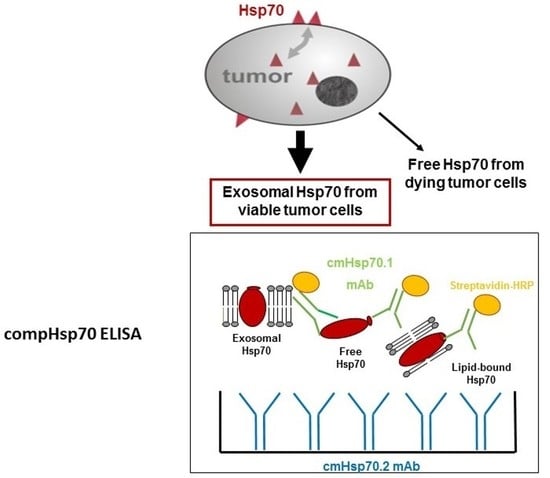
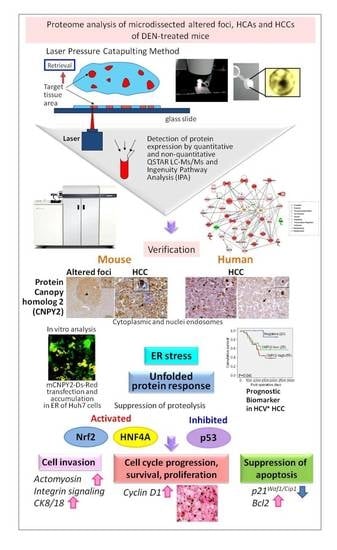
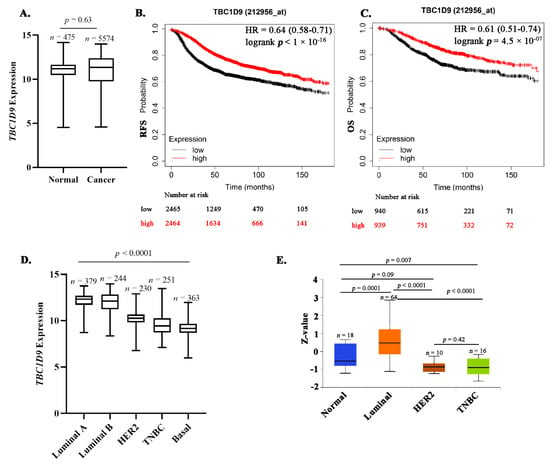
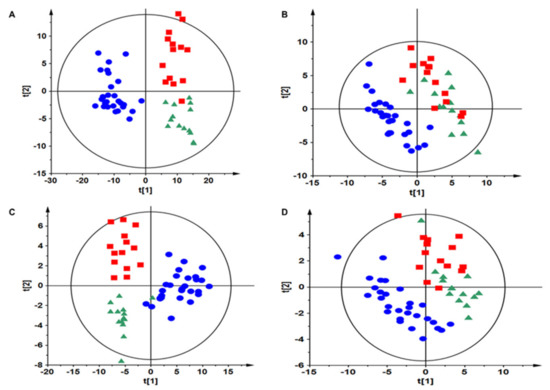
Novel Biomarkers and Molecular Targets in Cancer
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Molecular Cancer Biology".
