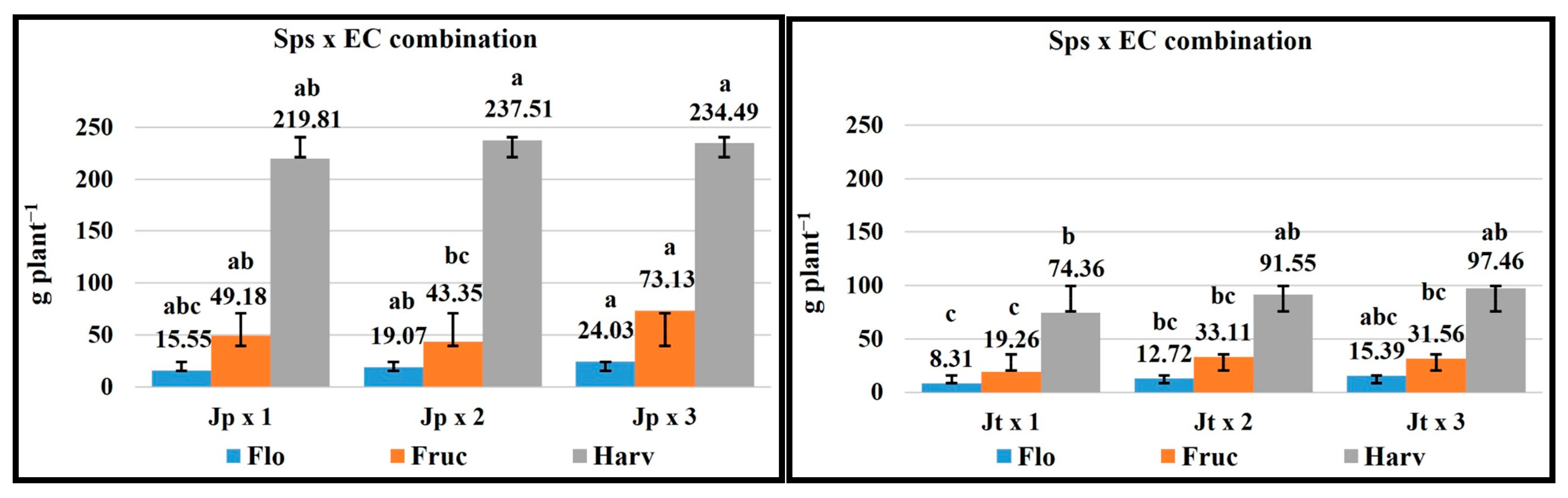
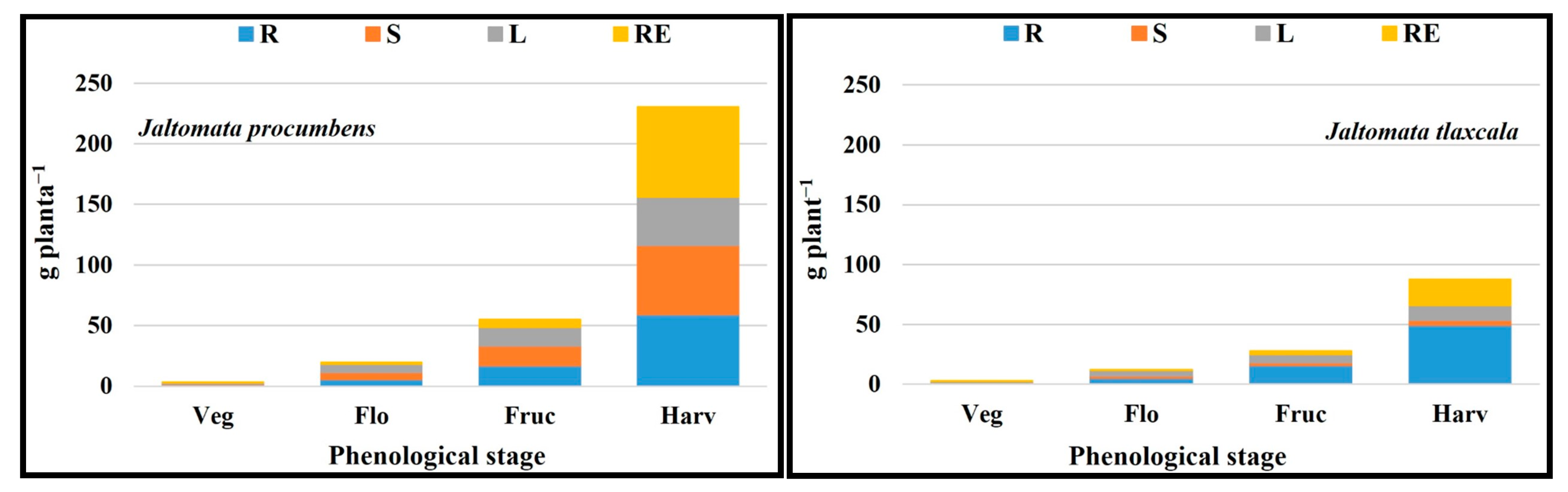
| FV | Macronutrients (g kg−1 Dry Matter) | Micronutrients (mg kg−1 Dry Matter) | ||||||
|---|---|---|---|---|---|---|---|---|
| EC (dS m−1) | Vegetative Stage (17 dat) | Vegetative Stage (17 dat) | ||||||
| N | P | Ca | Mg | Fe | Mo | |||
| 1 | 17.8 b | 3.5 b | 7.4 b | 4.8 a | 118.3 b | 1.9 b | ||
| 2 | 21.6 ab | 4.1 a | 8.7 a | 4.7 a | 146.6 ab | 4.1 a | ||
| 3 | 23.2 a | 4.1 a | 8.6 a | 4.0 b | 153.4 a | 5.2 a | ||
| Flowering stage (31 dat) | Flowering stage (31 dat) | |||||||
| N | P | Mg | Mn | Mo | Zn | |||
| 1 | 21.2 b | 2.3 c | 4.3 a | 94.4 a | 3.2 b | 24.2 b | ||
| 2 | 22.6 ab | 2.8 b | 3.3 b | 75.4 b | 2.5 b | 25.8 ab | ||
| 3 | 24.3 a | 3.4 a | 3.6 b | 59.7 c | 5.9 a | 30.0 a | ||
| Fructification stage (45 dat) | Fructification stage (45 dat) | |||||||
| P | K | Mg | Mn | Mo | Zn | |||
