Journal Description
Catalysts
Catalysts
is a peer-reviewed open access journal of catalysts and catalyzed reactions published monthly online by MDPI. The Romanian Catalysis Society (RCS) are partners of Catalysts journal and its members receive a discount on the article processing charge.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), Inspec, CAPlus / SciFinder, CAB Abstracts, and other databases.
- Journal Rank: JCR - Q2 (Chemistry, Physical) / CiteScore - Q1 (General Environmental Science )
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 13.9 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2024).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
3.8 (2023);
5-Year Impact Factor:
3.9 (2023)
Latest Articles
Computational Search for a Novel Effective Ligand for Ni-Catalyzed Asymmetric Hydrogenation
Catalysts 2025, 15(4), 352; https://doi.org/10.3390/catal15040352 (registering DOI) - 3 Apr 2025
Abstract
Using the DFT method, an analogue of R,R-t-Bu-BenzP* was tried as a potential ligand for Ni-catalyzed asymmetric hydrogenation. This ligand contains benzyl groups instead of the t-Bu groups in R,R-t-Bu-BenzP*. Computational results imply that the R
[...] Read more.
Using the DFT method, an analogue of R,R-t-Bu-BenzP* was tried as a potential ligand for Ni-catalyzed asymmetric hydrogenation. This ligand contains benzyl groups instead of the t-Bu groups in R,R-t-Bu-BenzP*. Computational results imply that the R,R-Benz-BenzP* ligand (1) is expected to provide excellent enantioselectivity in the Ni-catalyzed asymmetric hydrogenation of 1-phenylethanone oxime. The computed effectiveness of the R,R-Benz-BenzP* ligand is stipulated by its conformational flexibility, which helps stabilize the crucial transition states via a non-bonding interaction between the substrate and the catalyst. R,R-Benz-BenzP* ligands with CN- and OMe-substituted benzyl rings were also computed to possess the same effectiveness.
Full article
(This article belongs to the Section Computational Catalysis)
Open AccessArticle
Experimental Design and Numerical Optimization of Photochemical Oxidation Removal of Tetracycline from Water Using Fe3O4-Supported Fruit Waste Activated Carbon
by
Manasik M. Nour, Maha A. Tony, Hossam A. Nabwey and Shaaban M. Shaaban
Catalysts 2025, 15(4), 351; https://doi.org/10.3390/catal15040351 (registering DOI) - 3 Apr 2025
Abstract
The ever-increasing importance of sustainable environmental remediation calls for academics’ contribution to satisfy such a need. The 3R’s criteria of recover, recycle and reuse is designed to sustain the waste stream to produce a valuable product. In this regard, the circular economy looks
[...] Read more.
The ever-increasing importance of sustainable environmental remediation calls for academics’ contribution to satisfy such a need. The 3R’s criteria of recover, recycle and reuse is designed to sustain the waste stream to produce a valuable product. In this regard, the circular economy looks to deliver banana peel waste as a photocatalyst for pharmaceutical effluent oxidation, which we investigated in this study. Banana peel waste is treated thermally and chemically then augmented with magnetite nanoparticles and labeled as ACBP-Fe3O4. The mixture is characterized through Scanning Electron Microscopy (SEM) and the composition of the composite material is attained by energy dispersive X-ray spectroscopy (EDX), and then introduced as a Fenton catalyst. The notable oxidation of tetracycline (TC), evaluated by TC removal and chemical Oxygen Demand (COD) oxidation tenancy, is achieved. The effectiveness of the operational parameters is also assessed and the most influenced parameters are optimized through numerical optimization based on a Response Surface Methodology (RSM) tool. The effects of initial pH value, ACBP-Fe3O4 and H2O2 concentrations on the oxidation efficiency of the Tetracycline were optimized at pH 6.6 and 350 mg/L and 43 g/L for H2O2 and ACBP-Fe3O4, respectively. Thermodynamics and kinetics were also studied and the experimental and model data revealed the reaction is spontaneous and exothermic in nature and follows the first-order reaction kinetics. Also, the thermodynamic results the reaction proceeds at a low energy barrier of 34.33 kJ mol−1. Such a system introduces the role of engineers and academics for a sustainable world without a waste stream.
Full article
(This article belongs to the Special Issue Remediation of Natural Waters by Photocatalysis)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Fabricating Oxygen Vacancy-Rich Bi2WO6/Bi2S3 Z-Scheme Nano-Heterojunction on Carbon Fiber with Polydopamine for Enhanced Photocatalytic Activity
by
Jiantao Niu, Jiaqi Pan, Jianfeng Qiu and Chaorong Li
Catalysts 2025, 15(4), 350; https://doi.org/10.3390/catal15040350 - 2 Apr 2025
Abstract
The use of fibers or fabrics as frameworks for loading photocatalysts is beneficial in solving the problems of photocatalytic nanomaterials, which tend to agglomerate and are difficult to recycle. In this study, Bi2WO6/CFb and Bi2WO6/Bi
[...] Read more.
The use of fibers or fabrics as frameworks for loading photocatalysts is beneficial in solving the problems of photocatalytic nanomaterials, which tend to agglomerate and are difficult to recycle. In this study, Bi2WO6/CFb and Bi2WO6/Bi2S3/CFb photocatalytic fibers rich in oxygen vacancies were prepared using carbon fibers as the framework by the crystal seed attachment method and in situ growth method by using the self-polymerization and strong adhesion properties of dopamine. The results of SEM, TEM and XRD tests showed that Bi2WO6 and Bi2WO6/Bi2S3 nanosheets were uniformly and completely encapsulated on the surface of the carbon fibers. The results of XPS and EPR tests showed that Bi2WO6 nanosheets were rich in oxygen vacancies. The PL, transient photocurrent responses and EIS results showed that the introduction of Bi2S3 significantly improved the migration efficiency of the photogenerated carriers of Bi2WO6/Bi2S3/CFb, which effectively hindered the recombination of photogenerated electron–hole pairs. By conducting degradation experiments on p-nitrophenol and analyzing the bandgap structure, it was postulated that the heterojunction structure of Bi2WO6/Bi2S3/CFb in the Bi2WO6/Bi2S3 material was not Type-II but Z-scheme. As analyzed by the active species assay, the active species that played a major role in the degradation process were O2− and h+. The incorporation of a small amount of Bi2S3 resulted in enhanced photocatalytic degradation activity of Bi2WO6/Bi2S3/CFb toward tetracycline hydrochloride compared to Bi2WO6/CFb. The excellent photocatalytic performance of Bi2WO6/Bi2S3/CFb photocatalytic fibers can be attributed to the rapid transmission and separation performance and the high oxidation and reduction capacities of photogenerated electron–hole pairs formed by direct Z-scheme heterojunctions.