Submission Status: Closed | Viewed by 203035Editor
Interests: gastrointestinal cancer; diagnosis; biomarker; anti-cancer drug; molecular mechanisms
Special Issues, Collections and Topics in MDPI journals
Topical Collection Information
Dear Colleagues,
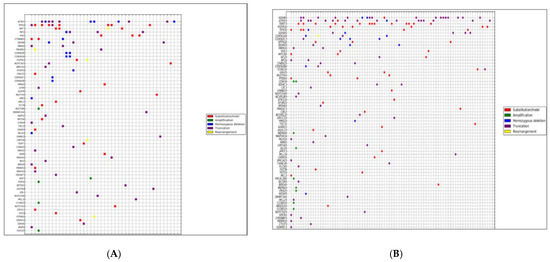
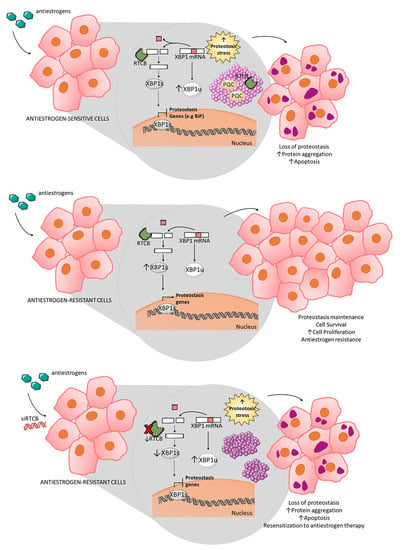
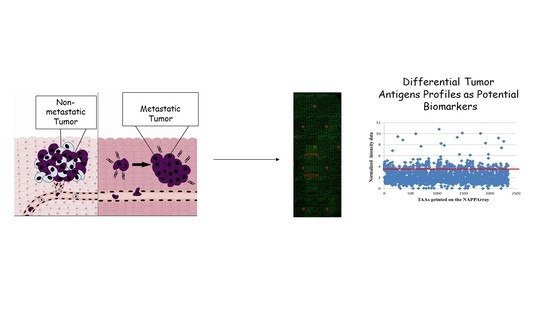
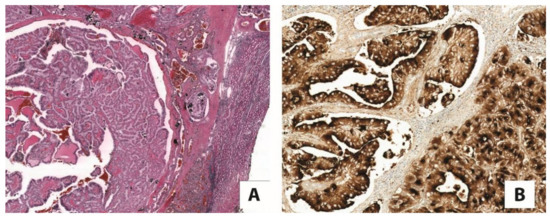
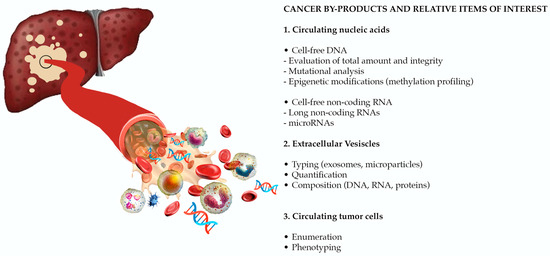
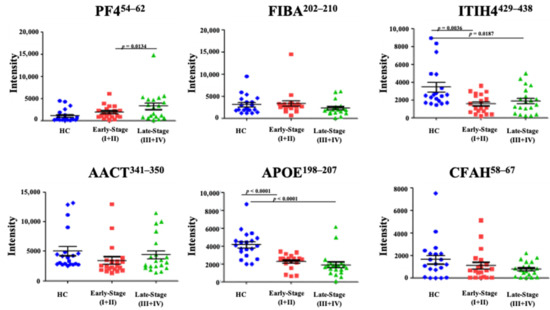
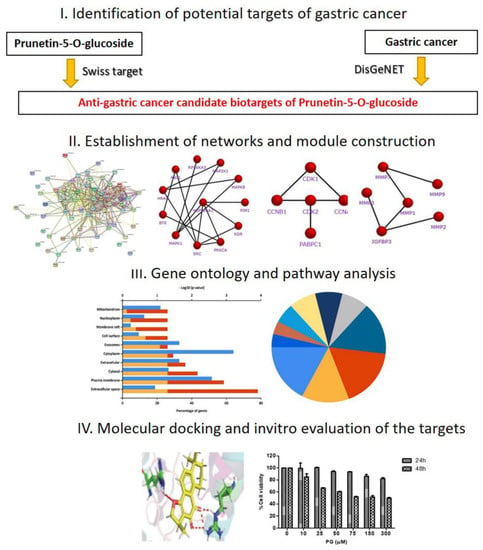
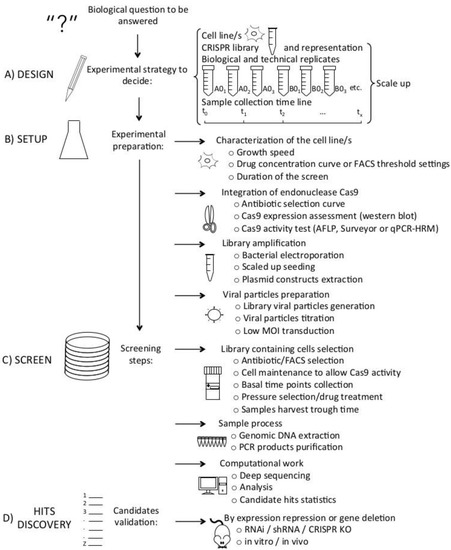
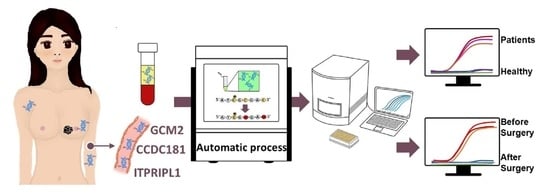