| 1 | 2.3 b | 24.4 b | 3.7 a | 118.3 a | 1.6 b | 18.6 c | ||
| 2 | 2.8 a | 27.4 a | 3.1 b | 112.0 ab | 4.8 a | 26.9 b | ||
| 3 | 2.8 a | 27.2 a | 2.8 b | 94.7 b | 4.3 a | 31.5 a | ||
| Harvesting stage (57 dat) | Harvesting stage (57 dat) | |||||||
| Ca | Mg | Zn | ||||||
| 1 | 14.3 a | 3.3 a | 18.1 b | |||||
| 2 | 12.0 b | 2.5 b | 24.7 a | |||||
| 3 | 11.6 b | 2.1 b | 28.1 a | |||||
| FV | Total Dry Matter (g plant−1) | |||
|---|---|---|---|---|
| Species | Vegetative | Flowering | Fructification | Harvesting |
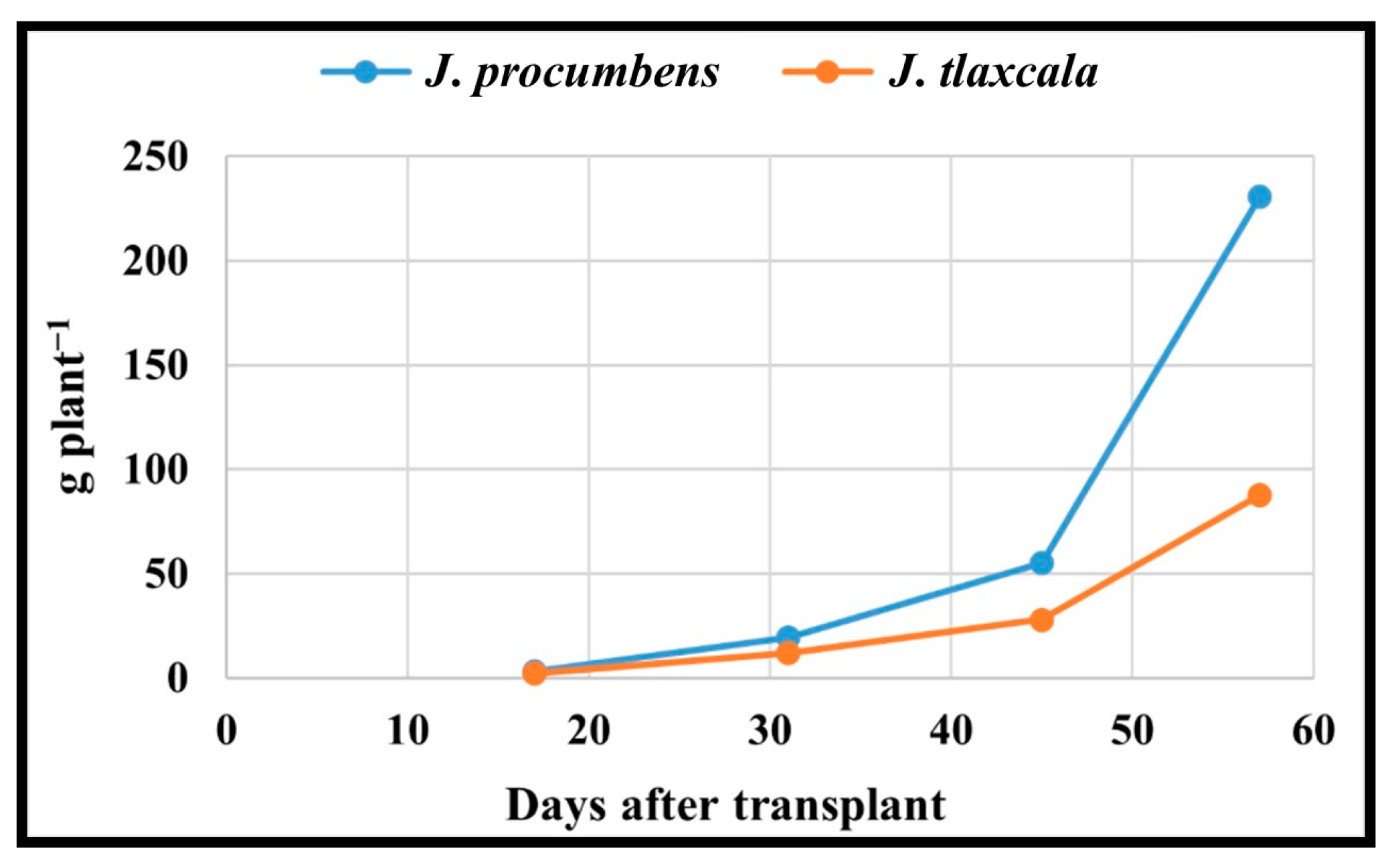
| Jaltomata procumbens | 3.1 a | 19.6 a | 55.2 a | 230.6 a |
| Jaltomata tlaxcala | 2.3 b | 12.1 b | 28.0 b | 87.8 b |
| EC (dS m−1) | ||||
| 1 | 2.4 a | 11.9 b | 34.2 a | 147.1 a |
| 2 | 2.7 a | 15.9 ab | 38.2 a | 164.5 a |