Full article
(This article belongs to the Section Catalytic Materials)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Synthesis of Black g-C3N4 and Exploration of the Mechanism Underlying the Enhancement of Photocatalytic CO2 Reduction
by
Shaokun Lv, Jun Zhang, Xiaoke Chen, Yue Zou, Qiuli Chen, Yongsheng Yan and Pengxin Li
Catalysts 2025, 15(4), 349; https://doi.org/10.3390/catal15040349 - 2 Apr 2025
Abstract
The use of solar energy to convert CO2 into value-added chemicals is a promising sustainable development strategy. In this study, a black graphitic carbon nitride (CN-B) photocatalyst was fabricated through a single-step calcination process, employing phloxine B and urea as the precursor
[...] Read more.
The use of solar energy to convert CO2 into value-added chemicals is a promising sustainable development strategy. In this study, a black graphitic carbon nitride (CN-B) photocatalyst was fabricated through a single-step calcination process, employing phloxine B and urea as the precursor materials. The catalysts were characterized using TEM, XRD, FTIR, XPS and so on. The amount of prepolymer phloxine B was 25 mg, 35 mg and 45 mg, respectively, and the obtained samples were CN-B-0.025, CN-B-0.035 and CN-B-0.045. All samples were used for visible-catalyzed CO2 reduction. The experimental findings indicate that the CO evolution rate of the optimal photocatalyst CN-B-0.035 reaches 27.56 μmol gcat.−1 h−1. This value is nine-fold higher than that of pure CN, which has a CO evolution rate of 3.22 μmol gcat.−1 h−1. The excellent photocatalytic reduction performance is due to the following factors: Firstly, the exceedingly thin nanosheet structure of the catalyst enhances the velocity of the charge transfer, and transmission electron microscopy (TEM) analysis shows that the nanosheet thickness of the catalyst CN-B is significantly thinner. Secondly, the light absorption capacity of the catalyst is enhanced. The absorbance of CN-B increases significantly in the ultraviolet region and extends to the near-infrared region, as shown with UV diffuse reflection spectroscopy. Finally, the photothermal effect of CN-B causes the catalyst temperature to rise rapidly from 20 °C to 131 °C within 120 s, which further promotes photogenerated carrier separation. This research offers a novel approach to the development of photocatalysts aimed at the photothermal-assisted photocatalytic conversion of CO2.
Full article
(This article belongs to the Special Issue Mineral-Based Composite Catalytic Materials)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Selective Conversion of Glycerol to Acetol: Effect of the Preparation Method of CuAl Catalysts and Reaction Phase
by
Francisco Maldonado-Martín, Lucía García, Joaquín Ruiz, Miriam Oliva and Jesús Arauzo
Catalysts 2025, 15(4), 348; https://doi.org/10.3390/catal15040348 - 2 Apr 2025
Abstract
A group of CuAl catalysts were synthesized with a Cu/Al molar ratio of 1:1 using different preparation methods: coprecipitation, surfactant assisted coprecipitation, polymeric precursor, and self-combustion and then screened for the selective dehydration of glycerol to acetol. The catalysts were employed in glycerol
[...] Read more.
A group of CuAl catalysts were synthesized with a Cu/Al molar ratio of 1:1 using different preparation methods: coprecipitation, surfactant assisted coprecipitation, polymeric precursor, and self-combustion and then screened for the selective dehydration of glycerol to acetol. The catalysts were employed in glycerol conversion at the same temperature (227 °C) in two different laboratory-scale systems, the first one at atmospheric pressure (gas phase) and the second one in a pressurized system at 34 absolute bar (liquid phase). The preparation method of the CuAl catalysts influenced the carbon yield to liquids and acetol selectivity. However, the reaction phase had a greater influence than the preparation method of the catalyst. In the gas phase, the carbon yield to liquids reached values above 40% and the carbon selectivity to acetol was higher than 90%. The highest acetol yield, 462.6 mgacetol/gglycerol, was obtained with the CuAl catalyst prepared by the surfactant-assisted coprecipitation method. This study provides a new perspective on catalyst design by highlighting the crucial role of preparation techniques in determining CuAl catalyst performance in the liquid and gas phases.
Full article
(This article belongs to the Special Issue Topical Advisory Panel Members' Collection Series: Biomass Catalytic Conversion)
►▼
Show Figures
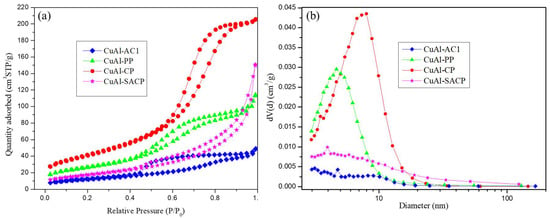
Figure 1
Open AccessArticle
Correlation Between the Apparent Rate Constant and the Dye Concentration—Effect of the Bottom Reflection Ability on the Photocatalytic Reaction Rate
by
Emil Lilov, Svetlozar Nedev, Vanya Lilova, Christian Girginov and Stephan Kozhukharov
Catalysts 2025, 15(4), 347; https://doi.org/10.3390/catal15040347 - 2 Apr 2025
Abstract
The dependence of the reaction rate on the solution layer thickness discovered in a previous work could be a powerful tool for investigating photocatalytic reactions. A reduction of the apparent rate constant with the growth of the dye concentration was found using this
[...] Read more.
The dependence of the reaction rate on the solution layer thickness discovered in a previous work could be a powerful tool for investigating photocatalytic reactions. A reduction of the apparent rate constant with the growth of the dye concentration was found using this dependence. This decrement follows a hyperbolic law. This dependence is explained based on the observed increment of the solution conductivity. In addition, it is confirmed experimentally that the reaction rate decreases with the solution depth growth. The possibility of independent determination of the reaction rate constant and the adsorption equilibrium constant has been discussed. Additionally, it is demonstrated that the vessel’s reflective bottom could increase the chemical reaction rate. The reason why other authors have not yet reported this effect is also discussed.
Full article
(This article belongs to the Section Photocatalysis)
►▼
Show Figures

Figure 1
Open AccessArticle
Frequency-Resolved Modulation Excitation Spectroscopy Methodology for Identifying Surface Reaction Species in Ethanol Oxidation on Gold Catalysts
by
Bhagyesha S. Patil, Alejandra Torres-Velasco and Juan J. Bravo-Suárez
Catalysts 2025, 15(4), 346; https://doi.org/10.3390/catal15040346 - 1 Apr 2025
Abstract
This study used in situ modulation excitation spectroscopy (MES) with varying frequencies in a single experiment to identify surface species during ethanol oxidation on Au/SiO2, Au/TiO2, Au/ZnO, and Au/SrTiO3. Fixed-bed reactor (FBR) tests (1 kPa ethanol, 1.5
[...] Read more.