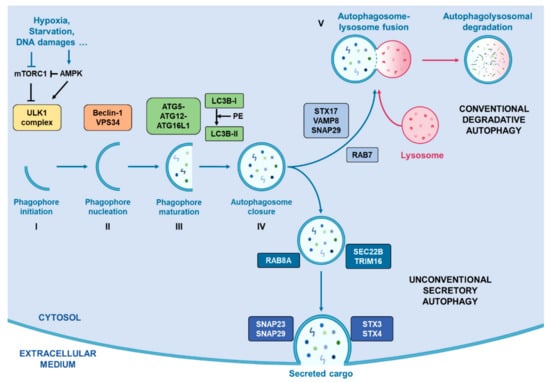
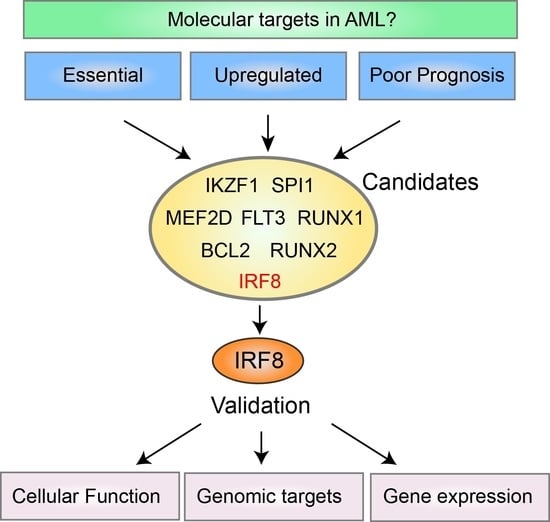
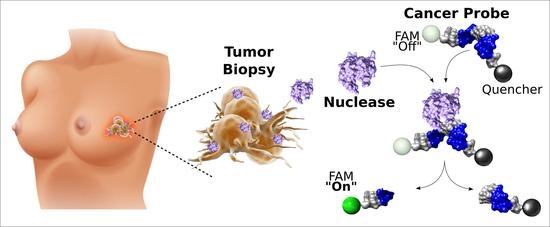
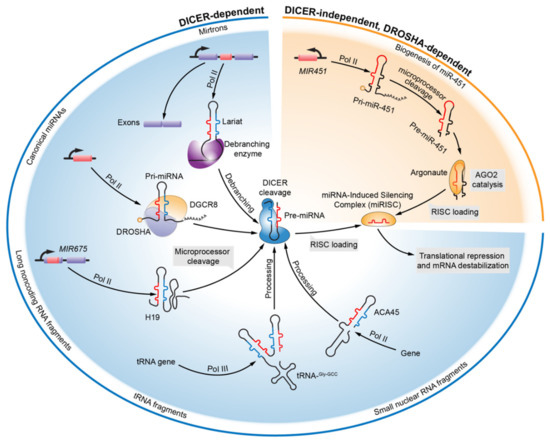
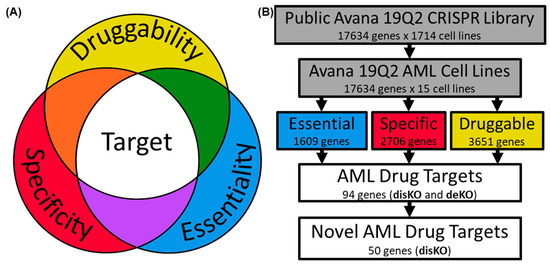
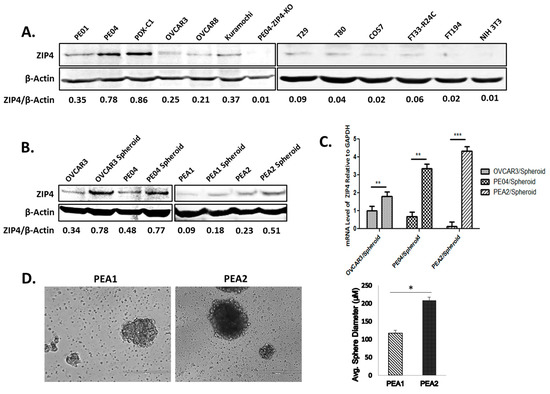
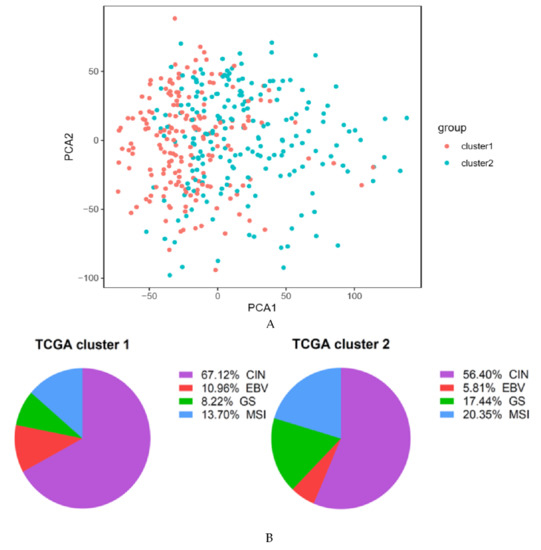
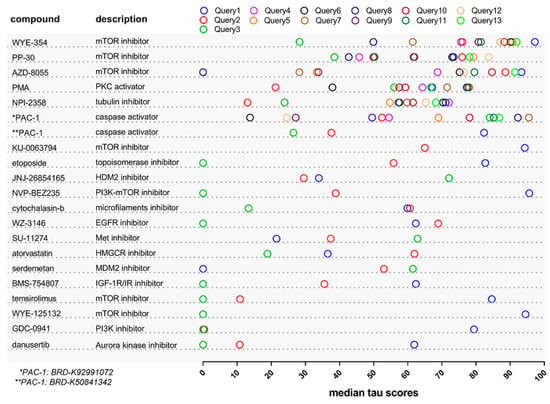
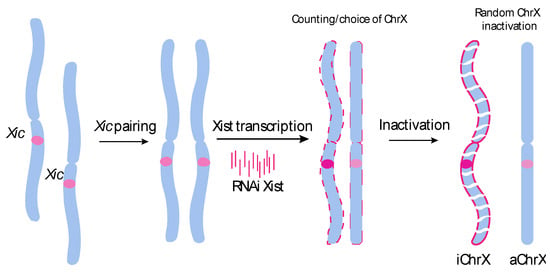
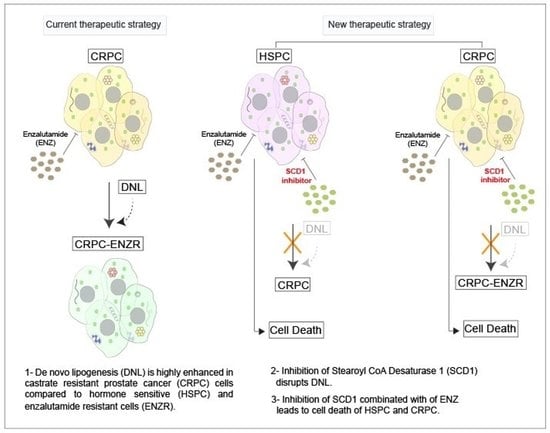
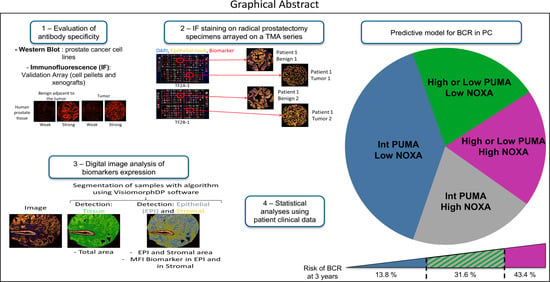
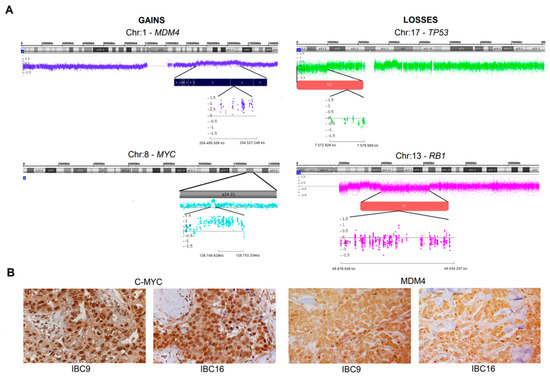
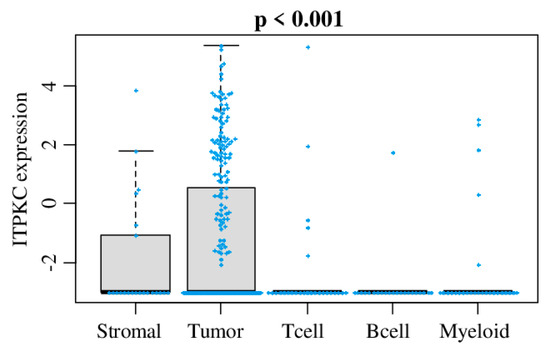
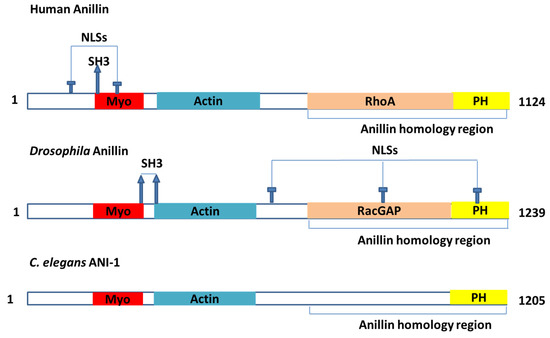
Today, the discovery of novel biomarkers and molecular targets to fight cancer remains the matter of great importance. Recent novel techniques, including cancer mutational analysis, genomics, proteomics and metabolomics, have given rise to new efforts in performing pathological, biochemical, molecular, and immunological research to study the mechanisms of carcinogenesis, with the final aim of finding the cancer molecular targets, which could be applied in clinical practice. This Collection aims to present the most recent studies related to the discovery of novel biomarkers and molecular targets in various cancers. The research could be performed in different aspects, including: 1) genetic alterations in cancer, 2) diagnosis using novel techniques, 3) cancer molecular mechanisms, 4) in vitro and in vivo carcinogenesis models for the discovery of cancer risk factors and innovative drugs to treat cancer, 5) cancer metabolic and proteomic signatures and altered pathways, 6) stem, stromal and immune cell biomarkers in cancer, and 7) preclinical innovative studies.
Dr. Anna Kakehashi
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- cancer
- biomarker
- molecular target
- diagnosis
- molecular mechanisms