| 3 | 3.0 a | 19.7 a | 52.4 a | 165.9 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Sánchez, I.D.; Sandoval-Villa, M.; Uscanga-Mortera, E. Nutrient Uptake of Two Semidomesticated Jaltomata Schltdl. Species for Their Cultivation. Plants 2025, 14, 1124. https://doi.org/10.3390/plants14071124
Flores-Sánchez ID, Sandoval-Villa M, Uscanga-Mortera E. Nutrient Uptake of Two Semidomesticated Jaltomata Schltdl. Species for Their Cultivation. Plants. 2025; 14(7):1124. https://doi.org/10.3390/plants14071124
Chicago/Turabian StyleFlores-Sánchez, Ignacio Darío, Manuel Sandoval-Villa, and Ebandro Uscanga-Mortera. 2025. "Nutrient Uptake of Two Semidomesticated Jaltomata Schltdl. Species for Their Cultivation" Plants 14, no. 7: 1124. https://doi.org/10.3390/plants14071124
APA StyleFlores-Sánchez, I. D., Sandoval-Villa, M., & Uscanga-Mortera, E. (2025). Nutrient Uptake of Two Semidomesticated Jaltomata Schltdl. Species for Their Cultivation. Plants, 14(7), 1124. https://doi.org/10.3390/plants14071124