This study used in situ modulation excitation spectroscopy (MES) with varying frequencies in a single experiment to identify surface species during ethanol oxidation on Au/SiO2, Au/TiO2, Au/ZnO, and Au/SrTiO3. Fixed-bed reactor (FBR) tests (1 kPa ethanol, 1.5 kPa O2, 513 K) showed that Au/SiO2 and Au/SrTiO3 had higher ethanol conversions. Au/SiO2 favored acetaldehyde, while Au/SrTiO3 yielded more acetates (acetic acid and ethyl acetate). Operando modulation excitation (ME)–phase sensitive detection (PSD)–DRIFTS, with ethanol and oxygen modulation, identified surface ethanol, acetaldehyde, acetates, ethoxy, and hydroxyl species. Oxygen modulation showed charge transfer to supports in Au/TiO2 and Au/ZnO. At the fundamental frequency (f0 = 1/90 Hz), ME–PSD–DRIFTS showed minimal adsorbed ethanol on Au/SiO2, indicating high ethanol conversion. Au/SrTiO3 had higher acetaldehyde consumption, correlating with increased acetates, consistent with FBR results. ME–PSD–DRIFTS at lower frequencies (0.07f0, 0.5 f0) and higher harmonics (2f0, 3f0) showed rapid ethoxy formation/decomposition, and slower acetaldehyde reactions, confirming acetaldehyde as a primary product and acetates as secondary products. Oxygen modulation revealed rapid hydrogen spillover and hydroxyl changes. Overall, operando spectroscopy via mass spectrometry confirmed the FBR findings.
Full article
(This article belongs to the Special Issue Spectroscopy in Modern Materials Science and Catalysis)
►▼
Show Figures
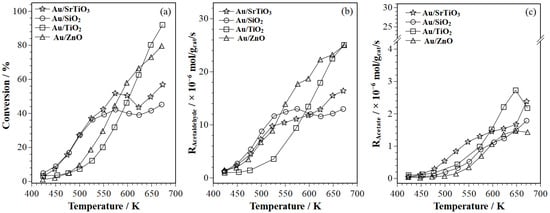
Figure 1
Open AccessArticle
Regulating the Structures of Carbon Cloth and Carbon Nanotubes to Boost the Positive Electrode Reaction of Vanadium Redox Flow Batteries
by
Xinyu Huang, Chuanyu Sun, Shuqi Liu, Bangsen Zhao, Mingming Ge and Huan Zhang
Catalysts 2025, 15(4), 345; https://doi.org/10.3390/catal15040345 - 1 Apr 2025
Abstract
Considering the various morphologies of carbon nanotubes (CNTs), it is expected to solve the contradiction between concentration polarization and electrochemical polarization in vanadium redox flow batteries (VRFBs). This paper investigates the structural evolution of CNTs grown on the surface of thermally oxidized carbon
[...] Read more.
Considering the various morphologies of carbon nanotubes (CNTs), it is expected to solve the contradiction between concentration polarization and electrochemical polarization in vanadium redox flow batteries (VRFBs). This paper investigates the structural evolution of CNTs grown on the surface of thermally oxidized carbon cloth (TCC) and their impact on the performance of VRFBs. The morphological results indicate that thermal oxidation treatment forms pores on the surface of the TCC, providing nucleation sites for CNT growth. Spiral-shaped CNTs (TCC@s-CNTs) were formed in a short growth time (1 h), and their high defect density originated from the non-steady-state supply of carbon sources and the dynamic behavior of the catalyst. While 3 h of growth forms a network structure (TCC@n-CNT), the van der Waals force drives the self-assembly of its three-dimensional network. Although the TCC@s-CNT exhibits high catalytic activity due to its high defect density and edge active sites, the performance of VRFBs is more dependent on the three-dimensional conductive network of the TCC@n-CNT. At 240 mA/cm2, the energy efficiency (EE) of a VRFB assembled with the TCC@n-CNT reaches 71%, and the capacity retention rate is 15% higher than that of the TCC@s-CNT. This work reveals the synergistic mechanism of CNT morphology regulation on electrode performance and provides theoretical guidance for the design of VRFB electrodes.
Full article
(This article belongs to the Section Catalysis for Sustainable Energy)
►▼
Show Figures

Figure 1
Open AccessFeature PaperArticle
Nitrone or Oxaziridine? Further Insights into the Selectivity of Imine Oxidation Catalyzed by Methyltrioxorhenium
by
Camilla Matassini, Marco Bonanni, Francesca Cardona and Andrea Goti
Catalysts 2025, 15(4), 344; https://doi.org/10.3390/catal15040344 - 1 Apr 2025
Abstract
The oxidation of imines may give several products, such as oxaziridines, nitrones, amides, and other rearranged compounds. Therefore, its selectivity is a challenge that various methods have to face. The controversial selectivity of the oxidation of imines using urea hydrogen peroxide (UHP) catalyzed
[...] Read more.
The oxidation of imines may give several products, such as oxaziridines, nitrones, amides, and other rearranged compounds. Therefore, its selectivity is a challenge that various methods have to face. The controversial selectivity of the oxidation of imines using urea hydrogen peroxide (UHP) catalyzed by methyltrioxorhenium (MTO) is addressed by varying the solvent, temperature, reaction time, amount of oxidant, and catalyst used. The reactivity and selectivity of the oxidation of imines proved to be particularly sensitive to the type of solvent. The use of methanol furnished the corresponding nitrones as the exclusive products, except for very hindered N-tert-alkyl substituted substrates. Using the ionic liquid [bmim]BF4 as a solvent resulted in a complete switch in reactivity and selectivity. N-methyl substituted imines gave the corresponding amides, while imines with bulkier substituents at nitrogen did not show any reactivity. An exception was the C-phenyl,N-tert-butyl imine—the only substrate that was oxidized to the corresponding oxaziridine, albeit with low conversion. The results reported herein reaffirm the oxidation of imines with UHP/MTO in MeOH as the method of choice for their interconversion to nitrones.
Full article
(This article belongs to the Section Catalysis in Organic and Polymer Chemistry)
►▼
Show Figures

Graphical abstract
Open AccessFeature PaperArticle
ZIF-67-Derived Co−N−C Supported Ni Nanoparticles as Efficient Recyclable Catalyst for Hydrogenation of 4-Nitrophenol
by
Juti Rani Deka, Diganta Saikia, Jia-Cheng Lin, Wan-Yu Chen, Hsien-Ming Kao and Yung-Chin Yang
Catalysts 2025, 15(4), 343; https://doi.org/10.3390/catal15040343 - 1 Apr 2025
Abstract
In this study, a novel, highly efficient, environment friendly, and low-cost nanocatalyst, denoted as Ni(x)@Co−N−C, was successfully developed by encapsulating Ni nanoparticles into N-doped porous carbon derived from ZIF-67. A variety of techniques including powder X-ray diffraction (XRD), nitrogen adsorption/desorption, scanning electron microscopy
[...] Read more.
In this study, a novel, highly efficient, environment friendly, and low-cost nanocatalyst, denoted as Ni(x)@Co−N−C, was successfully developed by encapsulating Ni nanoparticles into N-doped porous carbon derived from ZIF-67. A variety of techniques including powder X-ray diffraction (XRD), nitrogen adsorption/desorption, scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray photoelectron spectrometer (XPS) were used to characterize the prepared materials. The TEM images reveal that the nanoparticles were distributed homogeneously in the carbon support. The N atoms in the carbon support serve as the sites for the nucleation and uniform growth of Ni nanoparticles. The catalyst was used for the degradation of environmentally harmful 4-nitrophenol (4-NP) to 4-aminophenol (4-AP). Among all the catalysts investigated, Ni(10)@Co-N-C exhibited the highest catalytic activity for the hydrogenation of 4-NP, with a specific reaction rate of 6.1 × 10−3 s−1, activity parameter of 31 s−1g−1, and turn over frequency (TOF) of 1.78 × 1019 molecules gmetal−1s−1. On the other hand, the specific reaction rate and TOF value were 1.7 × 10−3 s−1 and 6.96 × 1018 molecules gmetal−1s−1, respectively, for Co−N−C. This suggests that Ni(10)@Co−N−C is about three times more catalytically active than the Co−N−C catalyst. The superb activity of Ni(10)@Co−N−C in comparison to Co−N−C can be ascribed to the homogeneous dispersion of small-sized Ni nanoparticles, the interconnected three-dimensional porous arrangement of the support Co−N−C, the presence of N atoms in the carbon framework that stabilize metal nanoparticles, and the synergistic electronic effect between Ni and Co. The Ni(10)@Co−N−C catalyst maintained consistent catalytic activity over multiple cycles, which suggests that porous N-containing carbon support can effectively prevent aggregation and leaching of metal nanoparticles. The ICP-AES analysis of the recycled Ni(10)@Co−N−C revealed a slight reduction in metal content compared to the fresh sample, suggesting almost negligible leaching of metal nanoparticles.
Full article
(This article belongs to the Special Issue Nanocatalysts for Degradation of Environmental Pollutants and Hydrogen Generation)
►▼
Show Figures

Graphical abstract
Open AccessFeature PaperArticle
Enhanced Nitrate Production via Electrocatalytic Nitric Oxide Oxidation Reaction over MnO2 with Different Crystal Facets
by
Xiaoyu Qin, Dongcai Shen, Quan Li, Xin Liu, Mingrui Wu and Wentai Wang
Catalysts 2025, 15(4), 342; https://doi.org/10.3390/catal15040342 - 1 Apr 2025
Abstract
The synthesis of nitrate (NO3−) via electrocatalytic nitric oxide oxidation reaction (NOOR) is a green and efficient strategy for nitrogen fixation, which has great advantages over conventional nitrate synthesis. Notably, it also presents a promising solution for the remediation of
[...] Read more.
The synthesis of nitrate (NO3−) via electrocatalytic nitric oxide oxidation reaction (NOOR) is a green and efficient strategy for nitrogen fixation, which has great advantages over conventional nitrate synthesis. Notably, it also presents a promising solution for the remediation of NO pollutants. In this study, the structure–performance correlations of α-, β-, and δ-MnO2 catalysts were investigated. These three polymorphs of MnO2 exhibited disparate surface chemistries, NO adsorption capabilities, and NOOR catalytic activities. In a 1.0 M KOH electrolyte, α-MnO2, characterized by its large-sized (2 × 2) one-dimensional tunnel structure, demonstrated the most outstanding NOOR catalytic performance, which achieved a remarkable NO3− yield of 665.2 mg·h−1·mgcat−1 at a potential of 1.9 V, along with excellent stability and durability. Furthermore, a Zn-NO system was constructed, employing α-MnO2 as the anode and a Zn plate as the cathode. This innovative setup integrated an energy storage system with NO electrochemical capture, yielding a NO3− production rate of 265.5 mg·h−1·mgcat−1. The density functional theory calculations confirm the NO adsorption performance and catalytic activity of three different crystal forms of MnO2.
Full article
(This article belongs to the Section Electrocatalysis)
►▼
Show Figures
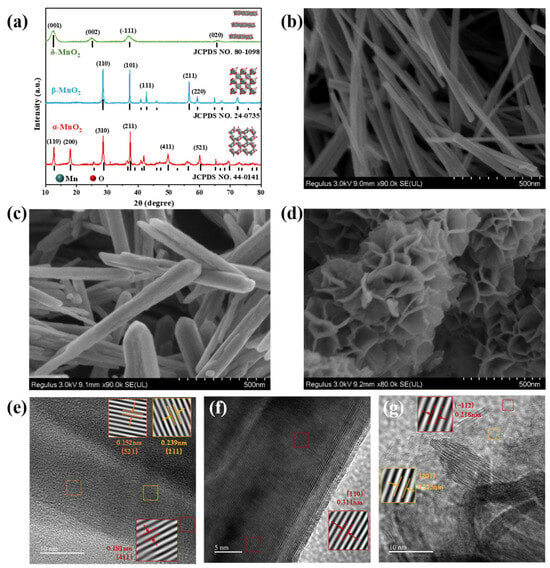
Figure 1
Open AccessArticle
Genome Mining of the Biocontrol Agent Trichoderma afroharzianum Unearths a Key Gene in the Biosynthesis of Anti-Fungal Volatile Sesquiterpenoids
by
Fang Zhang, Rui Ma, Yuyang Huang, Yang Cui, Qiong Zhou and Jingang Gu
Catalysts 2025, 15(4), 341; https://doi.org/10.3390/catal15040341 - 1 Apr 2025
Abstract
The volatile organic compounds (VOCs) in Trichoderma afroharzianum ACCC 33109 have the biological activities of both hydrolytic enzymes and antimicrobial peptides to mitigate attack by phytopathogens and spread over long distances in soil. However, the biosynthesis pathway of anti-fungal VOCs has not been
[...] Read more.
The volatile organic compounds (VOCs) in Trichoderma afroharzianum ACCC 33109 have the biological activities of both hydrolytic enzymes and antimicrobial peptides to mitigate attack by phytopathogens and spread over long distances in soil. However, the biosynthesis pathway of anti-fungal VOCs has not been elucidated yet. In this study, we identified 15 genes (TaTS1–15) coding for putative terpene synthase with low identities (<79.54%) to functionally characterized homologs through genome mining. Upon Fusarium induction, the relative expression levels of nine TaTS genes were up-regulated by up to 2793-fold (TaTS9). To verify the contribution of TaTS9 to the synthesis of anti-fungal VOCs, the TaTS9 knockout mutant strain was constructed and characterized by its antagonistic activities, transcript profiles, and VOC metabolomes. Heterologous expression of TaTS9 in Escherichia coli produced the target gene product, which converted the precursor farnesyl pyrophosphate (FPP) into β-cubenene (>90%) and γ-amorphene. Thus, TaTS9 was confirmed as the first β-cubenene synthase of Trichoderma, which catalyzes the biosynthesis of various sesquiterpenes with anti-fungal activities. This study provides insight into the key terpene synthase gene in the biosynthesis of anti-fungal sesquiterpenoids for potential applications in the agriculture and food industries.
Full article
(This article belongs to the Section Biocatalysis)
►▼
Show Figures
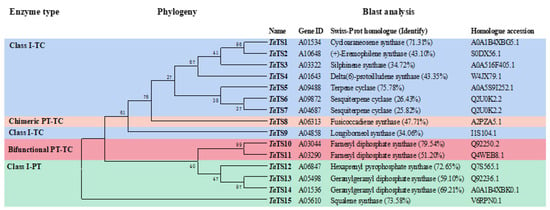
Figure 1
Open AccessArticle
Different Routes for the Hierarchization of *BEA Zeolite, Followed by Impregnation with Niobium and Application in Ethanol and 1-Propanol Dehydration
by
Deborah da Silva Valadares, Willian Henrique Ribeiro de Carvalho, Ana Lívia Fernandes Fonseca, Guilherme de França Machado, Matheus Ramos Silva, Pablo Teles Aragão Campos, José Alves Dias and Sílvia Cláudia Loureiro Dias
Catalysts 2025, 15(4), 340; https://doi.org/10.3390/catal15040340 - 31 Mar 2025
Abstract
This study examined the hierarchical structuring of *BEA zeolite using two distinct approaches: double aluminum removal with solid ammonium hexafluorosilicate (2x-AHFS) and a solution of 0.2 M sodium hydroxide followed by 0.5 M hydrochloric acid (T-NaOH). Additionally, niobium pentoxide (Nb2O5
[...] Read more.
This study examined the hierarchical structuring of *BEA zeolite using two distinct approaches: double aluminum removal with solid ammonium hexafluorosilicate (2x-AHFS) and a solution of 0.2 M sodium hydroxide followed by 0.5 M hydrochloric acid (T-NaOH). Additionally, niobium pentoxide (Nb2O5) was impregnated at different loadings (5, 10, 15, and 20 wt.%) onto the hierarchized materials. Both treatments increased the SiO2/Al2O3 ratio and produced crystals with domains of about the same size. The hierarchization methods generated secondary mesopores and reduced the micropores in the treated HB zeolite. The solid-state NMR analysis by 27Al and 29Si indicated that the 2x-AHFS treatment increased the hydrophobic character of the zeolite, while the treatment with NaOH/HCl resulted in a less hydrophobic material. A balanced quantity of Brønsted and Lewis sites was observed for all treated zeolites. Thus, these combined physicochemical characteristics of the new catalysts may explain their superior performance in the dehydration reactions. In the case of ethanol dehydration at 230 °C, the 20 wt.% Nb2O5 supported on the T-NaOH catalyst produced an 84% conversion and 86% selectivity for ethylene (EE), with 14% diethyl ether (DEE) as the only products. Conversely, in the 1-propanol dehydration reaction, the 20 wt.% Nb2O5 supported on 2x-AHFS achieved 99% conversion, producing 99% propene.
Full article
(This article belongs to the Special Issue Catalysis on Zeolites and Zeolite-Like Materials, 3rd Edition)
►▼
Show Figures
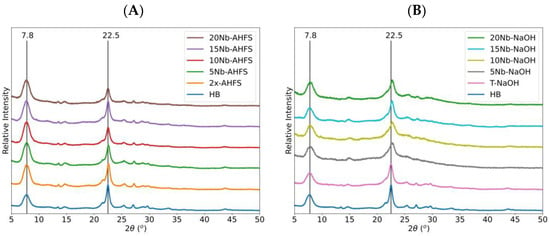
Figure 1
Open AccessArticle
Immobilized Phosphotriesterase as an Enzymatic Resolution for Sofosbuvir Precursor
by
Weerapha Panatdasirisuk, Suthathip Phetlum, Thanat Tiyasakulchai, Nitipol Srimongkolpithak, Tanaporn Uengwetwanit and Nongluck Jaito
Catalysts 2025, 15(4), 339; https://doi.org/10.3390/catal15040339 - 31 Mar 2025
Abstract
The enzymatic resolution of chiral sofosbuvir precursors is a critical step in producing stereoisomerically pure ProTide drugs, essential for their therapeutic efficacy. In this study, a mutated phosphotriesterase (W131M-PTE) was immobilized onto various polymeric macroporous beads, including commercial immobead 150P (IB), modified (IB-EDA
[...] Read more.
The enzymatic resolution of chiral sofosbuvir precursors is a critical step in producing stereoisomerically pure ProTide drugs, essential for their therapeutic efficacy. In this study, a mutated phosphotriesterase (W131M-PTE) was immobilized onto various polymeric macroporous beads, including commercial immobead 150P (IB), modified (IB-EDA and IB-MTD), and synthetic polyacrylamide (PAM) beads functionalized with glutaraldehyde (PAM-GA) or 1-Ethyl-3-(3-dimethyl aminopropyl) carbodiimide (PAM-EDC). The immobilization efficiency, stability, and reusability of the enzyme were systematically evaluated. Among the tested supports, PAM-EDC demonstrated superior performance, retaining high enzymatic activity across multiple cycles and achieving a 92% yield of the (Sp)-diastereomer. The study highlights the potential of immobilized W131M-PTE as a cost-effective and scalable solution for chiral separation in pharmaceutical manufacturing, with implications for broader applications in ProTide drug production.
Full article
(This article belongs to the Topic Green and Sustainable Chemical Processes)
►▼
Show Figures
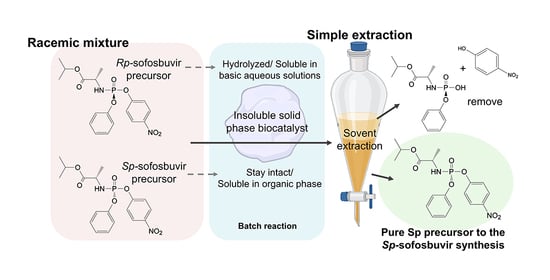
Graphical abstract
Open AccessArticle
Carbon-Encapsulated Ni Nanoparticles Catalysts Derived from Ni-Hexamine Coordination Frameworks for Oxygen Reduction Reaction and Oxygen Evolution Reaction
by
Huoxing Huang, Jiaxing Huang, Guoyu Zhong, Shurui Xu, Hongwei Chen, Xiaobo Fu, Shimin Kang, Junling Tu, Yongxiao Tuo, Wenbo Liao and Baizeng Fang
Catalysts 2025, 15(4), 338; https://doi.org/10.3390/catal15040338 - 31 Mar 2025
Abstract
►▼
Show Figures
Developing efficient bifunctional oxygen reduction (ORR) and oxygen evolution (OER) electrocatalysts is critical for renewable energy technologies. Noble metal catalysts face limitations in cost, scarcity, and bifunctional compatibility. Herein, we report the synthesis of nickel nanoparticles encapsulated in nitrogen-doped carbon nanosheets (Ni@NC-T) via
[...] Read more.
Developing efficient bifunctional oxygen reduction (ORR) and oxygen evolution (OER) electrocatalysts is critical for renewable energy technologies. Noble metal catalysts face limitations in cost, scarcity, and bifunctional compatibility. Herein, we report the synthesis of nickel nanoparticles encapsulated in nitrogen-doped carbon nanosheets (Ni@NC-T) via a solvothermal polymerization and pyrolysis process using a Ni-hexamine coordination framework (NiHMT) as a precursor. The Ni@NC-900 catalyst exhibits superior ORR and OER activity under alkaline conditions, with an ORR performance (half-wave potential = 0.86 V) comparable to commercial Pt/C and an OER overpotential of only 430 mV at 10 mA cm−2. Structural analysis indicates that the hierarchical porous structure and high specific surface area (409 m2 g−1) of Ni@NC-900 facilitate the exposure of active sites and enhance mass transport. The surface-doped nitrogen species, predominantly in the form of pyridinic N and graphitic N, promote electron transfer during the ORR. Furthermore, its application as a bifunctional cathode in rechargeable zinc-air batteries results in a high power density of 137 mW cm−2, surpassing the performance levels of many existing carbon-based bifunctional catalysts. This work highlights a facile strategy for the fabrication of transition metal-based catalysts encapsulated in MOF-derived carbon matrices, with promising potential for energy storage and conversion devices.
Full article
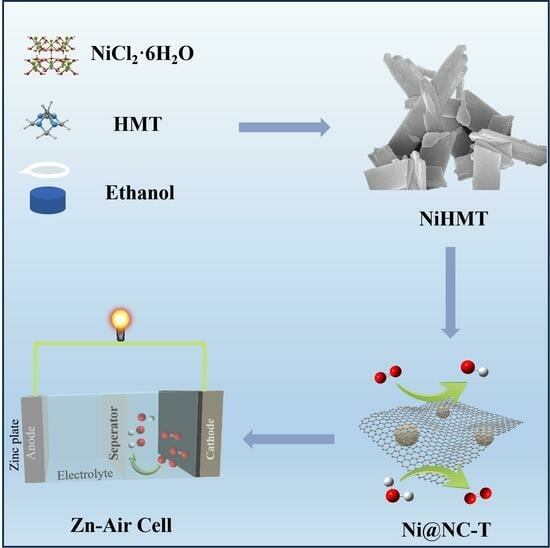
Graphical abstract
Open AccessArticle
Hydrogen Yield from the Partial Oxidation of Methane: Effect of Sc Promoter on Supported Ni/MCM-41 Catalyst
by
Ahmed A. Ibrahim, Omer A. Bellahwel, Fayez M. Al-Alweet, Mabrook S. Amer, Anis H. Fakeeha, Ahmed E. Abasaeed, Abdulaziz S. Bentalib and Ahmed S. Al-Fatesh
Catalysts 2025, 15(4), 337; https://doi.org/10.3390/catal15040337 - 31 Mar 2025
Abstract
This study explores the impact of scandium (Sc) as a promoter on the catalytic performance of 4Ni/MCM-41 catalysts for the partial oxidation of methane (POM). 4Ni+Sc/MCM-41 catalysts were synthesized with varying Sc loadings of 0, 0.2, 0.4, 0.6, and 0.8 wt.%. These catalysts
[...] Read more.
This study explores the impact of scandium (Sc) as a promoter on the catalytic performance of 4Ni/MCM-41 catalysts for the partial oxidation of methane (POM). 4Ni+Sc/MCM-41 catalysts were synthesized with varying Sc loadings of 0, 0.2, 0.4, 0.6, and 0.8 wt.%. These catalysts were characterized using several techniques, including N2 physisorption, X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), Raman spectroscopy, thermogravimetric analysis (TGA), and X-ray photoelectron spectroscopy (XPS). All catalysts exhibited a mesoporous structure characterized by narrow slit-shaped pores. Among them, the 4Ni+0.2Sc/MCM-41 catalyst showed the most consistent pore size distribution. The addition of Sc (scandium) facilitated the formation of strongly interacting nickel species, which enhanced the initial catalytic activity. However, a trade-off was observed between initial activity and long-term stability. The optimal Sc loading was determined to be 0.2 wt.%. This catalyst achieved the highest methane conversion rate of 63.9%, a hydrogen yield of 60%, and an H2/CO ratio of 2.7 while also demonstrating superior stability during extended operation. The 4Ni+0.2Sc/MCM-41 catalyst showed only a 7% weight loss in the thermogravimetric analysis (TGA), which shows that it will stay stable even after being used for a long time. The improved performance of the Sc-promoted catalysts is attributable to the increased availability of active sites, enhanced stability, and better dispersion of nickel. These efforts aim to create more sustainable and efficient methods for hydrogen production, minimizing the negative effects associated with traditional processes. By advancing these technologies, we can further support the transition to a cleaner energy future.
Full article
(This article belongs to the Section Industrial Catalysis)
►▼
Show Figures
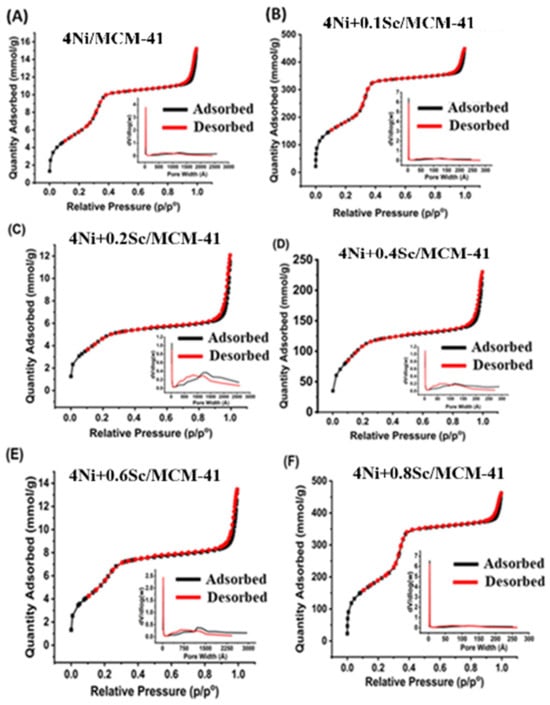
Figure 1
Open AccessReview
Application of Graphene-Based Solar Driven Interfacial Evaporation-Coupled Photocatalysis in Water Treatment
by
Yining Zhang, Huiqin Wang and Jisheng Zhang
Catalysts 2025, 15(4), 336; https://doi.org/10.3390/catal15040336 - 31 Mar 2025
Abstract
The global shortage of freshwater resources and the energy crisis have propelled solar-driven interfacial evaporation (SDIE) coupled with photocatalytic technology to become a research focus in efficient and low-carbon water treatment. Graphene-based materials demonstrate unique advantages in SDIE–photocatalysis integrated systems, owing to their
[...] Read more.
The global shortage of freshwater resources and the energy crisis have propelled solar-driven interfacial evaporation (SDIE) coupled with photocatalytic technology to become a research focus in efficient and low-carbon water treatment. Graphene-based materials demonstrate unique advantages in SDIE–photocatalysis integrated systems, owing to their broadband light absorption, ultrafast thermal carrier dynamics, tunable electronic structure, and low evaporation enthalpy characteristics. This review systematically investigates the enhancement mechanisms of graphene photothermal conversion on photocatalytic processes, including (1) improving light absorption through surface morphology modulation, defect engineering, and plasmonic material compositing; (2) reducing water evaporation enthalpy via hydrophilic functional group modification and porous structure design; (3) suppressing heat loss through thermal insulation layers and 3D structural optimization; and (4) enhancing water transport efficiency via fluid channel engineering and wettability control. Furthermore, salt resistance strategies and structural optimization significantly improve system practicality and stability. In water treatment applications, graphene-based SDIE systems achieve synergistic “adsorption–catalysis–evaporation” effects, enabling efficient the degradation of organic pollutants, reduction in/fixation of heavy metal ions, and microbial inactivation. However, practical implementation still faces challenges including low steam condensation efficiency, insufficient long-term material durability, and high scaling-up costs. Future research should prioritize enhancing heat and mass transfer in condensation systems, optimizing material environmental adaptability, and developing low-cost manufacturing processes to promote widespread application of graphene-based SDIE–photocatalysis integrated systems.
Full article
(This article belongs to the Special Issue Mineral-Based Composite Catalytic Materials)
►▼
Show Figures

Figure 1
Open AccessArticle
Hydrocracking of Polyethylene to Gasoline-Range Hydrocarbons over a Ruthenium-Zeolite Bifunctional Catalyst System with Optimal Synergy of Metal and Acid Sites
by
Qing Du, Xin Shang, Yangyang Yuan, Xiong Su and Yanqiang Huang
Catalysts 2025, 15(4), 335; https://doi.org/10.3390/catal15040335 - 31 Mar 2025
Abstract
Chemical recycling of plastic waste, especially polyolefins, into valuable liquid fuels is of considerable significance to address the serious issues raised by their threat on environmental and human health. Nevertheless, the construction of efficient and economically viable catalytic systems remains a significant hurdle.
[...] Read more.
Chemical recycling of plastic waste, especially polyolefins, into valuable liquid fuels is of considerable significance to address the serious issues raised by their threat on environmental and human health. Nevertheless, the construction of efficient and economically viable catalytic systems remains a significant hurdle. Herein, we developed an efficient bifunctional catalyst system comprising γ-Al2O3-supported ruthenium nanoparticles (Ru/γ-Al2O3) and β-zeolite for the conversion of polyolefins into gasoline-range hydrocarbons. A yield of C5–12 paraffins up to 73.4% can be obtained with polyethene as the reactant at 250 °C in hydrogen. The Ru sites primarily activate the initial cleavage of C–H bonds of polymer towards the formation of olefin intermediates, which subsequently go through further cracking and isomerization over the acid sites in β-zeolite. Employing in situ infrared spectroscopy and probe–molecule model reactions, our investigation reveals that the optimized proportion and spatial distribution of the dual catalytic sites are pivotal in the tandem conversion process. This optimization synergistically regulates the cracking kinetics and accelerates intermediate transfer, thereby minimizing the production of side C1–4 hydrocarbons resulting from over-cracking at the Ru sites and enhancing the yield of liquid fuels. This research contributes novel insights into catalyst design for the chemical upgrading of polyolefins into valuable chemicals, advancing the field of plastic waste recycling and sustainable chemical production.
Full article
(This article belongs to the Special Issue Zeolites as Catalysts: Applications in Chemical Engineering, Energy Sources and Environmental Protection, 2nd Edition)
►▼
Show Figures

Graphical abstract
Open AccessFeature PaperArticle
Enhanced H2 Production Efficiency in Photo-Reforming of PET Waste Plastic Using Dark-Deposited Atom/Nanocomposite Pt/TiO2 Photocatalysts
by
E. M. N. Thiloka Edirisooriya, Punhasa S. Senanayake, Pei Xu and Huiyao Wang
Catalysts 2025, 15(4), 334; https://doi.org/10.3390/catal15040334 - 31 Mar 2025
Abstract
Photo-reforming waste polyethylene terephthalate (PET) in alkaline aqueous solutions is a novel approach for green hydrogen production. This study focuses on improving the catalytic efficiency of Pt/TiO2 for the photo-reforming of waste PET using an innovative dark deposition method to deposit Pt
[...] Read more.
Photo-reforming waste polyethylene terephthalate (PET) in alkaline aqueous solutions is a novel approach for green hydrogen production. This study focuses on improving the catalytic efficiency of Pt/TiO2 for the photo-reforming of waste PET using an innovative dark deposition method to deposit Pt single atoms on nano TiO2 (Pt/TiO2), thereby increasing the catalytic efficiency while reducing the cost of the catalyst. The precursor concentration was optimized to control the size and distribution of the Pt clusters/atoms, and the TiO2 support was annealed at different temperatures to modify the properties of Pt/TiO2. Nine Pt/TiO2 catalysts were synthesized using different Pt precursor concentrations and annealing temperatures. The catalysts were characterized to measure their morphological, crystalline, and electronic properties, as well as their hydrogen yields via PET photo-reforming. The hydrogen conversion efficiency and external quantum yield (EQY) were calculated and compared with those of traditional direct-deposited catalysts. The correlation between the different characteristics of the dark-deposited and direct-deposited catalysts and their influence on the hydrogen yield in the photo-reforming process was statistically analyzed using principal component analysis. Catalysts deposited under dark conditions exhibited 5-fold and 7-fold enhancements in hydrogen conversion efficiency and EQY, respectively, compared to conventional catalytic systems. These findings indicate that the proposed catalytic system provides a viable solution for minimizing Pt loading, reducing the cost of the catalyst, and maintaining a higher hydrogen conversion efficiency.
Full article
(This article belongs to the Special Issue Advances in Photocatalytic Degradation)
►▼
Show Figures

Figure 1
Open AccessArticle
Electro-Refinery in Organics to Produce Energy Carriers: Co-Generation of Green Hydrogen and Carboxylic Acids by Glycerol Electrooxidation Using Dimensionally Stable Anode
by
Letícia M. G. da Silva, Letícia G. A. Costa, José E. L. Santos, Emily C. T. de A. Costa, Aruzza M. de Morais Araújo, Amanda D. Gondim, Lívia N. Cavalcanti, Marco A. Quiroz, Elisama V. dos Santos and Carlos A. Martínez-Huitle
Catalysts 2025, 15(4), 333; https://doi.org/10.3390/catal15040333 - 31 Mar 2025
Abstract
The urgency to decarbonize fuels has contributed to a rise in biofuel production, which has culminated in a significant increase in the waste quantity of glycerol produced. Therefore, to convert glycerol waste into high-value products, electrochemical oxidation (EO) is a viable alternative for
[...] Read more.
The urgency to decarbonize fuels has contributed to a rise in biofuel production, which has culminated in a significant increase in the waste quantity of glycerol produced. Therefore, to convert glycerol waste into high-value products, electrochemical oxidation (EO) is a viable alternative for the co-generation of carboxylic acids, such as formic acid (FA) and green hydrogen (H2), which are considered energy carriers. The aim of this study is the electroconversion of glycerol into FA by EO using a divided electrochemical cell, driven by a photovoltaic (PV) system, with a dimensionally stable anode (DSA, Ti/TiO2-RuO2-IrO2) electrode as an anode and Ni-Fe stainless steel (SS) mesh as a cathode. To optimize the experimental conditions, studies were carried out evaluating the effects of applied current density (j), electrolyte concentration, electrolysis time, and electrochemical cell configuration (undivided and divided). According to the results, the optimum experimental conditions were achieved at 90 mA cm−2, 0.1 mol L−1 of Na2SO4 as a supporting electrolyte, and 480 min of electrolysis. In this condition, 256.21 and 211.17 mg L−1 of FA were obtained for the undivided and divided cells, respectively, while the co-generation of 6.77 L of dry H2 was achieved in the divided cell. The electroconversion process under the optimum conditions was also carried out with a real sample, where organic acids like formic and acetic acids were co-produced simultaneously with green H2. Based on the preliminary economic analysis, the integrated-hybrid process is an economically viable and promising alternative when it is integrated with renewable energy sources such as solar energy.
Full article
(This article belongs to the Special Issue Photocatalyzed and Electrochemical Processes for a Cleaner Environment)
►▼
Show Figures
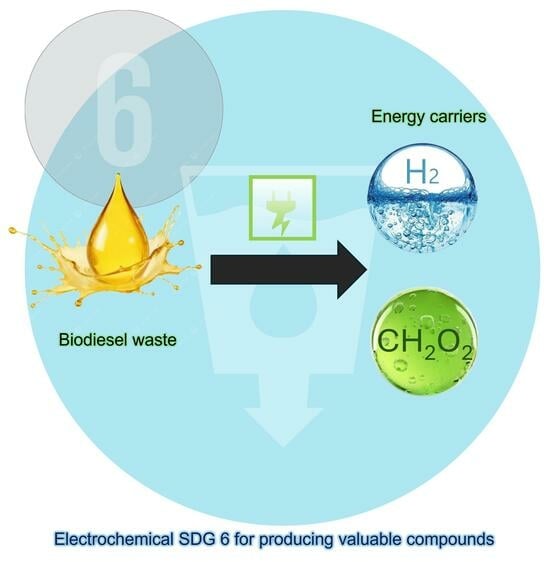
Graphical abstract

Journal Menu
► ▼ Journal Menu-
- Catalysts Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Applied Nano, Catalysts, Materials, Nanomaterials, Polymers, Molecules
Application of Nanomaterials in Environmental Analysis
Topic Editors: Yonggang Zhao, Yun ZhangDeadline: 30 September 2025
Topic in
Energies, Materials, Catalysts, Metals, Hydrogen
Hydrogen—The New Energy Vector for the Transition of Industries "Hard to Abate"
Topic Editors: Pasquale Cavaliere, Geoffrey BrooksDeadline: 20 October 2025
Topic in
Applied Nano, Catalysts, Molecules, Nanomaterials, Water, Gels, Polymers
Water Purification and Catalytic Disintegration at the Nanoscale
Topic Editors: Michael Arkas, Ioannis Pashalidis, Dimitrios A. Giannakoudakis, Ioannis P. AnastopoulosDeadline: 30 November 2025
Topic in
Energies, Materials, Catalysts, Processes, Biomass
Advances in Biomass Conversion, 2nd Edition
Topic Editors: Jacek Grams, Agnieszka RuppertDeadline: 20 December 2025

Conferences
Special Issues
Special Issue in
Catalysts
Environmentally Friendly Catalysts for Energy and Water Treatment Applications
Guest Editors: José Ignacio Lombraña, Hugo Olvera Vargas, Sandra Yazmin Arzate SalgadoDeadline: 5 April 2025
Special Issue in
Catalysts
Catalytic Effects in Electrochemical Energy Storage
Guest Editors: Bingsen Zhang, Ming Lu, Qinhua GuDeadline: 10 April 2025
Special Issue in
Catalysts
Nanomaterials in Environmental Catalysis
Guest Editors: Lixin Li, Lijie Xu, Langming Bai, Xin RenDeadline: 15 April 2025
Special Issue in
Catalysts
The State of the Art and Future Perspectives on Computational Catalysis
Guest Editors: Marta Castiñeira Reis, Carlo NerviDeadline: 15 April 2025
Topical Collections
Topical Collection in
Catalysts
Catalytic Conversion and Utilization of Carbon-Based Energy
Collection Editor: Jian Qi
Topical Collection in
Catalysts
Catalytic Conversion of Biomass to Bioenergy
Collection Editors: Sergio Nogales Delgado, Juan Félix González, Simona M. Coman
Topical Collection in
Catalysts
Nanocatalysis Towards Energy Transition and Environmental Sustainability
Collection Editors: Michalis Konsolakis, Sónia Carabineiro
Topical Collection in
Catalysts
Catalysis in Advanced Oxidation Processes for Pollution Control
Collection Editors: Rongfu Huang, Fei Chang









